 Back
BackChapter 5: The Structure and Function of Large Biological Molecules
Study Guide - Smart Notes

The Molecules of Life
Overview of Macromolecules
All living organisms are composed of four major classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic acids. These macromolecules are complex and possess unique properties due to the specific arrangement of their atoms. Understanding their structure is essential for explaining their biological functions.
Concept 5.1: Macromolecules are Polymers, Built from Monomers
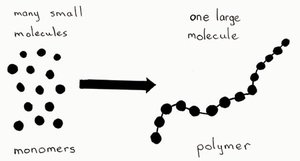
Polymers and Monomers
A polymer is a long molecule consisting of many similar or identical building blocks called monomers, linked by covalent bonds. Three of the four classes of biological macromolecules—carbohydrates, proteins, and nucleic acids—are polymers. Lipids are not true polymers.
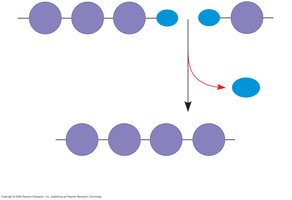
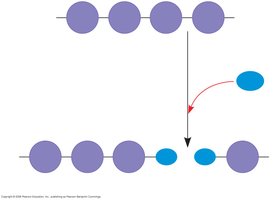
Synthesis and Breakdown of Polymers
Polymer synthesis and breakdown are mediated by enzymes:
Dehydration Reaction (Synthesis): Two monomers bond together through the loss of a water molecule.
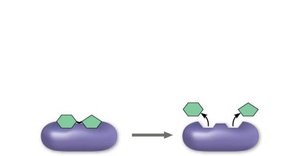
Hydrolysis (Breakdown): Polymers are disassembled to monomers by the addition of a water molecule, reversing the dehydration reaction.
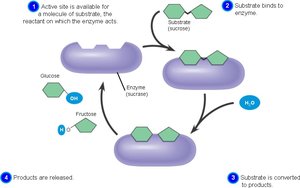
Enzyme-Mediated Reactions
Enzymes accelerate both dehydration and hydrolysis reactions, enabling cells to efficiently build and break down macromolecules.
Diversity of Polymers
The diversity of macromolecules arises from the sequence and length of monomers. For example, proteins are made from 20 different amino acids, while DNA is built from four kinds of nucleotides.
Concept 5.2: Carbohydrates Serve as Fuel and Building Material
Introduction to Carbohydrates
Carbohydrates are macromolecules that play crucial roles in energy storage, cell structure, and cell recognition. They include monosaccharides (simple sugars), oligosaccharides (short polymers), and polysaccharides (long polymers).

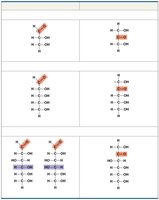
Monosaccharides: Structure and Variation
Monosaccharides vary in:
Location of the carbonyl group (aldose or ketose)
Number of carbon atoms (triose, pentose, hexose)
Spatial arrangement of atoms
Linear and ring forms (ring forms predominate in aqueous solutions)
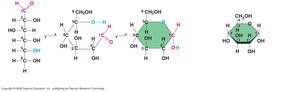
Disaccharides and Glycosidic Linkages
A disaccharide is formed when a dehydration reaction joins two monosaccharides via a glycosidic linkage.
Polysaccharides: Storage and Structural Roles
Polysaccharides serve as storage (starch in plants, glycogen in animals) and structural (cellulose in plants, chitin in arthropods, peptidoglycan in bacteria) molecules.
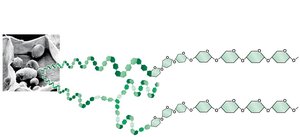

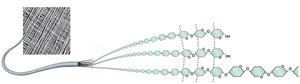
Carbohydrates and Cell Identity
Carbohydrates on cell surfaces (glycoproteins and glycolipids) are key for cell-cell recognition and signaling.
Summary Table: Carbohydrates
Components | Examples | Functions |
|---|---|---|
Monosaccharides | Glucose, Fructose | Fuel; carbon sources for other molecules |
Disaccharides | Lactose, Sucrose | Fuel; can be converted into polymers |
Polysaccharides | Cellulose, Starch, Glycogen, Chitin | Strengthens cell walls; stores glucose; structural support |

Concept 5.3: Lipids are a Diverse Group of Hydrophobic Molecules
Introduction to Lipids
Lipids are hydrophobic molecules, not true polymers, and include fats, phospholipids, and steroids. Their hydrophobic nature arises from nonpolar hydrocarbon chains.

Fats (Triglycerides)
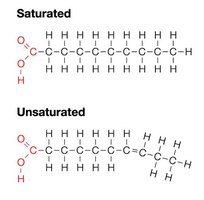
Fats are constructed from glycerol and fatty acids. They serve as energy storage, insulation, and protection. Fatty acids can be saturated (no double bonds, solid at room temperature) or unsaturated (one or more double bonds, liquid at room temperature).
Phospholipids
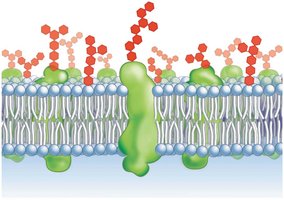
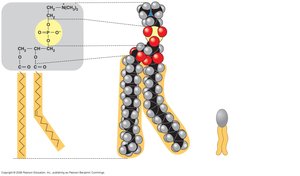
Phospholipids have two fatty acid tails (hydrophobic) and a phosphate group (hydrophilic head). They are amphipathic and form the basis of cell membranes.

Steroids
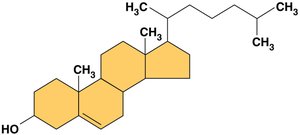
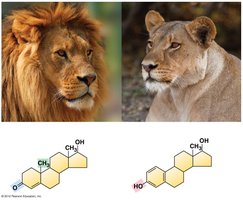
Steroids are characterized by a four-ring carbon skeleton. Examples include cholesterol (a membrane component) and hormones like estrogen and testosterone.
Concept 5.4: Proteins Include a Diversity of Structures, Resulting in a Wide Range of Functions
Functions of Proteins
Proteins are central to cell function, accounting for over 50% of cell dry mass. Functions include catalysis (enzymes), defense, storage, transport, communication, movement, and structural support.
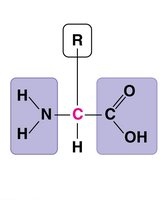
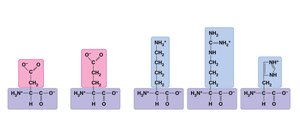
Amino Acids: Building Blocks of Proteins
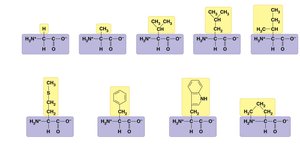
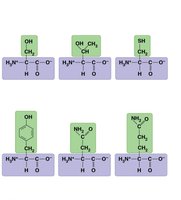
Proteins are polymers of amino acids. Each amino acid has an amino group, carboxyl group, and a unique side chain (R group) that determines its properties.
Polypeptides and Peptide Bonds
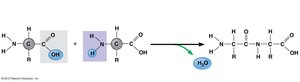
Amino acids are linked by peptide bonds via dehydration reactions, forming polypeptides. Each polypeptide has a unique sequence and directionality (N-terminus to C-terminus).
Protein Structure
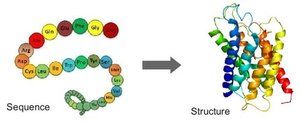
Protein function depends on its three-dimensional structure, which is determined by four levels:
Primary structure: Sequence of amino acids
Secondary structure: Coils and folds (α helix, β sheet) due to hydrogen bonding
Tertiary structure: Interactions among R groups (hydrogen bonds, ionic bonds, hydrophobic interactions, disulfide bridges)
Quaternary structure: Multiple polypeptide chains forming one macromolecule (e.g., hemoglobin, collagen)
Protein Denaturation and Misfolding
Proteins may lose their structure and function due to heat, pH, or chemicals, a process called denaturation. Misfolded proteins are associated with diseases such as Alzheimer's and sickle-cell anemia.
Concept 5.5: Nucleic Acids Store, Transmit, and Help Express Hereditary Information
Roles of Nucleic Acids
Nucleic acids (DNA and RNA) are polymers of nucleotides. DNA stores genetic information and directs its own replication and protein synthesis via RNA. The flow of genetic information is DNA → RNA → protein.
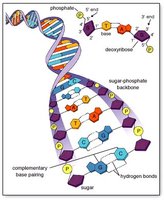
Nucleotide Structure
Each nucleotide consists of a nitrogenous base, a pentose sugar, and a phosphate group. Nitrogenous bases are classified as pyrimidines (cytosine, thymine, uracil) or purines (adenine, guanine).
DNA Structure and Base Pairing
DNA is a double helix with antiparallel strands. Complementary base pairing (A-T, G-C) enables accurate replication and transcription.

RNA Structure
RNA is typically single-stranded, with uracil replacing thymine. It is more variable in structure compared to DNA.
Summary Table: Nucleic Acids
Type | Components | Function |
|---|---|---|
DNA | Deoxyribose, A, T, G, C | Stores hereditary information |
RNA | Ribose, A, U, G, C | Transmits and expresses genetic information |
Summary
Large biological molecules—carbohydrates, lipids, proteins, and nucleic acids—are essential for life. Their structure determines their function, and understanding their properties is fundamental to biology.
