 Back
BackEnzymes: Structure, Function, and Regulation
Study Guide - Smart Notes

Enzymes: Structure, Function, and Regulation
Introduction to Enzymes
Enzymes are specialized biological catalysts that accelerate chemical reactions in living organisms. They are essential for sustaining life by enabling metabolic processes to occur efficiently and under mild cellular conditions.
Enzyme: A type of protein that speeds up the rate of a reaction without being consumed in the process. Enzymes are highly specific for their substrates.
Catalyst: A substance that increases the rate of a chemical reaction without undergoing permanent change itself.
Example: Sucrase catalyzes the hydrolysis of sucrose into glucose and fructose.
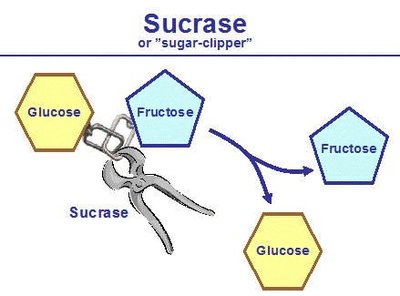

Enzyme Vocabulary and Mechanism
Understanding enzyme function requires familiarity with several key terms and concepts related to their mechanism of action.
Substrate: The specific reactant that an enzyme acts upon. The enzyme-substrate interaction is highly specific.
Active Site: The region on the enzyme where the substrate binds and the reaction occurs.
Enzyme-Substrate Complex: The temporary association formed when an enzyme binds its substrate(s).
Induced Fit: The active site of the enzyme changes shape slightly to fit the substrate more snugly upon binding, enhancing catalysis.


How Enzymes Work
Enzymes lower the activation energy required for reactions, making them proceed faster. They do not alter the overall energy change of the reaction but provide an alternative pathway with a lower energy barrier.
Activation Energy (EA): The minimum energy required for reactants to undergo a chemical reaction.
Mechanisms of Catalysis:
Orienting substrates correctly
Straining substrate bonds
Providing a favorable microenvironment
Direct participation in the reaction


Enzyme Specificity and Environmental Effects
Enzymes are highly specific for their substrates and are sensitive to environmental conditions such as temperature and pH. Each enzyme has optimal conditions under which it functions most efficiently.
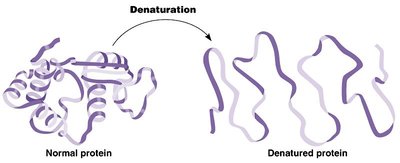
Optimal Temperature: The temperature at which an enzyme's activity is maximal. Too high or too low temperatures can denature the enzyme, reducing its activity.
Optimal pH: The pH at which an enzyme is most active. Deviations from this pH can lead to denaturation or reduced activity.
Cofactors and Coenzymes
Some enzymes require non-protein helpers to function properly. These molecules can be inorganic ions or organic molecules.
Cofactors: Inorganic ions (e.g., Mg2+, Fe2+, Zn2+) that are required for enzyme activity.
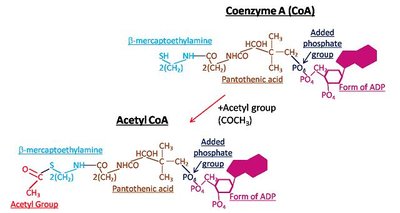
Coenzymes: Organic molecules (often derived from vitamins) that assist enzymes, such as NAD+ (from niacin, vitamin B3), FAD (from riboflavin, vitamin B2), and Coenzyme A.

Enzyme Inhibition and Regulation
Enzyme activity can be regulated by inhibitors and allosteric modulators, which are crucial for controlling metabolic pathways.
Competitive Inhibitors: Molecules that resemble the substrate and compete for binding at the active site, reducing enzyme activity. Example: Penicillin inhibits bacterial cell wall synthesis.
Non-Competitive (Allosteric) Inhibitors: Bind to a site other than the active site (allosteric site), causing a conformational change that reduces enzyme activity. Example: Some anti-cancer drugs inhibit enzymes involved in DNA synthesis.
Irreversible Inhibitors: Bind permanently to the enzyme, often at the active site or allosteric site, rendering the enzyme nonfunctional. Example: Sarin gas inhibits acetylcholinesterase, leading to nerve malfunction.

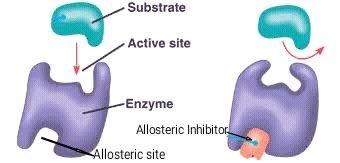

Allosteric Regulation
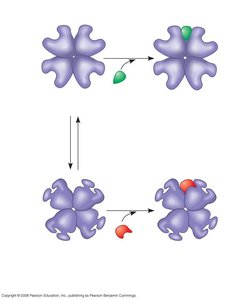
Allosteric regulation involves the binding of regulatory molecules at sites other than the active site, causing conformational changes that affect enzyme activity. Enzymes can be stabilized in either active or inactive forms by allosteric activators or inhibitors.
Allosteric Activators: Stabilize the active form of the enzyme, increasing activity.
Allosteric Inhibitors: Stabilize the inactive form, decreasing activity.
Feedback Inhibition
Feedback inhibition is a regulatory mechanism in which the end product of a metabolic pathway inhibits an enzyme involved earlier in the pathway. This prevents the unnecessary accumulation of products and maintains metabolic balance.
Mechanism: The final product acts as an allosteric inhibitor of an enzyme at the pathway's start.
Importance: Ensures efficient use of resources and prevents chemical chaos in the cell.


Summary Table: Types of Enzyme Inhibition
Type of Inhibition | Binding Site | Effect on Enzyme | Example |
|---|---|---|---|
Competitive | Active site | Blocks substrate binding | Penicillin |
Non-Competitive (Allosteric) | Allosteric site | Changes enzyme shape, reduces activity | Anti-cancer drugs |
Irreversible | Active or allosteric site | Permanently inactivates enzyme | Sarin gas |
Key Equations
General Enzyme Reaction:
Hydrolysis of Sucrose by Sucrase:
Additional info: Enzyme kinetics, including Michaelis-Menten kinetics, are often used to quantitatively describe enzyme activity, but are not detailed here. Enzyme regulation is crucial for homeostasis and adaptation to changing cellular conditions.
