 Back
BackChapter 6: An Introduction to Metabolism – Study Notes
Study Guide - Smart Notes

An Introduction to Metabolism
The Energy of Life
Cells are dynamic chemical factories where thousands of reactions occur, extracting energy from nutrients and using it to perform work. Metabolism encompasses all chemical reactions within an organism, including energy conversion processes such as cellular respiration and bioluminescence.
Concept 6.1: An Organism’s Metabolism Transforms Matter and Energy
Metabolic Pathways
Metabolic pathways are sequences of chemical reactions, each catalyzed by a specific enzyme, that transform a starting molecule into a final product. These pathways are fundamental to cellular function and energy management.
Catabolic pathways: Break down complex molecules, releasing energy (e.g., cellular respiration).
Anabolic pathways: Build complex molecules from simpler ones, consuming energy (e.g., protein synthesis).
Bioenergetics: The study of how energy flows through living organisms.

Forms of Energy
Energy exists in various forms and can be converted from one form to another. Understanding these forms is essential for studying metabolism.
Kinetic energy: Energy of motion.
Thermal energy: Kinetic energy from random movement of atoms/molecules; heat is thermal energy transferred between objects.
Light energy: Used by plants for photosynthesis.
Potential energy: Energy due to position or structure.
Chemical energy: Potential energy stored in chemical bonds, released during reactions.

The Laws of Thermodynamics
The First Law of Thermodynamics
The first law states that energy cannot be created or destroyed, only transferred or transformed. This principle is fundamental to understanding energy flow in biological systems.
Conservation of energy: Total energy in the universe remains constant.

The Second Law of Thermodynamics
Every energy transfer increases the entropy (disorder) of the universe. Some energy is lost as heat, increasing molecular disorder.
Entropy: Measure of disorder; always increases in spontaneous processes.

Concept 6.2: The Free-Energy Change of a Reaction
Free Energy (ΔG), Stability, and Equilibrium
Free energy (G) is the portion of a system’s energy available to do work. The change in free energy (ΔG) determines whether a reaction is spontaneous.
Spontaneous reactions: ΔG < 0; increase stability and can do work.
Nonspontaneous reactions: ΔG > 0; require energy input.
Equation:
Exergonic and Endergonic Reactions
Metabolic reactions are classified by their free energy changes:
Exergonic: Net release of free energy; spontaneous (ΔG negative).
Endergonic: Absorbs free energy; nonspontaneous (ΔG positive).
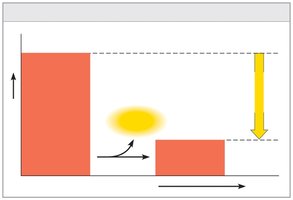
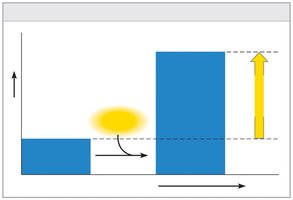
Equilibrium and Metabolism
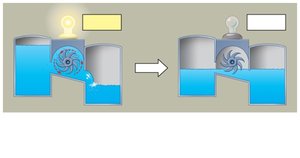
Cells avoid equilibrium by maintaining a constant flow of materials. In biological systems, metabolic pathways prevent equilibrium, allowing continuous work.
Isolated systems: Reach equilibrium, no work possible.
Open systems: Constant flow, never reach equilibrium.

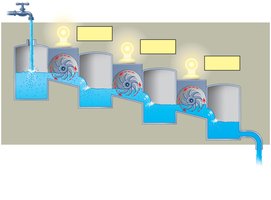
Concept 6.3: ATP Powers Cellular Work by Coupling Exergonic and Endergonic Reactions
The Structure and Hydrolysis of ATP
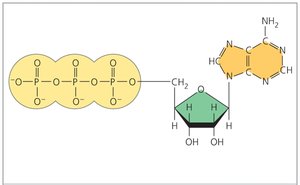
ATP (adenosine triphosphate) is the cell’s energy currency, composed of ribose, adenine, and three phosphate groups. Hydrolysis of ATP releases energy for cellular work.
ATP hydrolysis: ATP + H2O → ADP + Pi + energy
Phosphorylation: Transfer of phosphate group to another molecule.
How ATP Works
ATP hydrolysis powers chemical, transport, and mechanical work in cells by coupling exergonic and endergonic reactions. The energy released is used to phosphorylate target molecules, making them more reactive.
Phosphorylated intermediate: Molecule with added phosphate group, increasing its free energy.
Coupled reactions: Overall ΔG must be negative for spontaneity.
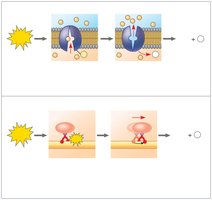
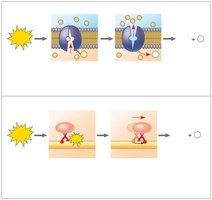
The Regeneration of ATP
ATP is regenerated from ADP and inorganic phosphate using energy from catabolic reactions. This cycle is essential for maintaining cellular energy supply.
Equation:
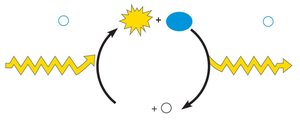
Concept 6.4: Enzymes Speed up Metabolic Reactions by Lowering Energy Barriers
Enzymes and Catalysis
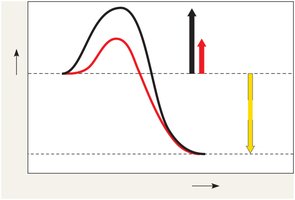
Enzymes are biological catalysts, usually proteins, that speed up reactions by lowering the activation energy (EA) without being consumed. They are highly specific for their substrates.
Activation energy (EA): Energy required to start a reaction.
Enzyme-substrate complex: Temporary association between enzyme and substrate.

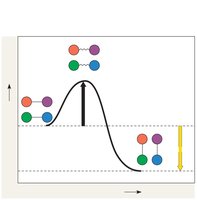
How Enzymes Speed up Reactions
Enzymes lower the activation energy barrier, allowing reactions to proceed faster. They do not alter the overall free energy change (ΔG) of the reaction.
Substrate Specificity of Enzymes
Enzymes are specific due to the fit between their active site and the substrate. The induced fit model describes how enzymes change shape to better accommodate the substrate.

Catalysis in the Enzyme’s Active Site
The active site of an enzyme facilitates catalysis by orienting substrates, straining bonds, providing a favorable environment, or forming temporary covalent bonds. After the reaction, products are released and the enzyme is ready for another cycle.

Effects of Local Conditions on Enzyme Activity
Enzyme activity is influenced by environmental factors such as temperature and pH. Each enzyme has optimal conditions for maximum activity.
Optimal temperature: Human enzymes typically function best at 37°C; thermophilic enzymes at higher temperatures.
Optimal pH: Digestive enzymes have different pH optima (e.g., pepsin in the stomach, trypsin in the intestine).
Example: Thermophilic cyanobacteria thrive in hot environments due to enzymes adapted to high temperatures.
*Additional info: Enzyme denaturation can occur if conditions deviate significantly from the optimum, leading to loss of function.*
