 Back
BackChapter 6: An Introduction to Metabolism – Study Notes
Study Guide - Smart Notes

Chapter 6: An Introduction to Metabolism
Overview of Metabolism
Metabolism encompasses all chemical reactions occurring within an organism, enabling the transformation of matter and energy. These reactions are organized into metabolic pathways, each catalyzed by specific enzymes, and are fundamental to life’s processes.
Metabolism: The sum of all chemical reactions in an organism.
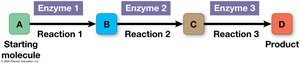
Metabolic pathway: Begins with a specific molecule and ends with a product, with each step catalyzed by a specific enzyme.
Catabolic pathways: Break down complex molecules, releasing energy (e.g., cellular respiration).
Anabolic pathways: Build complex molecules from simpler ones, consuming energy (e.g., protein synthesis).
Bioenergetics: The study of how energy flows through living organisms.
Forms of Energy
Energy is the capacity to cause change and exists in various forms, some of which can perform work. Biological systems utilize and transform energy to maintain order and drive cellular processes.
Kinetic energy: Energy of motion (e.g., movement of molecules).
Thermal energy: Kinetic energy associated with random movement of atoms or molecules; heat is thermal energy in transfer.
Potential energy: Energy due to location or structure (e.g., chemical energy in bonds).
Chemical energy: Potential energy available for release in a chemical reaction.
Energy can be converted from one form to another (e.g., chemical to kinetic).

The Laws of Thermodynamics in Biology
The First Law of Thermodynamics
The first law, also known as the principle of conservation of energy, states that energy can be transferred and transformed, but not created or destroyed. In biological systems, energy from food is converted into usable forms for cellular work.
Energy input (e.g., food, sunlight) is transformed but not lost.
Example: Chemical energy in food is converted to kinetic energy and heat in animals.

The Second Law of Thermodynamics
The second law states that every energy transfer or transformation increases the entropy (disorder) of the universe. Some energy is always lost as heat, increasing disorder.
Entropy (S): A measure of disorder or randomness.
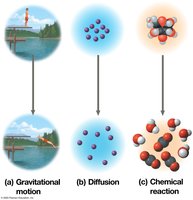
Spontaneous processes increase the entropy of the universe and do not require energy input.
Nonspontaneous processes decrease entropy and require energy input.


Biological Order and Disorder
Living organisms create ordered structures from less organized materials, but overall, the entropy of the universe increases. Organisms are islands of low entropy in a universe tending toward disorder.
Free Energy and Spontaneity
Free Energy Change (ΔG), Stability, and Equilibrium
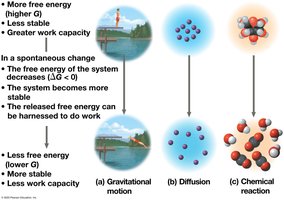
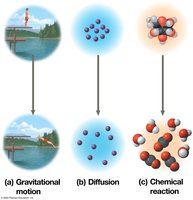
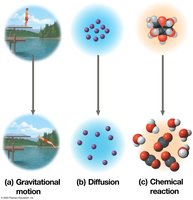
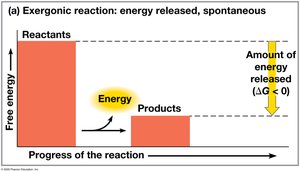
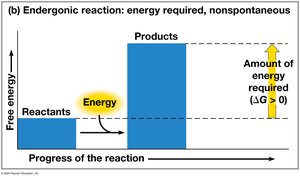
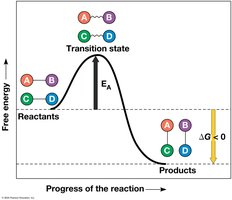
Free energy (G) is the portion of a system’s energy that can do work at constant temperature and pressure. The change in free energy (ΔG) determines whether a process is spontaneous.
ΔG < 0: Spontaneous process; system becomes more stable.
ΔG > 0: Nonspontaneous process; requires energy input.
At equilibrium, ΔG is at its lowest value and no work can be done.
Equation:
Where: = change in free energy = change in enthalpy (total energy) = temperature in Kelvin = change in entropy
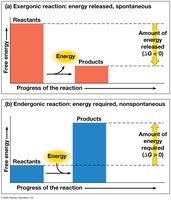
Exergonic and Endergonic Reactions
Metabolic reactions can be classified based on their free energy changes:
Exergonic reactions: Release free energy; spontaneous ().
Endergonic reactions: Absorb free energy; nonspontaneous ().
Cellular respiration is exergonic; photosynthesis is endergonic.

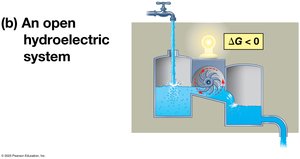
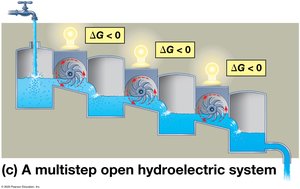
Equilibrium and Metabolism
Cells are open systems and never reach equilibrium, allowing continuous work. Catabolic pathways release free energy in a series of reactions, preventing equilibrium and enabling life.
ATP and Cellular Work
ATP: Structure and Function
ATP (adenosine triphosphate) is the cell’s main energy currency. It consists of ribose, adenine, and three phosphate groups. ATP hydrolysis releases energy used to power cellular work.
ATP hydrolysis:
Energy is released due to the repulsion of negatively charged phosphate groups.
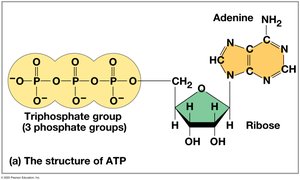
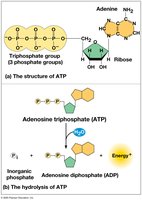
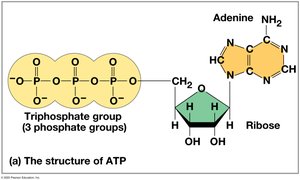
ATP Powers Cellular Work
ATP drives three main types of cellular work by coupling exergonic and endergonic reactions:
Chemical work: Pushing endergonic reactions.
Transport work: Pumping substances across membranes.
Mechanical work: Movement, such as muscle contraction.
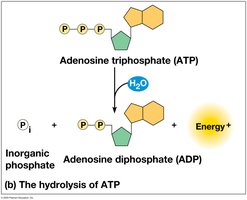
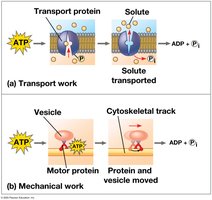
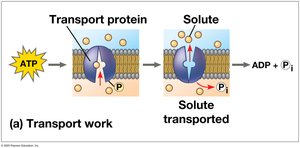
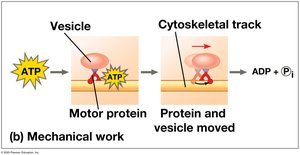
The ATP Cycle
ATP is regenerated by the addition of a phosphate group to ADP, using energy from catabolic reactions. This cycle is essential for maintaining cellular energy balance.
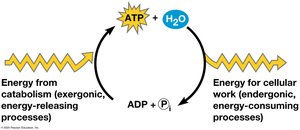
Enzymes and Metabolic Reactions
Enzymes as Catalysts
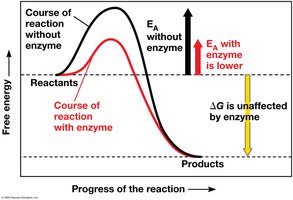
Enzymes are biological catalysts, usually proteins, that speed up metabolic reactions by lowering activation energy barriers without being consumed.
Catalyst: Substance that increases reaction rate without being consumed.
Enzyme: Macromolecule that acts as a catalyst (e.g., sucrase).
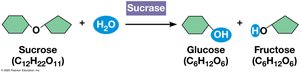
Activation Energy
Activation energy (EA) is the energy required to start a reaction. Enzymes lower EA, allowing reactions to proceed at cellular temperatures.
How Enzymes Work
Enzymes lower activation energy by:
Orienting substrates correctly
Straining substrate bonds
Providing a favorable microenvironment
Covalently bonding to the substrate
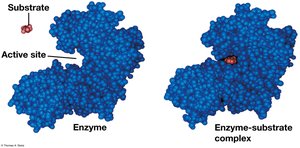
Substrate Specificity and the Active Site
Enzymes are specific for their substrates, binding at the active site to form an enzyme-substrate complex. The induced fit model describes how enzymes change shape to fit substrates.
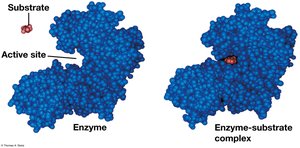
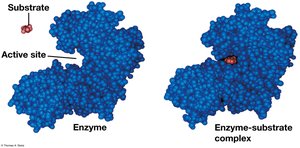
The Catalytic Cycle of an Enzyme
The enzyme’s active site binds substrates, catalyzes the reaction, releases products, and is ready for another cycle.
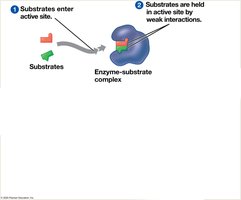
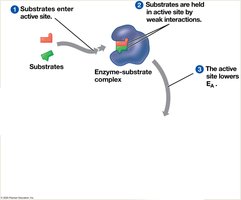
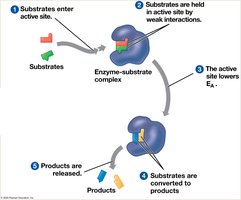
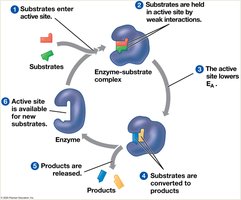
Factors Affecting Enzyme Activity
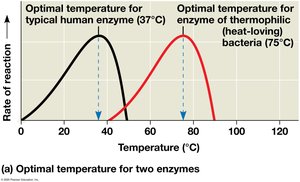
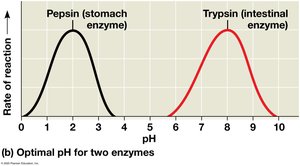
Enzyme activity is influenced by temperature, pH, and chemicals. Each enzyme has optimal conditions for activity.
High temperatures can denature enzymes.
Optimal pH varies (e.g., pepsin in the stomach, trypsin in the intestine).
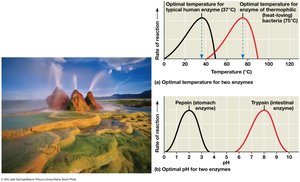
Cofactors and Coenzymes
Cofactors are nonprotein helpers required for enzyme function. They can be inorganic (e.g., metal ions) or organic (coenzymes, often derived from vitamins).
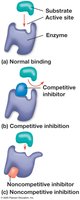
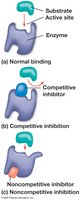
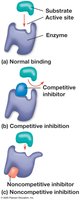
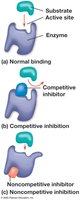
Enzyme Inhibitors
Enzyme inhibitors regulate enzyme activity:
Competitive inhibitors: Bind to the active site, blocking substrate binding.
Noncompetitive inhibitors: Bind elsewhere, altering enzyme shape and function.
Inhibitors can be reversible or irreversible (e.g., toxins, antibiotics).
Regulation of Enzyme Activity
Cells regulate metabolism by controlling enzyme activity through:
Switching genes on/off
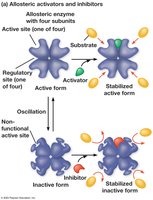
Allosteric regulation: Regulatory molecules bind to sites other than the active site, stabilizing active or inactive forms.
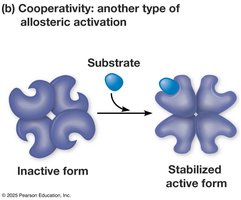
Cooperativity: Substrate binding increases enzyme activity.
Feedback inhibition: End product of a pathway inhibits an early enzyme, preventing overproduction.

Organization of Enzymes Within the Cell
Cellular compartmentalization brings order to metabolic pathways. Some enzymes are structural components of membranes or are localized within specific organelles (e.g., mitochondria for cellular respiration).
Additional info: These notes provide a comprehensive overview of metabolism, energy transformations, enzyme function, and regulation, as covered in a typical introductory biology course.
