 Back
BackCarbon and the Molecular Diversity of Life: Study Notes
Study Guide - Smart Notes

Carbon and Molecular Diversity
Carbon’s Electron Configuration and Covalent Bonding
Carbon is unique among elements due to its electron configuration, which allows it to form four covalent bonds. This property enables carbon to act as a versatile backbone for a vast array of molecular structures essential to life.
Electron Configuration: Carbon has the electron configuration 1s2 2s2 2p2, with four valence electrons available for bonding.
Tetrahedral Geometry: When carbon forms four single covalent bonds, the atoms are arranged at the corners of a tetrahedron with bond angles of 109.5°.
Bonding Diversity: Carbon can bond with many elements, including hydrogen, oxygen, nitrogen, sulfur, and phosphorus, forming diverse organic compounds.
Example: Methane (CH4) is a simple molecule where carbon forms four single bonds with hydrogen.
Variation in Carbon Skeletons
The diversity of organic molecules arises from variations in the carbon skeleton, including differences in length, branching, double bonds, and ring structures.
Length: Carbon chains can vary in length, forming molecules from a few to many carbon atoms.
Branching: Carbon chains may be unbranched or branched, affecting molecular properties.
Double Bonds: The presence and position of double bonds influence molecular shape and reactivity.
Rings: Carbon atoms can form ring structures, such as cyclohexane.
Example: Butane and 2-methylpropane (isobutane) are structural isomers with different branching.
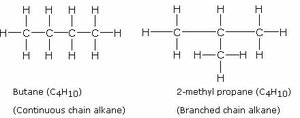
Isomers and Molecular Diversity
Isomers are compounds with the same molecular formula but different structures, leading to distinct chemical properties and biological functions.
Structural Isomers: Differ in the covalent arrangement of atoms.
Cis-Trans Isomers: Differ in spatial arrangement around a double bond.
Enantiomers: Are mirror images of each other, differing in spatial arrangement around an asymmetric carbon.
Biological Significance: Enantiomers can have dramatically different effects in biological systems (e.g., L- and D-amino acids).
Example: The molecules shown above are structural isomers; they have different molecular structures but the same molecular formula.

The Seven Major Functional Groups
Functional groups are specific groups of atoms within molecules that confer characteristic chemical properties. They are critical in determining solubility, acidity, basicity, and reactivity.
Hydroxyl (-OH): Found in alcohols; increases solubility in water.
Carbonyl (C=O): Found in aldehydes and ketones; affects reactivity.
Carboxyl (-COOH): Found in organic acids; acts as an acid.
Amino (-NH2): Found in amines; acts as a base.
Sulfhydryl (-SH): Found in thiols; forms disulfide bonds in proteins.
Phosphate (-PO4): Found in nucleotides; contributes negative charge and energy transfer.
Methyl (-CH3): Found in many compounds; affects gene expression and hydrophobicity.
Interpreting Structural Formulas and Functional Groups
Recognizing functional groups and asymmetric carbons in structural formulas is essential for predicting molecular behavior and biological activity.
Functional Groups: Identify groups such as hydroxyl, carbonyl, carboxyl, amino, etc., in molecular diagrams.
Asymmetric Carbon: A carbon atom attached to four different groups; key for enantiomerism.
Example: The molecule below contains amino, carboxyl, and carbonyl groups.
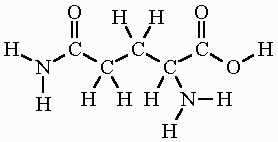
Hydrocarbons and Functionalized Hydrocarbons
Hydrocarbons consist of only carbon and hydrogen. Their nonpolar C–H bonds make them hydrophobic and excellent for energy storage.
Energy Storage: Hydrocarbons release energy when oxidized.
Hydrophobicity: Nonpolar C–H bonds cause hydrocarbons to repel water.
Functionalization: Addition of functional groups increases reactivity and solubility.
Biological Macromolecules and Carbon Chemistry
The structure and function of biological macromolecules—carbohydrates, lipids, proteins, and nucleic acids—are determined by carbon skeletons and functional groups.
Carbohydrates: Contain hydroxyl and carbonyl groups; provide energy and structural support.
Lipids: Mostly hydrocarbons; store energy and form membranes.
Proteins: Contain amino and carboxyl groups; perform diverse functions.
Nucleic Acids: Contain phosphate, hydroxyl, and amino groups; store genetic information.
Summary Table: Functional Groups and Their Properties
Functional Group | Structure | Properties | Example |
|---|---|---|---|
Hydroxyl | -OH | Polar, increases solubility | Alcohols |
Carbonyl | C=O | Polar, reactive | Aldehydes, Ketones |
Carboxyl | -COOH | Acidic, donates H+ | Acetic acid |
Amino | -NH2 | Basic, accepts H+ | Amines |
Sulfhydryl | -SH | Forms disulfide bonds | Thiols |
Phosphate | -PO4 | Negative charge, energy transfer | ATP |
Methyl | -CH3 | Nonpolar, affects expression | Methylated DNA |
Key Equations
Lewis Dot Structure of Carbon:
General Formula for Alkanes:
Additional info: The notes above expand on brief points and fill in missing context to provide a comprehensive overview of carbon chemistry and its role in biological molecular diversity.
