 Back
BackChapter 2: The Chemical Context of Life – Study Notes
Study Guide - Smart Notes

The Chemical Context of Life
Introduction
Understanding the chemical basis of life is essential for studying biology. Living organisms are composed of matter, which consists of chemical elements in pure form and in combinations called compounds. The properties and interactions of these elements and compounds underlie all biological processes.
Elements and Compounds
Elements: The Building Blocks of Matter
Elements are substances that cannot be broken down into other substances by chemical reactions. There are 92 naturally occurring elements, but only a small subset is essential for life.
Essential Elements: Oxygen (O), carbon (C), hydrogen (H), and nitrogen (N) make up about 96% of living matter.
Trace Elements: Required in minute quantities, such as iron (Fe) and iodine (I).
Compounds: Substances consisting of two or more different elements combined in a fixed ratio, e.g., water (H2O) and sodium chloride (NaCl).
Emergent Properties: Compounds have characteristics different from their constituent elements.
Example: Sodium (Na) is a reactive metal, and chlorine (Cl) is a poisonous gas, but together they form edible table salt (NaCl).



The Elements of Life
Distribution and Importance
Major Elements: O, C, H, N
Minor Elements: Ca, P, K, S, Na, Cl, Mg
Trace Elements: B, Cr, Co, Cu, F, I, Fe, Mn, Mo, Se, Si, Sn, V, Zn
Toxic Elements: Some elements, such as arsenic, are toxic and can cause disease if present in the environment.
Adaptation to Toxic Elements
Evolutionary Adaptations
Some species have evolved tolerance to environments containing toxic elements, such as serpentine soils rich in chromium, nickel, and cobalt. Natural selection favors variants that can survive in these conditions.



Atomic Structure and Properties
Atoms and Subatomic Particles
An atom is the smallest unit of matter that retains the properties of an element. Atoms are composed of:
Protons: Positively charged particles in the nucleus; determine the element's identity.
Neutrons: Neutral particles in the nucleus; determine isotope.
Electrons: Negatively charged particles orbiting the nucleus; determine chemical behavior.
Atomic Number: Number of protons in the nucleus.
Mass Number: Total number of protons and neutrons.
Atomic Mass: Measured in daltons (atomic mass units).
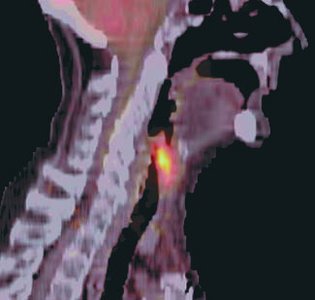
Isotopes and Radioactivity
Isotopes: Atoms of the same element with different numbers of neutrons.
Radioactive Isotopes: Unstable isotopes that decay, emitting particles and energy.
Applications: Used as tracers in medicine and for radiometric dating of fossils.

Electron Configuration and Chemical Properties
Electron Shells and Energy Levels
Electrons occupy specific energy levels called shells. The distribution of electrons among these shells determines the chemical behavior of an atom.
Valence Electrons: Electrons in the outermost shell; determine reactivity.
Inert Elements: Atoms with full valence shells (e.g., helium, neon, argon) are chemically unreactive.
Electron Orbitals
Orbitals are three-dimensional spaces where electrons are likely to be found. The first shell has one s orbital; the second shell has one s and three p orbitals.
Chemical Bonds
Covalent Bonds
Covalent bonds involve the sharing of valence electrons between atoms. The number of covalent bonds an atom can form is determined by the number of electrons needed to complete its valence shell.
Single Bond: Sharing one pair of electrons (e.g., H2).
Double Bond: Sharing two pairs of electrons (e.g., O2).
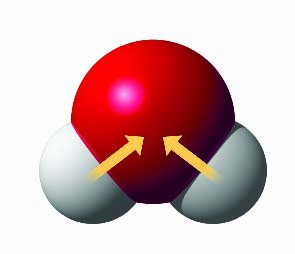
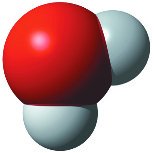
Polar Covalent Bond: Electrons are shared unequally due to differences in electronegativity (e.g., H2O).
Nonpolar Covalent Bond: Electrons are shared equally (e.g., H2, O2).
Ionic Bonds
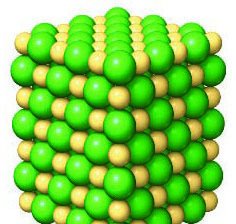
Ionic bonds form when electrons are transferred from one atom to another, resulting in oppositely charged ions that attract each other.
Cation: Positively charged ion (e.g., Na+).
Anion: Negatively charged ion (e.g., Cl-).
Ionic Compounds: Aggregates of cations and anions (e.g., NaCl).

Weak Chemical Interactions
Hydrogen Bonds: Attraction between a hydrogen atom carrying a partial positive charge and an electronegative atom carrying a partial negative charge (e.g., between water molecules).
Van der Waals Interactions: Weak attractions due to transient regions of positive and negative charge.


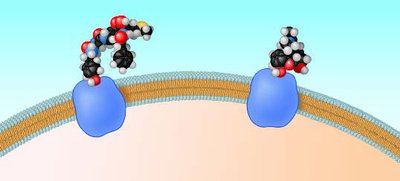
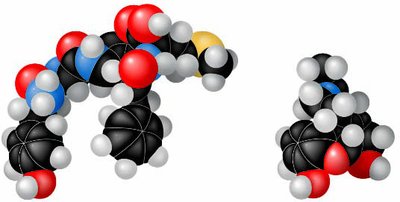
Molecular Shape and Function
Shape Determines Function
The shape of a molecule is determined by the arrangement of its atoms' orbitals. Molecular shape is crucial for biological recognition and function, such as the binding of signaling molecules to receptors.

Chemical Reactions
Making and Breaking Bonds
Chemical reactions involve the making and breaking of chemical bonds, resulting in changes in the composition of matter. Reactants are converted to products, and matter is conserved.
Photosynthesis: Conversion of CO2 and H2O into glucose and O2 using sunlight.
Reversibility: All chemical reactions are theoretically reversible; equilibrium is reached when forward and reverse reaction rates are equal.
Summary Table: Elements in the Human Body
Element | Symbol | Percentage of Body Mass |
|---|---|---|
Oxygen | O | 65.0% |
Carbon | C | 18.5% |
Hydrogen | H | 9.5% |
Nitrogen | N | 3.3% |
Calcium | Ca | 1.5% |
Phosphorus | P | 1.0% |
Potassium | K | 0.4% |
Sulfur | S | 0.3% |
Sodium | Na | 0.2% |
Chlorine | Cl | 0.2% |
Magnesium | Mg | 0.1% |
Trace elements | - | <0.01% |
Key Equations
Mass Number:
Number of Neutrons:
Chemical Reaction Example:
Photosynthesis:
Additional info:
Radioactive isotopes are used for dating fossils and medical imaging.
Molecular mimicry explains how drugs like morphine can bind to natural receptors due to similar shapes.
Weak chemical interactions are essential for the structure and function of biological macromolecules.
