 Back
BackChapter 6 Study Guide
Study Guide - Smart Notes

Metabolism
Definition and Overview
Metabolism is the sum of all chemical reactions taking place in a living cell. These reactions are organized into metabolic pathways, which are sequences of steps catalyzed by enzymes, transforming a starting molecule into a product.
Catabolic pathways: Break down complex molecules into simpler ones, releasing energy. Example: Cellular respiration.
Anabolic pathways: Build complex molecules from simpler ones, requiring energy input. Example: Protein synthesis.
Hydrolysis reactions: Typically associated with catabolic pathways.
Dehydration reactions: Typically associated with anabolic pathways.
Energy Concepts
Forms of Energy
Energy is the capacity to cause change, and it exists in various forms:
Kinetic energy: Energy of motion (e.g., thermal, light).
Potential energy: Stored energy (e.g., chemical bonds).
Examples:
A roller coaster at the top of a hill: Potential energy
A pitcher throws a baseball: Kinetic energy
Chemical bonds in glucose: Potential energy
A book falling: Kinetic energy

Thermodynamics
Systems and Energy Exchange
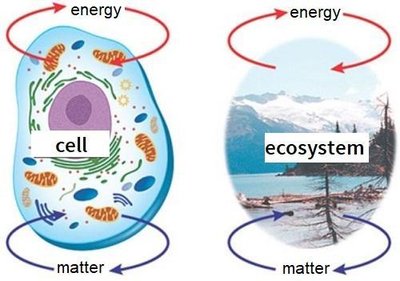
Biological systems can be classified as:
Closed systems: Isolated from surroundings; no exchange of matter or energy.
Open systems: Exchange matter and energy with surroundings; cells and ecosystems are open systems.
Laws of Thermodynamics
First Law: Energy of the universe is constant; it can be transferred or transformed, but not created or destroyed. (Law of Conservation of Energy)
Second Law: Natural processes tend to move toward greater disorder (entropy).

Gibbs Free Energy (ΔG)
Definition and Application
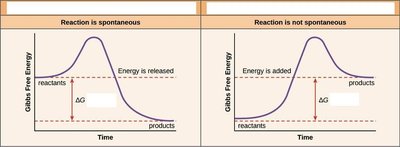
Gibbs free energy (G) is the amount of energy available to do work in a system. The change in free energy () determines whether a reaction is spontaneous.
Exergonic reactions: Release energy, , spontaneous.
Endergonic reactions: Require energy input, , non-spontaneous.
Examples of Free Energy Change
Cellular respiration: Exergonic, catabolic, releases energy.
Photosynthesis: Endergonic, anabolic, requires energy input.
Energy Coupling and ATP
Energy Coupling
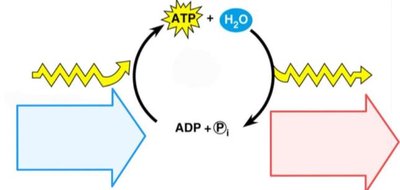
Energy coupling occurs when energy released from an exergonic reaction is used to drive an endergonic reaction. ATP is the primary molecule responsible for mediating energy coupling in cells.
Structure and Function of ATP
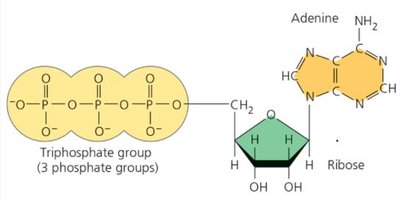
ATP (adenosine triphosphate) is structurally similar to a nucleotide and contains three phosphate groups. The bonds between phosphate groups are unstable due to negative charges, making ATP an efficient energy carrier.
Energy is stored in the chemical bonds between phosphates.
ATP hydrolysis releases energy for cellular work.
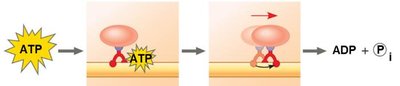
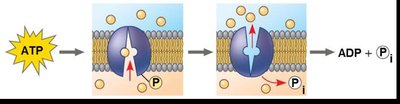
Types of Cellular Work Driven by ATP
Chemical work: Driving endergonic reactions.
Transport work: Moving substances across membranes.
Mechanical work: Moving cellular structures.
Enzymes and Catalysis
Role of Enzymes
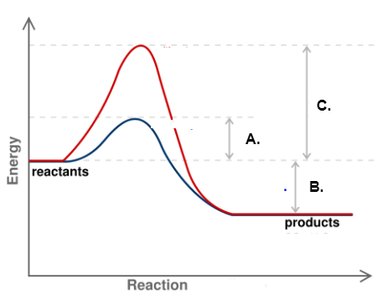
Enzymes are biological catalysts that increase the rate of metabolic reactions by lowering the activation energy (EA) required. They do not affect the overall change in free energy () of the reaction.
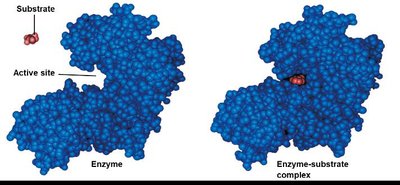
Substrate Binding and Specificity
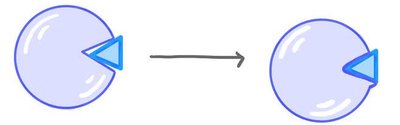
Enzymes bind to specific substrates at their active site. The shape of the active site determines specificity. Substrate binding can induce a change in the active site, leading to a tighter fit (induced fit model).
Lock and key model: Substrate fits exactly into the active site.
Induced fit model: Active site changes shape to fit substrate.
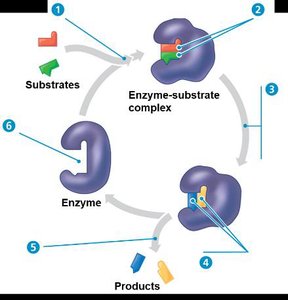
Steps of Enzyme-Substrate Interaction
Substrate enters active site.
Substrate is held by weak interactions (hydrogen bonds, ionic bonds).
Active site lowers activation energy.
Substrate is converted to products.
Products are released.
Active site is available for new substrate.
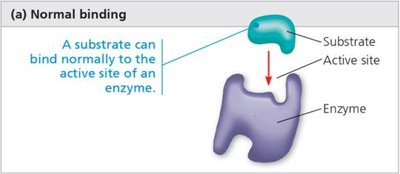
Enzyme Inhibition and Regulation
Types of Enzyme Inhibition
Irreversible inhibition: Inhibitor binds permanently to active site.
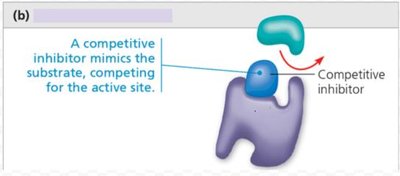
Competitive inhibition: Inhibitor mimics substrate and competes for active site.
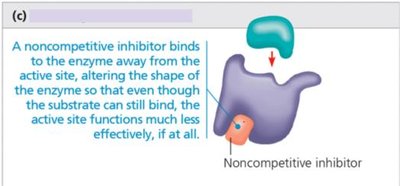
Noncompetitive inhibition: Inhibitor binds away from active site, altering enzyme shape and function.
Factors Affecting Enzyme Activity
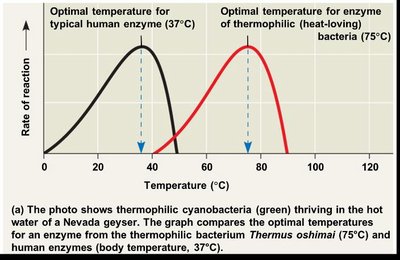
Temperature
pH
Relative concentrations of enzyme and substrate
Presence of inhibitors or activators
Feedback Inhibition and Allosteric Regulation
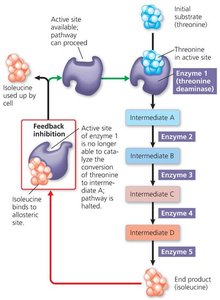
Feedback Inhibition
Feedback inhibition is a common mode of control for metabolic pathways. The end product of a pathway acts as an inhibitor of an enzyme early in the pathway, preventing overproduction and conserving resources.
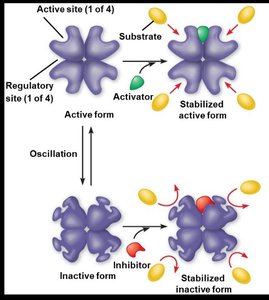
Allosteric Regulation
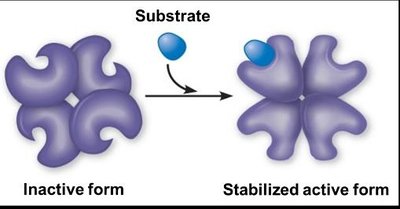
Enzymes with quaternary structure can be regulated allosterically. Allosteric activators stabilize the active form, while allosteric inhibitors stabilize the inactive form. Cooperativity is a special kind of allosteric activation where the substrate itself acts as an activator.
Summary Table: Metabolic Pathways
Pathway | Process | Energy | Example |
|---|---|---|---|
Catabolic | Breakdown | Released | Cellular respiration |
Anabolic | Synthesis | Absorbed | Protein synthesis |
Summary Table: Enzyme Inhibition
Type | Mechanism | Effect |
|---|---|---|
Irreversible | Permanent binding to active site | Enzyme permanently inactivated |
Competitive | Competes for active site | Can be outcompeted by substrate |
Noncompetitive | Binds away from active site | Alters enzyme shape, reduces activity |
