 Back
BackChapter 8: An Introduction to Metabolism – Study Notes
Study Guide - Smart Notes

Chapter 8: An Introduction to Metabolism
Overview of Metabolism
Metabolism encompasses all chemical reactions occurring within a living organism. These reactions are organized into metabolic pathways, which involve the transformation of molecules through a series of enzyme-catalyzed steps. Metabolism is a fundamental property of life, arising from the orderly interactions between molecules.
Key Points:
Metabolism: The sum of all chemical reactions in an organism.
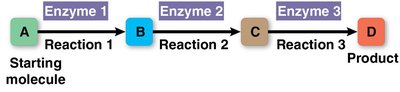
Metabolic Pathway: A sequence of chemical reactions, each catalyzed by a specific enzyme, that transforms a starting molecule into a product.
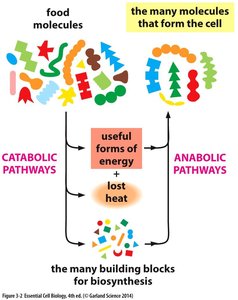
Catabolic and Anabolic Pathways
Metabolic pathways are classified as either catabolic or anabolic, depending on whether they release or consume energy.
Catabolic Pathways: Break down complex molecules into simpler ones, releasing energy (e.g., cellular respiration).
Anabolic Pathways: Build complex molecules from simpler ones, consuming energy (e.g., protein synthesis, glycogen synthesis).
Forms of Energy in Biological Systems
Energy is the capacity to cause change and exists in various forms relevant to biological systems.
Kinetic Energy: Energy of motion (e.g., muscle contraction).
Thermal Energy: Kinetic energy associated with random movement of atoms or molecules; transfer of thermal energy is called heat.
Potential Energy: Stored energy due to position or structure (e.g., water behind a dam, chemical bonds).
Chemical Energy: Potential energy available for release in a chemical reaction (e.g., glucose breakdown).

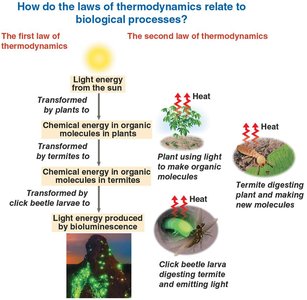
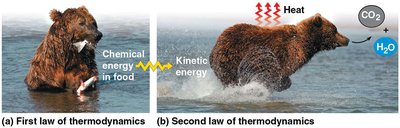
The Laws of Thermodynamics in Biology
Biological processes obey the laws of thermodynamics, which govern energy transformations.
First Law (Conservation of Energy): Energy can be transferred and transformed, but not created or destroyed.
Second Law: Every energy transfer increases the entropy (disorder) of the universe; some energy is lost as heat.
Biological Order and Disorder
Cells create ordered structures from less ordered materials, but overall, the entropy of the universe increases. Organisms maintain order locally while increasing disorder in their surroundings.

Free Energy and Spontaneity of Reactions
Free Energy (G) and Delta G
Free energy is the portion of a system’s energy that can perform work at constant temperature and pressure. The change in free energy (ΔG) during a reaction determines whether the process is spontaneous.
ΔG Equation: where:
= change in free energy
= change in enthalpy (total energy)
= temperature in Kelvin
= change in entropy
Spontaneous Process: Occurs without energy input; ΔG is negative.
Nonspontaneous Process: Requires energy input; ΔG is zero or positive.
Free Energy, Stability, and Equilibrium
Systems with higher free energy are less stable and tend to change to more stable states with lower free energy. The released free energy can be harnessed to do work.
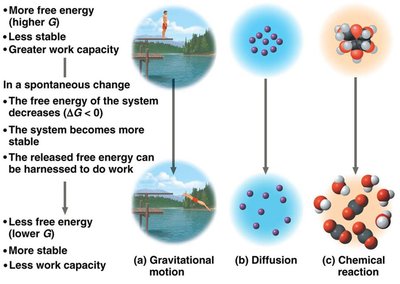
Exergonic and Endergonic Reactions
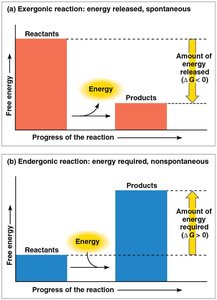
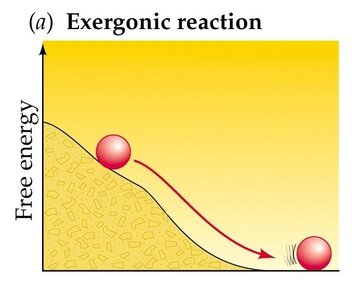
Chemical reactions are classified by their free-energy changes:
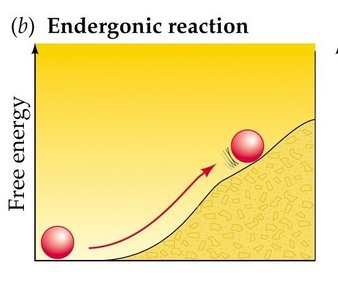
Exergonic Reaction: Releases free energy (ΔG < 0); spontaneous.
Endergonic Reaction: Absorbs free energy (ΔG > 0); nonspontaneous.
ATP and Energy Coupling
ATP: Structure and Function
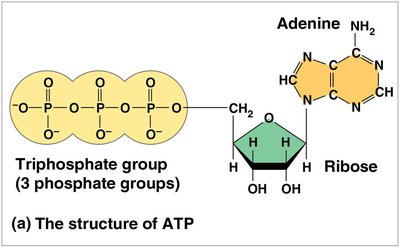
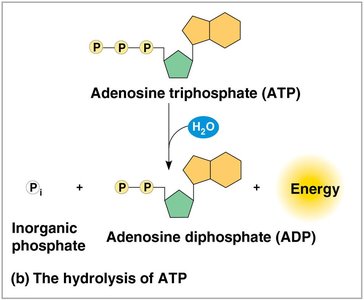
ATP (adenosine triphosphate) is the cell’s main energy currency. It consists of ribose, adenine, and three phosphate groups. ATP hydrolysis releases energy that can be used to drive endergonic reactions.
ATP Powers Cellular Work
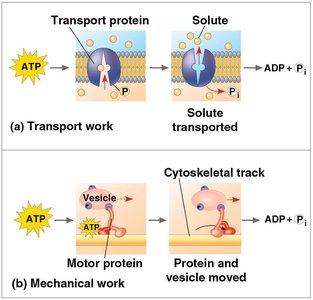
Cells use ATP to perform three main types of work:
Chemical Work: Driving endergonic reactions (e.g., synthesis of macromolecules).
Transport Work: Pumping substances across membranes against gradients.
Mechanical Work: Movement, such as muscle contraction or movement of cilia.
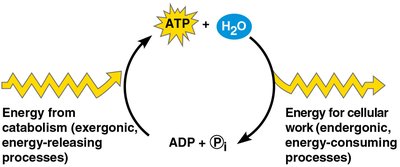
The ATP Cycle
ATP is regenerated from ADP and inorganic phosphate using energy from catabolic (exergonic) reactions. This cycle couples energy-releasing and energy-consuming processes in the cell.
Enzymes and Metabolic Reactions
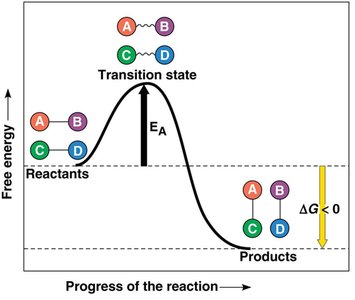
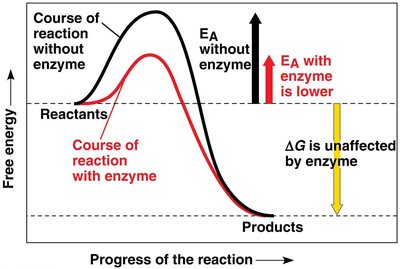
Activation Energy and Catalysis
Every chemical reaction requires an initial input of energy (activation energy, EA) to break bonds. Enzymes act as biological catalysts, lowering the activation energy barrier and speeding up reactions without being consumed.
Enzyme Structure and Function
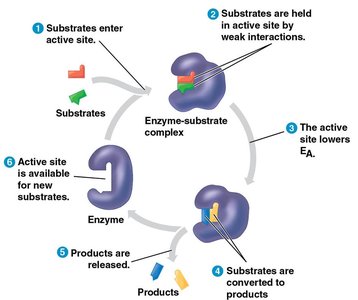
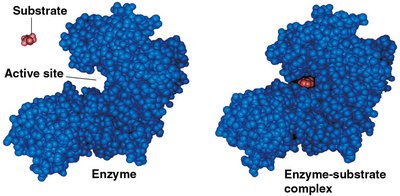
Enzymes are highly specific for their substrates, binding them at the active site to form an enzyme-substrate complex. The induced fit model describes how the enzyme changes shape to better fit the substrate, enhancing catalysis.
Factors Affecting Enzyme Activity
Enzyme activity is influenced by environmental conditions such as temperature and pH, as well as by cofactors and inhibitors.
Optimal Temperature and pH: Each enzyme has specific conditions under which it functions best.
Cofactors: Nonprotein helpers (inorganic ions or organic coenzymes) required for enzyme activity.
Inhibitors: Molecules that decrease enzyme activity. Competitive inhibitors bind the active site, while noncompetitive inhibitors bind elsewhere, altering enzyme shape.
Summary Table: Types of Enzyme Inhibition
Type of Inhibitor | Binding Site | Effect on Enzyme | Can Be Overcome By |
|---|---|---|---|
Competitive | Active site | Blocks substrate binding | Increasing substrate concentration |
Noncompetitive | Allosteric site (not active site) | Changes enzyme shape, reduces activity | Not overcome by substrate increase |
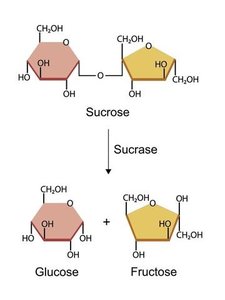
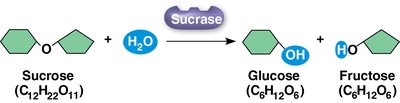
Example: Sucrase catalyzes the hydrolysis of sucrose into glucose and fructose, demonstrating enzyme specificity and the lowering of activation energy.
Additional info: Most vitamins are coenzymes or precursors for coenzymes, highlighting the importance of micronutrients in metabolism.
