 Back
BackCellular Respiration: Mechanisms, Pathways, and Energy Yield
Study Guide - Smart Notes

Cellular Respiration and Energy Flow in Ecosystems
Overview of Energy and Chemical Flow
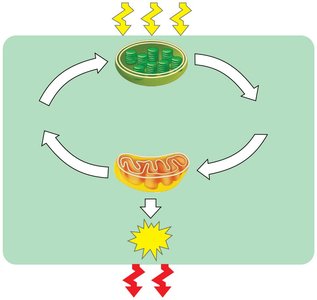
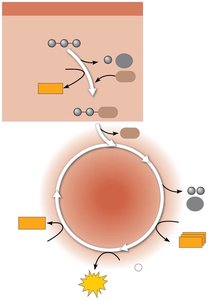
Cellular respiration is a fundamental metabolic process that allows cells to extract energy from organic molecules. This process is tightly linked to photosynthesis in the ecosystem, creating a cycle of energy and matter.
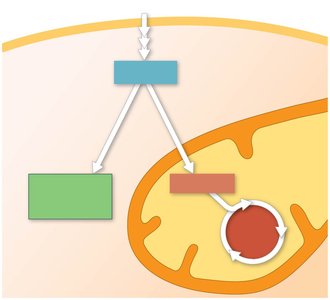
Photosynthesis in chloroplasts converts light energy into chemical energy stored in organic molecules.
Cellular respiration in mitochondria breaks down these molecules, releasing energy as ATP and heat.
ATP powers most cellular work.
Energy flows through ecosystems, while chemicals are recycled.
Redox Reactions in Cellular Respiration
Oxidation-Reduction (Redox) Reactions
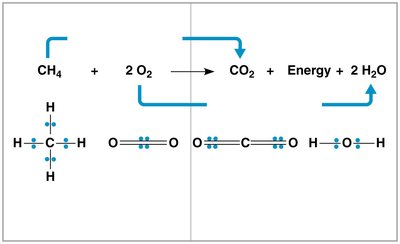
Redox reactions are central to cellular respiration, involving the transfer of electrons between molecules. These reactions change the electron sharing in covalent bonds, releasing energy.
Oxidation: Loss of electrons from a molecule.
Reduction: Gain of electrons by a molecule.
Example: Methane reacts with oxygen to form carbon dioxide and water, with electrons transferred from methane to oxygen.
General Redox Equation
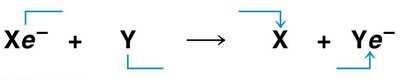
Electron transfer can be represented generically:
Xe- + Y → X + Ye-
Xe- is oxidized to X; Y is reduced to Ye-.
Cellular Respiration Redox Equation
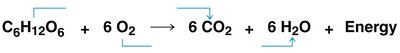
The overall equation for cellular respiration is:
Glucose is oxidized; oxygen is reduced.
Mechanisms of ATP Generation
ATP Synthesis Pathways
ATP is generated through two main mechanisms during cellular respiration:
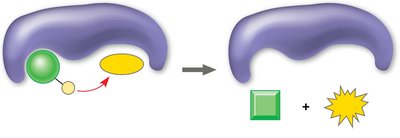
Substrate-level phosphorylation: Direct transfer of a phosphate group to ADP by an enzyme.
Oxidative phosphorylation: Indirect synthesis of ATP via the electron transport chain and chemiosmosis.
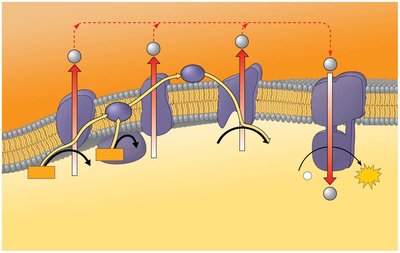
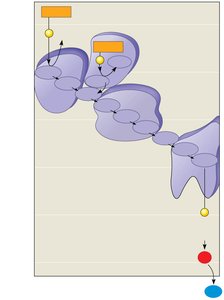
Electron Transport Chain and Chemiosmosis
The electron transport chain (ETC) is a series of protein complexes embedded in the inner mitochondrial membrane. Electrons from NADH and FADH2 are transferred through the chain, creating a proton gradient used by ATP synthase to generate ATP.
ETC receives electrons from NADH and FADH2.
Protons are pumped into the intermembrane space, creating a gradient.
ATP synthase uses this gradient to synthesize ATP.
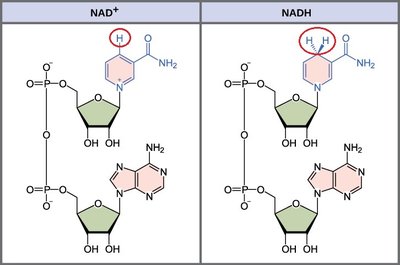
NAD+ and NADH: Electron Carriers
Role of NAD+ in Cellular Respiration
NAD+ (nicotinamide adenine dinucleotide) is a key electron carrier in cellular respiration. It traps electrons from glucose and other organic molecules, becoming reduced to NADH.
NAD+: Oxidized form, accepts electrons.
NADH: Reduced form, donates electrons to the ETC.
Enzyme: Dehydrogenase catalyzes the transfer.
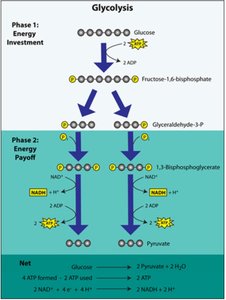
Stages of Cellular Respiration
Glycolysis
Glycolysis is the first stage of cellular respiration, occurring in the cytosol. It breaks down glucose into two pyruvate molecules, producing ATP and NADH.
Two phases: Energy investment and energy payoff.
Net yield: 2 ATP, 2 NADH, 2 pyruvate per glucose.
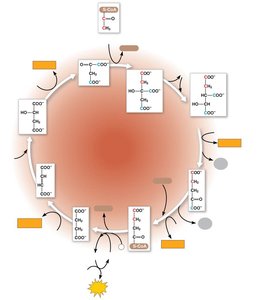
Pyruvate Oxidation and Citric Acid Cycle
Pyruvate is transported into the mitochondrion and converted to acetyl CoA, which enters the citric acid cycle. The cycle completes the oxidation of glucose, generating NADH, FADH2, ATP, and CO2.
Pyruvate oxidation produces acetyl CoA, NADH, and CO2.
Citric acid cycle produces NADH, FADH2, ATP, and CO2.
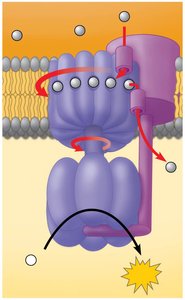
Oxidative Phosphorylation
Most ATP is produced during oxidative phosphorylation, which includes the electron transport chain and chemiosmosis. NADH and FADH2 donate electrons, and oxygen acts as the final electron acceptor, forming water.
Electron transport chain creates a proton gradient.
ATP synthase uses the gradient to produce ATP.
Oxygen is essential for the process; without it, the chain stops.
Fermentation and Anaerobic Respiration
Alternative Pathways Without Oxygen
When oxygen is unavailable, cells can use fermentation or anaerobic respiration to generate ATP. Fermentation allows glycolysis to continue by regenerating NAD+ using organic molecules as electron acceptors.
Aerobic respiration: Uses oxygen as final electron acceptor.
Anaerobic respiration: Uses other molecules (e.g., sulfate) as electron acceptors.
Fermentation: No ETC; organic molecules accept electrons.
Types of Fermentation
There are two main types of fermentation:
Alcohol fermentation: Pyruvate is converted to ethanol and CO2.
Lactic acid fermentation: Pyruvate is converted to lactate.
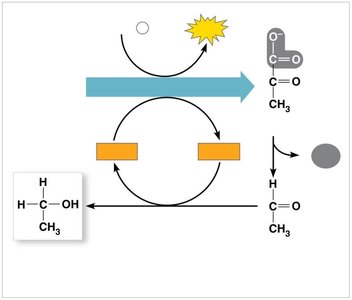
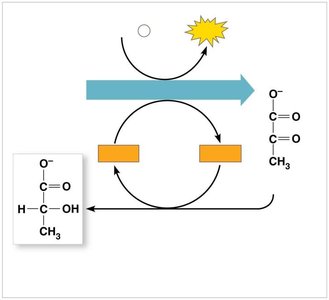
Comparing Fermentation and Cellular Respiration
Energy Yield and Electron Acceptors
Fermentation and cellular respiration differ in their mechanisms for oxidizing NADH and their energy yield.
Fermentation: Organic molecule is final electron acceptor; yields 2 ATP per glucose.
Cellular respiration: Electrons transferred to ETC; yields 32 ATP per glucose.
Evolutionary Basis of Glycolysis
Origins of Glycolysis
Glycolysis is believed to be an ancient metabolic pathway, predating the presence of oxygen in Earth's atmosphere. It is found in nearly all organisms, suggesting its evolutionary importance.
Glycolysis likely arose before the citric acid cycle and oxidative phosphorylation.
It provides a mechanism for energy production in anaerobic conditions.
Additional info: Glycolysis is considered a universal pathway, supporting the theory that early life forms relied on anaerobic metabolism before the evolution of oxygenic photosynthesis and aerobic respiration.
