 Back
BackChapter 5: The Working Cell – Membrane Structure, Transport, Energy, and Enzymes
Study Guide - Smart Notes

The Working Cell
Introduction
The plasma membrane and its associated proteins are essential for cell survival and function. This chapter explores how cells utilize membranes, energy, and enzymes to perform vital biological processes.
Membrane Structure and Function
Fluid Mosaic Model
Biologists describe the structure of cell membranes using the fluid mosaic model. This model depicts the membrane as a dynamic arrangement of diverse protein molecules suspended in a fluid phospholipid bilayer. The plasma membrane exhibits selective permeability, allowing certain substances to pass while restricting others. Membrane proteins serve various functions, including transport, signaling, and structural support.

Phospholipid bilayer: Provides the fundamental structure and barrier of the membrane.
Membrane proteins: Facilitate transport, act as receptors, and maintain cell structure.
Selective permeability: Enables the cell to control its internal environment.
Example: Transport proteins allow glucose to enter cells while keeping out unwanted substances.
Spontaneous Formation of Membranes
Phospholipids spontaneously self-assemble into simple membranes, forming enclosed collections of molecules. This process was a critical step in the origin of life, enabling the evolution of the first cells.
Self-assembly: Driven by the amphipathic nature of phospholipids (hydrophilic heads, hydrophobic tails).
Evolutionary significance: Membrane-enclosed structures allowed compartmentalization and complex cellular functions.
Additional info: A functional membrane requires not only lipids but also proteins for transport and signaling.
Transport Across Membranes
Passive Transport: Diffusion
Diffusion is the tendency of particles to spread out evenly in an available space. When diffusion occurs across a cell membrane without energy input, it is called passive transport.
Passive transport: Movement of substances down their concentration gradient, requiring no energy.
Selective permeability: Only certain molecules can diffuse freely (e.g., small, nonpolar molecules).
Example: Oxygen and carbon dioxide diffuse across cell membranes by passive transport.
Osmosis: Diffusion of Water
Osmosis is the diffusion of water across a selectively permeable membrane. Water moves down its concentration gradient until solute concentrations are equal on both sides of the membrane.
Hypertonic solution: Higher solute concentration outside the cell; cell loses water and shrinks.
Hypotonic solution: Lower solute concentration outside the cell; cell gains water and swells.
Isotonic solution: Equal solute concentration; animal cells remain normal, plant cells become flaccid.
Example: Red blood cells placed in a hypotonic solution swell and may burst.
Facilitated Diffusion
Hydrophobic substances easily diffuse across membranes, but polar or charged substances require transport proteins for facilitated diffusion. This process does not require energy and relies on the concentration gradient.
Transport proteins: Provide channels or carriers for specific molecules.
Aquaporins: Specialized channels for rapid water transport.
Example: Glucose transporters facilitate the movement of glucose into cells.
Active Transport
In active transport, cells expend energy (usually from ATP) to move solutes against their concentration gradients. This process is essential for maintaining cellular homeostasis.
ATP: Supplies energy for active transport.
Concentration gradients: Active transport maintains differences in ion concentrations across membranes.
Example: Sodium-potassium pump moves Na+ out and K+ into cells.
Bulk Transport: Exocytosis and Endocytosis
Cells use exocytosis to export bulky molecules and endocytosis to import large molecules. Endocytosis includes phagocytosis (engulfment of particles) and receptor-mediated endocytosis (specific uptake of molecules).
Vesicles: Membrane-bound sacs transport materials across the membrane.
Phagocytosis: Cell engulfs particles by wrapping membrane around them.
Receptor-mediated endocytosis: Specific molecules are taken in after binding to receptors.
Example: White blood cells use phagocytosis to engulf bacteria.
Energy and the Cell
Energy Transformations
Energy is the capacity to cause change. Cells transform energy and matter as they perform work. Kinetic energy is energy of motion, while potential energy is stored energy, including chemical energy in bonds. The laws of thermodynamics state that energy can change form but cannot be created or destroyed, and energy transformations increase disorder (entropy).
First law: Energy conservation.
Second law: Increase in entropy; some energy lost as heat.
Example: Cellular respiration transforms chemical energy in glucose into ATP and heat.
Chemical Reactions: Exergonic and Endergonic
Exergonic reactions release energy, while endergonic reactions require energy input and yield products rich in potential energy. Metabolism encompasses all of a cell’s chemical reactions.
Exergonic: Cellular respiration.
Endergonic: Photosynthesis.
Example: ATP hydrolysis is an exergonic reaction that powers cellular work.
ATP and Energy Coupling
ATP (adenosine triphosphate) powers nearly all forms of cellular work by transferring a phosphate group. This process couples exergonic and endergonic reactions.
ATP hydrolysis:
Energy coupling: Exergonic reactions provide energy for endergonic processes.
Example: ATP powers muscle contraction and active transport.
How Enzymes Function
Enzyme Catalysis
Enzymes are biological catalysts that speed up chemical reactions by lowering the activation energy required. They are not consumed in the reaction.
Activation energy: The energy barrier that must be overcome for a reaction to proceed.
Enzyme specificity: Each enzyme catalyzes a specific reaction.
Example: Amylase catalyzes the breakdown of starch into sugars.
The Catalytic Cycle of Enzymes
An enzyme’s substrate fits specifically into the active site of the enzyme. The catalytic cycle involves substrate binding, conversion to products, and release.
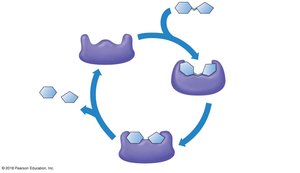
Substrate: The reactant molecule acted upon by the enzyme.
Active site: The region of the enzyme where the substrate binds.
Induced fit: The enzyme changes shape to better fit the substrate.
Products: The molecules released after the reaction.
Example: The enzyme sucrase binds sucrose and breaks it into glucose and fructose.
Enzyme Inhibition
Enzyme activity can be regulated by inhibitors. Competitive inhibitors block the active site, while noncompetitive inhibitors bind elsewhere and change the enzyme’s shape. Feedback inhibition regulates metabolism by shutting down pathways when products accumulate.
Competitive inhibition: Inhibitor competes with substrate for active site.
Noncompetitive inhibition: Inhibitor binds to a different site, altering enzyme function.
Feedback inhibition: End product inhibits an earlier enzyme in the pathway.
Example: The end product of a metabolic pathway inhibits the first enzyme, preventing overproduction.
Enzyme Inhibitors: Drugs, Pesticides, and Poisons
Many drugs, pesticides, and poisons act as enzyme inhibitors. Some are beneficial (e.g., antibiotics), while others are harmful or deadly.
Reversible inhibition: Inhibitor can be removed, restoring enzyme activity.
Irreversible inhibition: Inhibitor permanently disables the enzyme.
Example: Penicillin inhibits bacterial enzymes; nerve gases irreversibly inhibit enzymes in humans.
Summary Table: Types of Membrane Transport
Type | Energy Required? | Direction | Example |
|---|---|---|---|
Passive Transport | No | Down concentration gradient | Oxygen diffusion |
Facilitated Diffusion | No | Down concentration gradient | Glucose transport |
Active Transport | Yes (ATP) | Against concentration gradient | Sodium-potassium pump |
Osmosis | No | Water down concentration gradient | Water movement in cells |
Exocytosis | Yes | Bulk export | Secretion of proteins |
Endocytosis | Yes | Bulk import | Phagocytosis |
Key Concepts for Exam Preparation
Describe the fluid mosaic structure of cell membranes and the functions of membrane proteins.
Explain passive transport, osmosis, and the effects of tonicity on cells.
Distinguish between facilitated diffusion, active transport, exocytosis, and endocytosis.
Define kinetic, potential, chemical energy, and heat; explain the laws of thermodynamics.
Compare exergonic and endergonic reactions; describe ATP’s role in energy coupling.
Explain enzyme function, inhibition, and the effects of drugs and poisons on enzymes.
