 Back
BackEnzymes: Structure, Function, and Regulation
Study Guide - Smart Notes

Enzymes: Structure, Function, and Regulation
Enzyme Basics
Enzymes are specialized biological catalysts, primarily proteins, that accelerate chemical reactions in living organisms without being consumed in the process. They are essential for sustaining life by enabling metabolic reactions to occur rapidly and efficiently under physiological conditions.
Enzyme: A protein that speeds up the rate of a reaction by lowering the activation energy required. Example: sucrase.
Catalyst: A substance that increases the rate of a chemical reaction without undergoing permanent change.
Substrate: The specific reactant that an enzyme acts upon. When bound to the enzyme, it forms the enzyme-substrate complex. Example: sucrose.
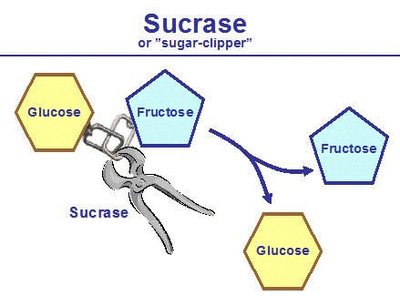

Enzyme Structure and Mechanism
Enzymes possess a unique three-dimensional structure that determines their specificity and function. The region where the substrate binds is called the active site, and the enzyme-substrate interaction is highly specific.
Active Site: The region of the enzyme where the substrate binds and the reaction occurs.
Induced Fit: The active site undergoes a conformational change to fit the substrate more snugly upon binding, enhancing catalytic efficiency.
Enzyme-Substrate Complex: The temporary association between enzyme and substrate during the reaction.


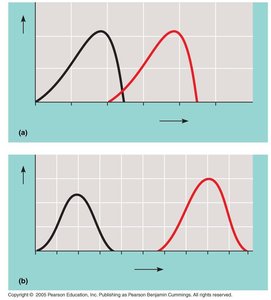
How Enzymes Lower Activation Energy
Enzymes accelerate reactions by lowering the activation energy (EA), which is the minimum energy required for reactants to undergo a chemical transformation. This allows reactions to proceed rapidly at cellular temperatures.
Activation Energy (EA): The energy barrier that must be overcome for a reaction to proceed.
Enzymes lower EA by:
Orienting substrates correctly
Straining substrate bonds
Providing a favorable microenvironment
Participating directly in the reaction

Enzyme Specificity and Environmental Effects
Enzymes are highly specific for their substrates and are sensitive to environmental conditions such as temperature and pH. Each enzyme has an optimal temperature and pH at which it functions most efficiently.
Optimal Temperature: The temperature at which an enzyme's activity is maximal.
Optimal pH: The pH at which an enzyme's activity is maximal.
Extreme conditions can lead to denaturation, where the enzyme loses its functional shape.

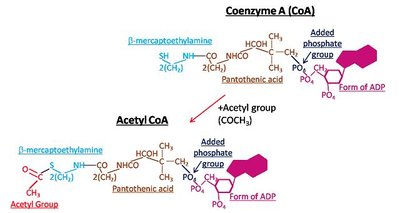
Cofactors and Coenzymes
Some enzymes require non-protein helpers to function properly. These can be inorganic ions (cofactors) or organic molecules (coenzymes).
Cofactors: Inorganic ions such as Mg2+, K+, Ca2+, Zn2+, Fe2+, and Cu2+ that assist enzyme activity.
Coenzymes: Organic molecules, often derived from vitamins, that bind near the active site and are essential for enzyme function. Examples include NAD (from niacin, B3), FAD (from riboflavin, B2), and Coenzyme A.

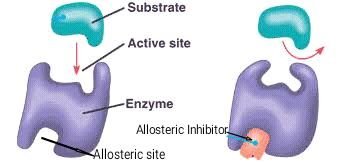
Enzyme Inhibition and Regulation
Enzyme activity can be regulated by inhibitors and allosteric modulators, which are crucial for controlling metabolic pathways.
Competitive Inhibitors: Molecules that resemble the substrate and compete for binding at the active site, reducing enzyme activity. Example: Penicillin.
Non-Competitive (Allosteric) Inhibitors: Molecules that bind to a site other than the active site (allosteric site), causing a conformational change that reduces enzyme activity. Example: Some anti-cancer drugs.
Irreversible Inhibitors: Inhibitors that bind permanently to the enzyme, often leading to permanent loss of activity. Example: Sarin gas.
Allosteric Regulation: Regulatory molecules bind to allosteric sites, stabilizing the enzyme in either an active or inactive form.

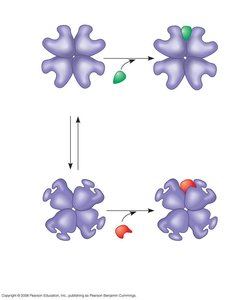

Feedback Inhibition in Metabolic Pathways
Feedback inhibition is a regulatory mechanism in which the end product of a metabolic pathway inhibits an earlier step, preventing the overaccumulation of products and maintaining metabolic balance.
The final product acts as an allosteric inhibitor of an enzyme involved in an early step of the pathway.
This ensures efficient use of resources and prevents chemical chaos within the cell.


Summary Table: Types of Enzyme Inhibition
Type of Inhibition | Binding Site | Effect on Enzyme | Example |
|---|---|---|---|
Competitive | Active site | Blocks substrate binding | Penicillin |
Non-Competitive (Allosteric) | Allosteric site | Changes enzyme shape, reduces activity | Anti-cancer drugs |
Irreversible | Active or allosteric site | Permanently inactivates enzyme | Sarin gas |
Key Equations
General Enzyme Reaction:
Activation Energy Lowering:
Additional info:
Enzyme regulation is critical for homeostasis and adaptation to changing cellular conditions. Many drugs and toxins act by targeting enzymes, highlighting their importance in medicine and toxicology.
