 Back
BackCatalysts and Enzyme Function in Biological Systems
Study Guide - Smart Notes

Catalysts and Activation Energy
Introduction to Catalysts
Catalysts are substances that increase the rate of a chemical reaction without being consumed in the process. In biological systems, most catalysts are enzymes, which are typically proteins. Catalysts function by lowering the activation energy required for a reaction to proceed, making it easier for reactants to be converted into products.
Activation Energy (Ea): The minimum energy required to initiate a chemical reaction.
Enzyme: A biological catalyst, usually a protein, that speeds up specific biochemical reactions.
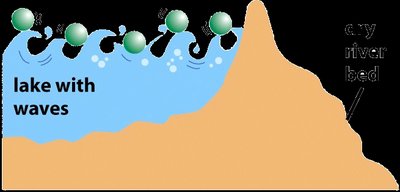
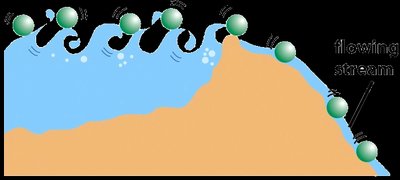
Energy Diagrams and the Effect of Catalysts
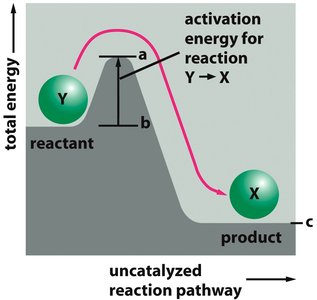
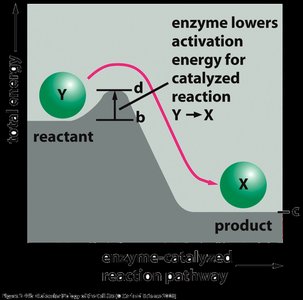
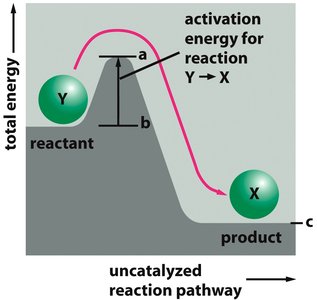
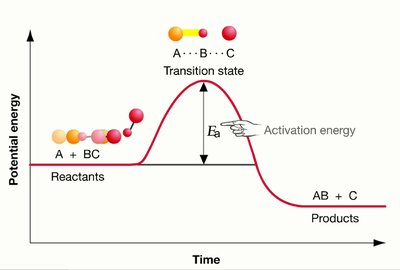
Energy diagrams illustrate the energy changes during a reaction. The difference in energy between reactants and products is the Gibbs free energy change (ΔG), while the peak represents the activation energy barrier. Catalysts lower this barrier, allowing more molecules to react at a given temperature.
ΔG (Gibbs Free Energy): The overall energy change of a reaction; determines if a reaction is spontaneous.
Uncatalyzed vs. Catalyzed Pathways: The catalyzed pathway has a lower activation energy but the same ΔG as the uncatalyzed pathway.
Enzyme Efficiency
Enzymes can increase reaction rates by factors of millions or more, making biological processes feasible under physiological conditions.
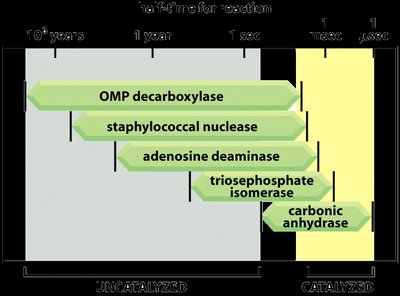
Enzyme Structure and Function
Protein Folding and Active Sites
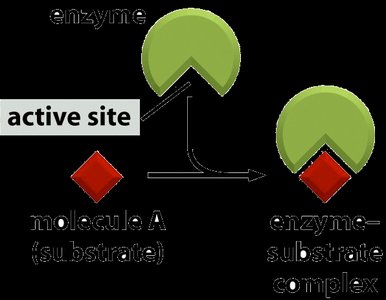
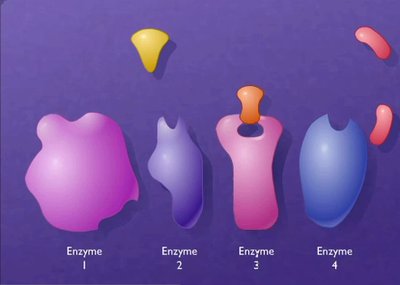
Enzymes are proteins that must fold into specific three-dimensional shapes to function. The active site is a region where substrates bind and reactions are catalyzed. Protein folding brings together amino acid side chains to form the active site.
Active Site: The region of an enzyme where substrate molecules bind and undergo a chemical reaction.
Protein Folding: The process by which a protein assumes its functional shape.
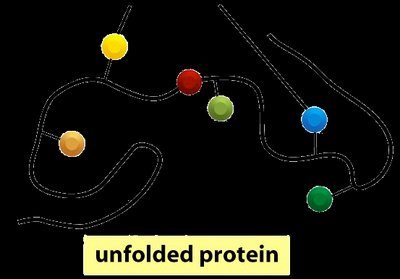
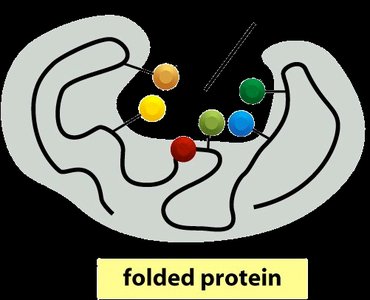
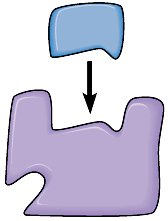
Enzyme-Substrate Interactions
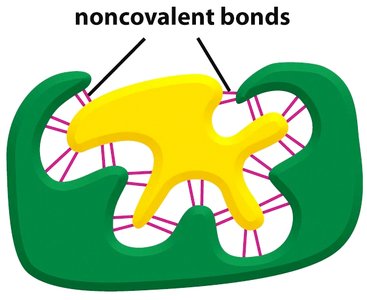
Enzyme-substrate binding is highly specific, often involving noncovalent interactions such as hydrogen bonds, ionic bonds, and van der Waals forces. The substrate fits into the enzyme's active site, forming an enzyme-substrate complex.
Noncovalent Interactions: Weak chemical bonds that help stabilize the enzyme-substrate complex.
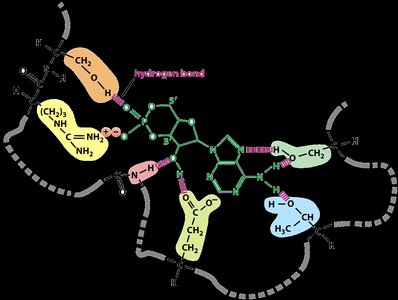
Mechanism of Enzyme Action
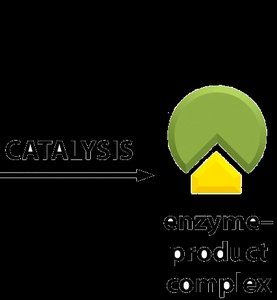
Enzymes function by binding substrates, facilitating the conversion to products, and releasing the products while remaining unchanged. The process involves the formation of a transition state, which is the highest energy and most unstable state during the reaction.
Transition State: A high-energy, unstable state that reactants must pass through to become products.

Coupling Reactions and High-Energy Intermediates
Reaction Coupling
Many cellular reactions are energetically unfavorable (positive ΔG) and must be coupled to favorable reactions (negative ΔG) to proceed. This is often achieved using high-energy intermediates such as ATP.
Coupled Reactions: The energy released from an exergonic reaction is used to drive an endergonic reaction.
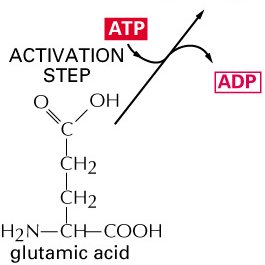
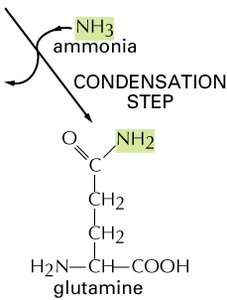
Example: Glutamine Biosynthesis
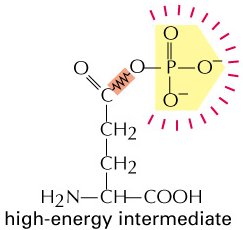
The synthesis of glutamine from glutamate and ammonia is an endergonic process, but it is driven by coupling to ATP hydrolysis, which is highly exergonic.
Glutamine Synthetase: The enzyme that catalyzes the formation of glutamine from glutamate and ammonia.

Free Energy and Reaction Progress
Cells use high-energy intermediates to ensure that the overall ΔG of coupled reactions is negative, making the process spontaneous.
ATP Hydrolysis:
Glutamine Synthesis:
Overall Coupled Reaction:
Enzyme Kinetics and Regulation
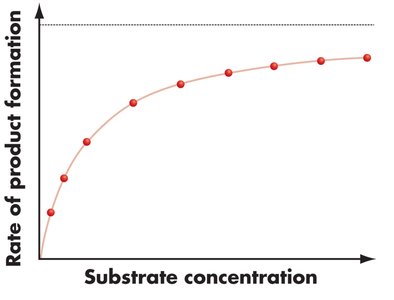
Substrate Concentration and Enzyme Activity
The rate of an enzyme-catalyzed reaction increases with substrate concentration until it reaches a maximum rate (Vmax), where all enzyme active sites are saturated.
Vmax: The maximum rate of an enzyme-catalyzed reaction when the enzyme is saturated with substrate.
Environmental Effects on Enzyme Activity
Enzyme activity is sensitive to environmental factors such as temperature, pH, and ionic strength. Each enzyme has optimal conditions under which it functions most efficiently.
Optimal Conditions: The specific set of environmental parameters where an enzyme's activity is maximal.

Enzyme Inhibition and Regulation
Types of Enzyme Inhibitors
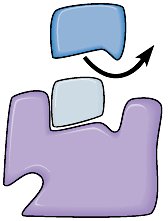
Enzyme inhibitors are molecules that decrease or abolish enzyme activity. They can be classified as competitive or noncompetitive inhibitors.
Competitive Inhibitors: Molecules that resemble the substrate and bind to the active site, preventing substrate binding.
Noncompetitive Inhibitors: Molecules that bind to a site other than the active site (allosteric site), altering enzyme function.
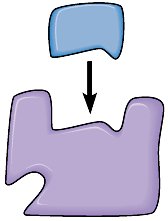
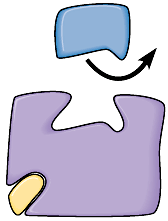
Example: Sulfa Drugs as Competitive Inhibitors
Sulfanilamide is a competitive inhibitor of the enzyme that converts PABA into folic acid in bacteria, blocking nucleic acid synthesis and thus bacterial growth.
Biochemical Pathways and Feedback Regulation
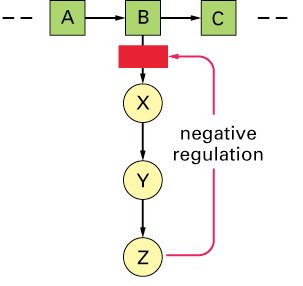
Enzymes often function in metabolic pathways, where the product of one reaction becomes the substrate for the next. Feedback inhibition is a regulatory mechanism in which the end product of a pathway inhibits an earlier step, preventing overproduction.
Feedback Inhibition: The process by which the accumulation of an end product inhibits an enzyme involved in its synthesis.
Additional info: Enzyme regulation is essential for maintaining homeostasis and efficient cellular function. Allosteric regulation, covalent modification, and gene expression control are other mechanisms by which cells fine-tune enzyme activity.
