 Back
BackEnzymes: Structure, Function, and Laboratory Analysis
Study Guide - Smart Notes

Enzymes: Structure and Function
Introduction to Enzymes
Enzymes are biological catalysts, primarily proteins, that accelerate specific metabolic reactions without being consumed in the process. They are essential for sustaining life by enabling biochemical reactions to occur rapidly and efficiently under physiological conditions.
Definition: Enzymes are proteins that catalyze chemical reactions by lowering the activation energy required for the reaction to proceed.
Naming: Most enzyme names end with the suffix “-ase” (e.g., lactase, peroxidase).
Example: Peroxidase catalyzes the breakdown of hydrogen peroxide.
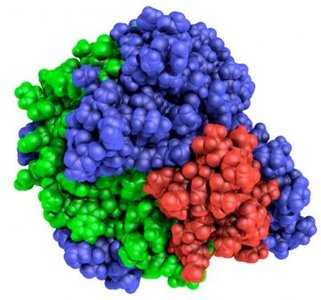
Protein Structure of Enzymes
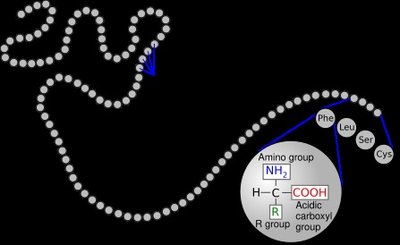
Enzymes are composed of one or more polypeptide chains that fold into specific three-dimensional structures. The structure of an enzyme determines its function and specificity.
Primary Structure: Linear sequence of amino acids.
Secondary Structure: Local folding into alpha-helices and beta-sheets.
Tertiary Structure: Overall three-dimensional shape of a single polypeptide chain.
Quaternary Structure: Association of multiple polypeptide subunits.
Enzyme Function and Mechanism
How Enzymes Work
Enzymes function by binding to specific reactant molecules called substrates at a region known as the active site. This interaction forms an enzyme-substrate complex, facilitating the conversion of substrates into products.
Active Site: The region on the enzyme where the substrate binds and the reaction occurs.
Induced Fit: The active site undergoes a conformational change to better fit the substrate upon binding.
Enzyme-Substrate Complex: Temporary association between enzyme and substrate during catalysis.
Enzyme Specificity
Enzymes are highly specific, typically catalyzing only one type of reaction or acting on a specific substrate due to the precise interaction between the enzyme's active site and the substrate.
Lock-and-Key Model: The substrate fits into the enzyme's active site like a key into a lock.
Induced Fit Model: The enzyme changes shape slightly to accommodate the substrate.
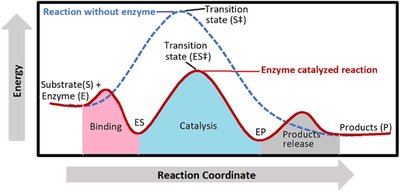
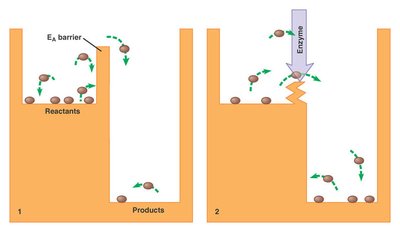
Enzyme Catalysis and Activation Energy
Enzymes accelerate reactions by lowering the activation energy barrier, making it easier for reactants to reach the transition state and form products.
Activation Energy (Ea): The minimum energy required to initiate a chemical reaction.
Catalysis: Enzymes stabilize the transition state, reducing the energy needed for the reaction.
Factors Affecting Enzyme Activity
Optimal Conditions
Each enzyme has optimal conditions under which it functions most efficiently. These include specific temperature, pH, and substrate concentration ranges.
Temperature: Enzyme activity increases with temperature up to an optimum, after which activity declines due to denaturation.
pH: Each enzyme has an optimal pH; deviations can reduce activity or denature the enzyme.
Substrate Concentration: Increasing substrate concentration increases reaction rate up to a saturation point.
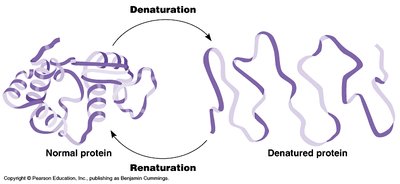
Denaturation
Denaturation is the loss of an enzyme’s native structure, resulting in loss of function. This can be caused by extreme temperatures, pH changes, or chemical agents.
Effect: Denatured enzymes cannot bind substrates or catalyze reactions effectively.
Reversibility: Some denaturation is reversible, but often it is permanent.
Laboratory Analysis of Enzyme Activity
Peroxidase Activity Assay
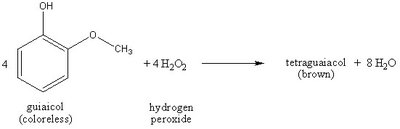
In laboratory experiments, peroxidase activity can be measured by monitoring the conversion of guaiacol (colorless) to tetraguaiacol (brown) in the presence of hydrogen peroxide. The rate of color change reflects enzyme activity.
Reaction: Guaiacol + Hydrogen Peroxide → Tetraguaiacol + Water
Measurement: The increase in brown color is quantified using a spectrophotometer.
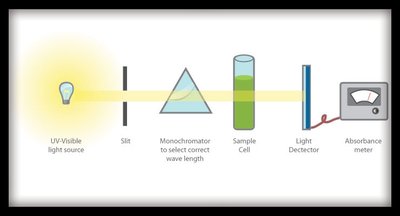
Using a Spectrophotometer
A spectrophotometer measures the absorbance of light by a solution, allowing quantification of product formation in enzyme assays. Proper calibration and wavelength selection are essential for accurate measurements.
Calibration: The instrument must be calibrated before use to ensure accurate readings.
Wavelength Selection: The wavelength is set to match the absorbance maximum of the product (e.g., tetraguaiacol).
Data Collection: Absorbance is measured over time to determine the rate of reaction.
Experimental Design Considerations
When designing enzyme experiments, it is important to control variables such as enzyme and substrate concentrations, temperature, and pH. Data should be recorded systematically for analysis and reporting.
Controls: Include negative and positive controls to validate results.
Replicates: Perform multiple trials to ensure reliability.
Data Recording: Record all observations and measurements promptly and accurately.
Summary Table: Key Properties of Enzymes
Property | Description | Example |
|---|---|---|
Catalyst | Speeds up reactions without being consumed | Peroxidase |
Specificity | Acts on specific substrates | Sucrase acts on sucrose |
Optimal Conditions | Works best at specific temperature and pH | Pepsin (pH 2), Trypsin (pH 8) |
Denaturation | Loss of structure and function under extreme conditions | Boiling an enzyme solution |
Key Terms
Substrate: The reactant molecule upon which an enzyme acts.
Active Site: The region of the enzyme where substrate binding and catalysis occur.
Induced Fit: The dynamic adjustment of the enzyme’s active site to fit the substrate.
Denaturation: The process by which an enzyme loses its structure and function.
Optimal Conditions: The specific environmental conditions where an enzyme is most active.
