 Back
BackStudy guide 12
Study Guide - Smart Notes

Energy and Enzymes in Biological Systems
Free Energy and Chemical Reactions
Chemical reactions in biological systems are governed by changes in free energy, which determines whether a reaction is spontaneous or requires energy input. The Gibbs free energy equation is central to understanding these processes:
Gibbs Free Energy Equation:
Spontaneous Reactions: Occur when (exergonic; energy is released).
Non-spontaneous Reactions: Occur when (endergonic; energy is required).
Endergonic and exergonic reactions can be coupled so that the energy released from one drives the other.
Coupled Reactions
Cells often couple exergonic and endergonic reactions to ensure necessary processes occur. For example, ATP hydrolysis (exergonic) is used to drive many endergonic cellular reactions.
Exergonic Reaction: Releases energy ().
Endergonic Reaction: Requires energy input ().
Coupling: The energy released from ATP hydrolysis is used to power endergonic reactions, often by transferring a phosphate group to a substrate (phosphorylation).
Enzymes and Catalysis
Role of Enzymes
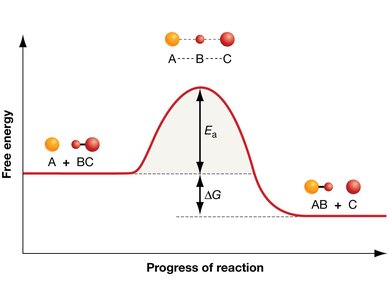
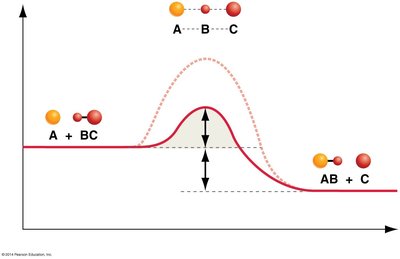
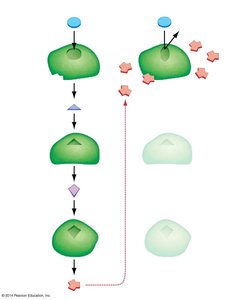
Enzymes are biological catalysts that speed up chemical reactions by lowering the activation energy required for the reaction to proceed. They do not alter the overall free energy change () of the reaction.
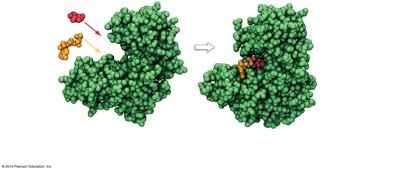
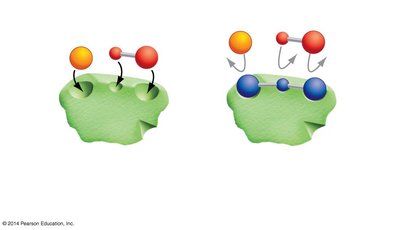
Substrate: The reactant molecule(s) upon which an enzyme acts.
Active Site: The region of the enzyme where substrates bind and undergo a chemical reaction.
Induced Fit: When substrates bind, the enzyme changes shape to better fit the substrate and facilitate the reaction.
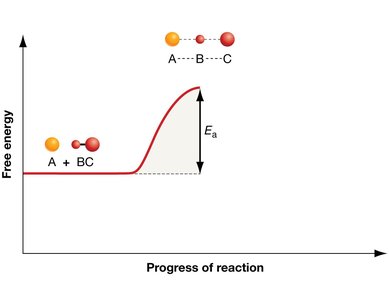
Activation Energy and Reaction Progress
Enzymes lower the activation energy () required to reach the transition state, thus increasing the rate of reaction without being consumed in the process.
Transition State: A high-energy intermediate state during the reaction.
Activation Energy (): The energy required to reach the transition state from the reactants.
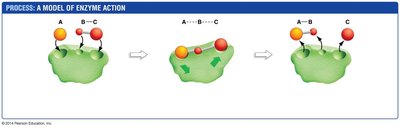
Mechanism of Enzyme Action
Enzyme-catalyzed reactions typically proceed through three main steps:
Initiation: Substrates bind to the enzyme's active site, forming an enzyme-substrate complex.
Transition State Facilitation: The enzyme stabilizes the transition state, lowering the activation energy.
Termination: Products are released, and the enzyme is free to catalyze another reaction.
Enzyme Activity and Regulation
Enzyme activity can be affected by substrate concentration, temperature, pH, and regulatory molecules.
Substrate Concentration: Enzyme activity increases with substrate concentration until a maximum rate (Vmax) is reached.
Temperature and pH: Each enzyme has an optimal temperature and pH for activity; deviations can reduce efficiency or denature the enzyme.
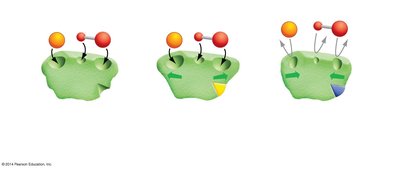
Regulation: Enzymes can be regulated by competitive inhibitors (bind active site) or allosteric regulators (bind elsewhere, changing enzyme shape and activity).
Feedback Inhibition
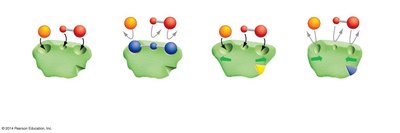
Feedback inhibition is a regulatory mechanism in which the end product of a metabolic pathway inhibits an enzyme involved earlier in the pathway, preventing overproduction of the product.
Allosteric Site: The site on an enzyme where a regulatory molecule can bind, altering enzyme activity.
Example: In a biosynthetic pathway, high concentrations of the final product can inhibit the first enzyme, conserving resources.
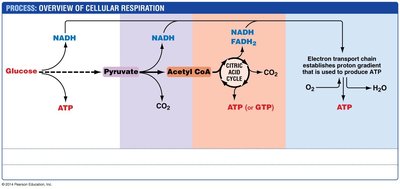
Cellular Respiration: Overview and Glycolysis
Introduction to Cellular Respiration
Cellular respiration is a series of metabolic processes that convert biochemical energy from nutrients into ATP, the energy currency of the cell. It consists of four main stages:
Glycolysis
Pyruvate Processing
Citric Acid Cycle
Electron Transport and Oxidative Phosphorylation
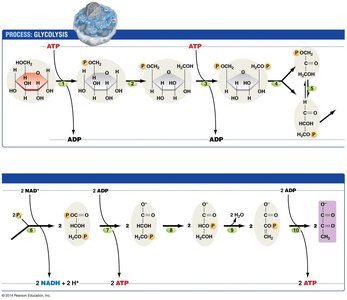
Glycolysis
Glycolysis is the first step of cellular respiration, occurring in the cytosol. It breaks down one molecule of glucose (6 carbons) into two molecules of pyruvate (3 carbons each), producing a net gain of ATP and NADH.
Location: Cytoplasm of both prokaryotes and eukaryotes.
Phases: Energy investment (uses 2 ATP) and energy payoff (produces 4 ATP and 2 NADH).
Net Yield: 2 ATP, 2 NADH, 2 pyruvate per glucose molecule.
Phase | ATP Used | ATP Produced | NADH Produced | End Product |
|---|---|---|---|---|
Energy Investment | 2 | 0 | 0 | Glyceraldehyde-3-phosphate |
Energy Payoff | 0 | 4 | 2 | Pyruvate |
Net | 2 | 2 | 2 | 2 Pyruvate |
Pyruvate Processing and Mitochondrial Structure
After glycolysis, pyruvate is transported into the mitochondria (in eukaryotes) where it is converted to acetyl CoA, releasing CO2 and generating NADH. The mitochondrion's structure is specialized for efficient energy production.
Matrix: Site of the citric acid cycle and pyruvate processing.
Cristae: Folds of the inner mitochondrial membrane, increasing surface area for the electron transport chain.
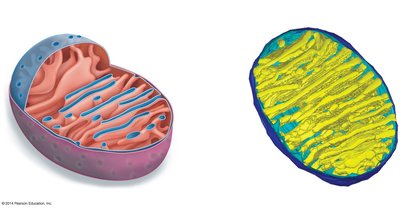
Additional info: The electron transport chain and oxidative phosphorylation occur on the inner mitochondrial membrane, using the proton gradient established by electron carriers to drive ATP synthesis via ATP synthase.
