 Back
BackCellular Respiration and Fermentation: Mechanisms and Pathways
Study Guide - Smart Notes

Cellular Respiration and Fermentation
Overview: Life Is Work
Living cells require energy to perform essential functions, which they obtain from external sources. Animals acquire energy by consuming other organisms, while photosynthetic organisms such as plants and algae capture energy from sunlight. Cellular respiration is the process by which cells extract energy from organic molecules to regenerate ATP, the universal energy currency.

Energy Flow and Chemical Recycling in Ecosystems
Energy enters ecosystems as sunlight and exits as heat. Photosynthesis in chloroplasts produces oxygen and organic molecules, which serve as fuel for cellular respiration in mitochondria. Cellular respiration uses these molecules to generate ATP, powering most cellular work.

Catabolic Pathways Yield Energy by Oxidizing Organic Fuels
Catabolic pathways break down organic molecules, releasing energy. Aerobic respiration uses oxygen as the final electron acceptor, while anaerobic respiration uses other molecules. Fermentation is a partial degradation of sugars that occurs without oxygen.
Redox Reactions: Oxidation and Reduction
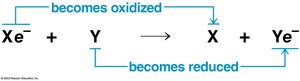
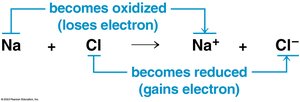
Cellular respiration involves redox reactions, where electrons are transferred between molecules. Oxidation is the loss of electrons, and reduction is the gain of electrons. These reactions release energy stored in organic molecules, which is used to synthesize ATP.
Oxidation of Organic Fuel Molecules During Cellular Respiration
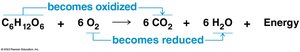
During cellular respiration, fuel molecules such as glucose are oxidized, and oxygen is reduced. Organic molecules rich in hydrogen, like carbohydrates and fats, are excellent fuels. The transfer of hydrogen to oxygen releases energy for ATP synthesis.
Harvesting Energy via NAD+ and Electron Transport Chain
Electrons from glucose are transferred to NAD+, a coenzyme that acts as an oxidizing agent. The reduced form, NADH, stores energy for ATP synthesis. Enzymes called dehydrogenases facilitate this transfer.

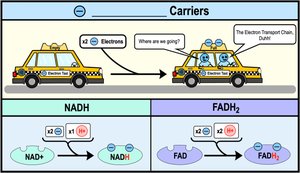
Stepwise Energy Harvest via NAD+ and Electron Transport Chain
NADH passes electrons to the electron transport chain (ETC), where electrons are transferred through a series of redox reactions to increasingly electronegative carriers, ultimately to oxygen. This stepwise transfer releases energy in manageable amounts for ATP regeneration.
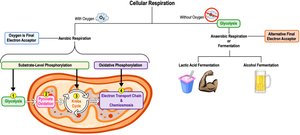
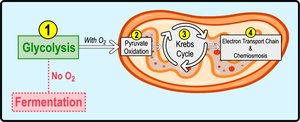
Stages of Cellular Respiration
Cellular respiration consists of three main stages:
Glycolysis: Breaks down glucose into two pyruvate molecules in the cytosol.
Pyruvate Oxidation and Citric Acid Cycle: Completes glucose breakdown in the mitochondrial matrix.
Oxidative Phosphorylation: Accounts for most ATP synthesis (~90%) in the inner mitochondrial membrane.

Types of Phosphorylation
Substrate-level phosphorylation: Direct transfer of a phosphate group to ADP by an enzyme, occurs in glycolysis and the citric acid cycle.
Oxidative phosphorylation: Uses energy from the ETC to phosphorylate ADP, occurs in ETC and chemiosmosis.
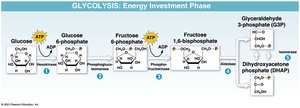
Glycolysis
Glycolysis is the first stage of cellular respiration, breaking down glucose into two pyruvate molecules. It occurs in the cytoplasm and consists of two phases: energy investment and energy payoff. The net yield is 2 ATP and 2 NADH per glucose molecule.
Pyruvate Oxidation
If oxygen is present, pyruvate enters the mitochondrion, where it is converted to acetyl CoA and CO2 is released. This step yields 1 NADH per pyruvate (2 NADH per glucose).
Citric Acid Cycle (Krebs Cycle)
The citric acid cycle completes the breakdown of pyruvate to CO2. Each turn of the cycle oxidizes molecules derived from one pyruvate, and the cycle turns twice per glucose molecule, generating 2 ATP, 6 NADH, and 2 FADH2.
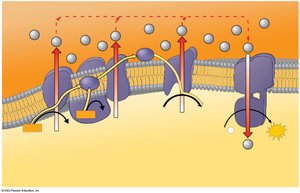
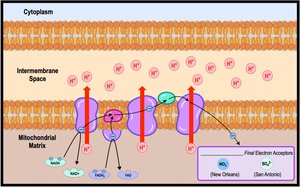
Electron Transport Chain
The ETC is located in the inner mitochondrial membrane and consists of protein complexes (I-IV) and cytochromes. Electrons are transferred through these complexes, dropping in free energy and releasing energy for ATP synthesis. Water is formed when oxygen is reduced.
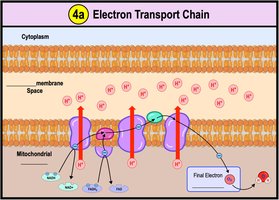
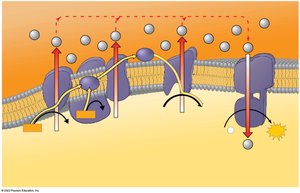
Chemiosmosis: Energy-Coupling Mechanism
Energy released by the ETC is used to pump H+ ions into the intermembrane space, creating a gradient. H+ diffuses back into the mitochondrial matrix through ATP synthase, driving ATP synthesis. This process is called chemiosmosis.
ATP Production by Cellular Respiration
Most ATP is produced by oxidative phosphorylation. Approximately 34% of the energy in glucose is transferred to ATP (~32 ATP per glucose), with the remainder lost as heat.
Fermentation and Anaerobic Respiration
Without oxygen, cells generate ATP through anaerobic respiration or fermentation. Anaerobic respiration uses an alternative final electron acceptor, such as sulfate, while fermentation allows continuous ATP production by substrate-level phosphorylation.
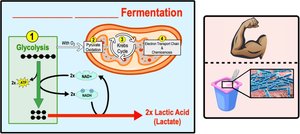
Types of Fermentation
Fermentation consists of glycolysis plus reactions that regenerate NAD+ by transferring electrons from NADH to pyruvate or its derivatives. The two main types are:
Alcohol fermentation: Pyruvate is converted to ethanol, releasing CO2 and regenerating NAD+.
Lactic acid fermentation: Pyruvate is converted directly to lactate, regenerating NAD+ without producing CO2.
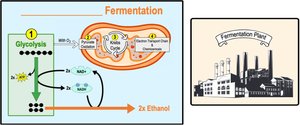
Comparing Fermentation with Anaerobic and Aerobic Respiration
Process | Final Electron Acceptor | ATP Yield (per glucose) |
|---|---|---|
Aerobic Respiration | Oxygen (O2) | ~32 |
Anaerobic Respiration | Alternative molecule (e.g., sulfate) | Variable, less than aerobic |
Fermentation | Organic molecule (pyruvate or acetaldehyde) | 2 |
Evolutionary Significance of Glycolysis
Glycolysis is the most widespread metabolic pathway, suggesting it evolved early in the history of life. Its occurrence in the cytosol indicates it predates mitochondria and was likely used by early prokaryotes.
Glycolysis and the Citric Acid Cycle Connect to Many Other Metabolic Pathways
These pathways serve as major intersections for various catabolic and anabolic processes. Catabolic pathways funnel electrons from diverse organic molecules into cellular respiration, while anabolic pathways use intermediates for biosynthesis.
Versatility of Catabolism
Glycolysis accepts a wide range of carbohydrates.
Proteins are digested to amino acids, which are deaminated before entering glycolysis or the citric acid cycle.
Fats are digested to glycerol (used in glycolysis) and fatty acids (used in the citric acid cycle). Fatty acids undergo beta oxidation to yield acetyl CoA.
Biosynthesis (Anabolic Pathways)
Cells use food not only for energy but also as building blocks for new organic molecules. Intermediates from glycolysis and the citric acid cycle are used in biosynthetic pathways.
Key Equations
General redox reaction:
Cellular respiration:
NAD+ reduction:
Additional info: The notes have been expanded to provide academic context, definitions, and examples for clarity and completeness.
