 Back
BackEnergy and Enzymes: An Introduction to Metabolism (Chapter 8 Study Notes)
Study Guide - Smart Notes

Energy and Enzymes: An Introduction to Metabolism
8.1 What Happens to Energy in Chemical Reactions?
Chemical reactions in biological systems involve the transformation of energy, which is essential for life processes. Understanding the types of energy and how they change during reactions is fundamental to studying metabolism.
Kinetic Energy: The energy of motion. In biological systems, this is often observed as thermal energy, which is the energy of molecules moving.
Potential Energy: Energy stored in position or configuration. In cells, chemical energy is stored in the bonds of molecules.
Energy can be transformed from one type to another, but it cannot be created or destroyed (First Law of Thermodynamics).
Chemical Reactions Involve Energy Transformations:
First Law of Thermodynamics: Energy is conserved; it can only be transferred or transformed.
Enthalpy (H): The total energy in a molecule, including both potential energy in bonds and the effect of kinetic energy.
Exothermic Reactions: Release heat; products have less potential energy than reactants; is negative.
Endothermic Reactions: Absorb heat; products have more potential energy than reactants; is positive.
Entropy (S): A measure of disorder. When products are less ordered than reactants, entropy increases ( is positive).
Second Law of Thermodynamics: The total entropy of a system always increases in a spontaneous process.
Gibbs Free Energy (G): Determines whether a reaction is spontaneous or requires energy input. The change in free energy () is calculated as:
If , the reaction is spontaneous (exergonic).
If , the reaction is nonspontaneous (endergonic).
Temperature and Concentration Affect Reaction Rates:
For reactions to proceed, bonds must break and form, requiring specific molecular collisions.
Higher concentrations and temperatures increase the rate of reaction by increasing collision frequency and energy.
8.2 Nonspontaneous Reactions May Be Driven Using Chemical Energy
Cells often drive nonspontaneous (endergonic) reactions by coupling them with spontaneous (exergonic) reactions, allowing the overall process to proceed.
Energetic Coupling: The energy released from an exergonic reaction is used to drive an endergonic reaction.
This coupling occurs via:
Transfer of electrons (redox reactions)
Transfer of phosphate groups (e.g., ATP hydrolysis)
Redox Reactions:
Oxidation: Loss of electrons (exergonic)
Reduction: Gain of electrons (endergonic)
These always occur together and represent energetic coupling of two half-reactions.
ATP Transfers Energy via Phosphate Groups:
Adenosine Triphosphate (ATP): The primary energy currency of the cell, with high potential energy due to repulsion between three negatively charged phosphate groups.
Hydrolysis of ATP releases energy ( kcal/mol under standard conditions), which can be used to drive endergonic reactions.
Phosphorylation: The addition of a phosphate group to a molecule, increasing its potential energy and making it more reactive.
8.3 How Enzymes Work
Enzymes are biological catalysts that speed up chemical reactions by lowering the activation energy required for the reaction to proceed.
Activation Energy (Ea): The energy required to strain chemical bonds so they can react.
Transition State: The high-energy intermediate state during a reaction.
Enzymes bring substrates together in precise orientations, facilitating bond breaking and formation.
Substrates bind to the enzyme's active site, often inducing a conformational change called induced fit.
Enzymes lower the activation energy by stabilizing the transition state.
Three Steps of Enzyme Catalysis:
Initiation: Substrates are precisely oriented as they bind to the active site.
Transition State Facilitation: Interactions between substrate and active site lower activation energy.
Termination: Products are released from the enzyme.
What Limits the Rate of Catalysis?
The rate of enzyme-catalyzed reactions depends on substrate concentration and enzyme saturation.
At low substrate concentrations, the reaction rate increases linearly with substrate concentration.
At high substrate concentrations, the reaction rate plateaus as all enzyme active sites are occupied (saturation kinetics).
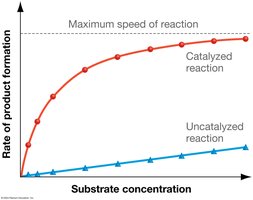
Enzyme activity is limited by the number of available active sites.
Do Enzymes Work Alone?
Many enzymes require non-protein molecules to function properly:
Cofactors: Inorganic ions (e.g., Mg2+, Zn2+) that reversibly interact with enzymes.
Coenzymes: Organic molecules (e.g., NADH, FADH2) that interact with enzymes.
Prosthetic Groups: Non-amino acid molecules permanently attached to proteins.
8.4 What Factors Affect Enzyme Function?
Enzyme structure is critical to function and is sensitive to environmental changes.
Temperature: Affects enzyme folding, movement, and kinetic energy.
pH: Affects enzyme shape, reactivity, and the charge on acidic and basic groups.
Both temperature and pH can alter enzyme activity by changing protein structure.
Most Enzymes Are Regulated
Enzyme activity can be regulated by molecules that alter their structure or substrate binding ability.
Competitive Inhibition: A molecule competes with the substrate for the active site.
Allosteric Regulation: A molecule binds at a site other than the active site, causing a conformational change that can activate or deactivate the enzyme.
Covalent Modifications: Changes to the enzyme's primary structure, such as phosphorylation, can reversibly activate or deactivate the enzyme.
8.5 Enzymes Can Work Together in Metabolic Pathways
Metabolic pathways are series of enzyme-catalyzed reactions that build or break down biological molecules.
Catabolic Pathways: Break down molecules for energy and building blocks.
Anabolic Pathways: Use energy and building blocks to synthesize molecules.
Feedback Inhibition: A regulatory mechanism where the final product of a pathway inhibits an enzyme earlier in the pathway, preventing overproduction and conserving resources.
As product concentration increases, it "feeds back" to inhibit the pathway.
This ensures efficient use of substrates and energy.
