 Back
BackChapter 2: The Chemical Context of Life – Study Notes
Study Guide - Smart Notes

The Chemical Context of Life
Introduction
The study of biology requires a foundational understanding of chemistry, as all living organisms are composed of matter and undergo chemical processes. This chapter explores the structure of atoms, the nature of chemical bonds, and the behavior of elements and molecules essential to life.
Structure of Atoms and Subatomic Particles
Atomic Structure
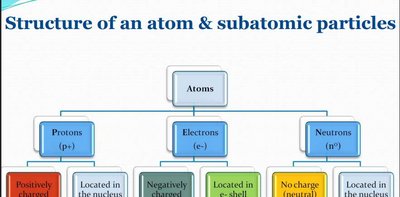
Atoms are the fundamental units of matter, composed of three main subatomic particles: protons, neutrons, and electrons. The arrangement and properties of these particles determine the chemical behavior of elements.
Protons (p+): Positively charged particles located in the nucleus. The number of protons defines the atomic number and the identity of the element.
Neutrons (n0): Neutral particles (no charge) also located in the nucleus. Neutrons contribute to the atomic mass and can vary in number, resulting in isotopes.
Electrons (e-): Negatively charged particles found in electron shells (energy levels) surrounding the nucleus. Electrons are involved in chemical bonding and reactions.
Electron Energy Levels
Energy Shells and Electron Distribution
Electrons occupy specific energy levels or shells around the nucleus. The distribution of electrons among these shells influences an atom's reactivity and bonding behavior.
First shell: Closest to the nucleus, lowest energy level.
Second and third shells: Progressively farther from the nucleus, higher energy levels.
Electrons can absorb energy and move to higher shells (excited state) or lose energy and fall to lower shells (ground state).
Example: When an atom absorbs energy (e.g., from light), an electron may jump to a higher energy shell. When the electron returns to its original shell, energy is released, often as light.
Chemical Bonds
Types of Chemical Bonds
Atoms interact to form molecules through chemical bonds, which can be classified as strong or weak based on their stability and energy requirements.
Strong Bonds:
Covalent Bonds: Atoms share pairs of electrons. Can be polar (unequal sharing) or nonpolar (equal sharing).
Ionic Bonds: Electrons are transferred from one atom to another, resulting in oppositely charged ions that attract each other.
Weak Bonds:
Hydrogen Bonds: Weak attractions between a hydrogen atom covalently bonded to an electronegative atom (like oxygen or nitrogen) and another electronegative atom.
Van der Waals Interactions: Weak, transient attractions between molecules due to temporary charge fluctuations.

Additional info: Covalent bonds are the most common in biological molecules, providing stability to macromolecules such as DNA and proteins.
Ions and Ionic Compounds
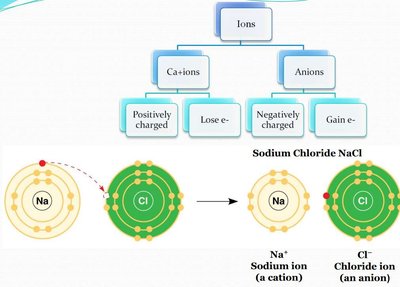
Formation of Ions
Ions are atoms or molecules that have gained or lost electrons, resulting in a net electrical charge. The formation of ions is fundamental to many biological processes, including nerve impulse transmission and muscle contraction.
Cations: Positively charged ions formed by the loss of electrons (e.g., Na+).
Anions: Negatively charged ions formed by the gain of electrons (e.g., Cl-).
Ionic Bond Example: Sodium Chloride (NaCl)
When sodium (Na) transfers an electron to chlorine (Cl), Na becomes a cation (Na+) and Cl becomes an anion (Cl-). The resulting electrostatic attraction forms the ionic compound sodium chloride.
electronegativity: the attraction for electrons in covalent bonds
The Trend for the Periodic table" Across it increases and up it increases
nonpolar covalent= less than 0.5
polarcovalnet = 0.5-1.7
ionic = more than 1.7
isotopes: difference in neutrons in elements
founded through radioactive decay
Summary Table: Subatomic Particles
Particle | Charge | Location |
|---|---|---|
Proton (p+) | +1 | Nucleus |
Neutron (n0) | 0 (neutral) | Nucleus |
Electron (e-) | -1 | Electron shell |
Key Equations
Atomic Number (Z):
Mass Number (A):
Charge of Ion:
