 Back
BackEpigenetics: Mechanisms and Biological Implications
Study Guide - Smart Notes

The Wonderful World of Epigenetics
Introduction to Epigenetics
Epigenetics is the study of heritable changes in gene expression that do not involve changes to the underlying DNA sequence. These changes regulate how genes are turned on or off and are crucial for cellular differentiation and organismal development.
Epigenetic mechanisms include DNA methylation, histone modification, and non-coding RNAs.
Epigenetic regulation allows genetically identical cells to develop into different cell types with specialized functions.
Gene Expression and Cellular Differentiation
Why Are Brain Cells Different from Liver Cells?
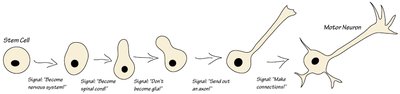
Although all cells in an organism contain the same DNA, they express different sets of genes, leading to diverse cell types such as neurons, muscle cells, and skin cells. This selective gene expression is regulated by epigenetic mechanisms.
Gene expression refers to the process by which information from a gene is used to synthesize functional gene products (proteins or RNAs).
Cell differentiation is driven by the activation or repression of specific genes in response to developmental signals.

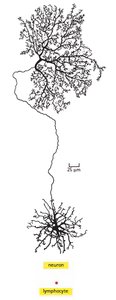
Single Cell Gene Expression
Different cell types express unique sets of genes, which determine their structure and function. For example, muscle cells express muscle-specific genes, while skin cells express skin-specific genes.
Mechanisms of Epigenetic Regulation
DNA Methylation
DNA methylation involves the addition of a methyl group (–CH3) to the cytosine base in DNA, typically at CpG sites. This modification generally represses gene expression by preventing transcription factors from binding to DNA.
High levels of DNA methylation are associated with gene silencing.
DNA methylation patterns are heritable during cell division.
Histone Modification via Acetylation
Histones are proteins around which DNA is wrapped. Acetylation of histone tails by histone acetyltransferases (HATs) reduces the positive charge on histones, loosening DNA and promoting gene expression. Deacetylation by histone deacetylases (HDACs) condenses chromatin and represses gene expression.
Acetylation = gene activation
Deacetylation = gene repression
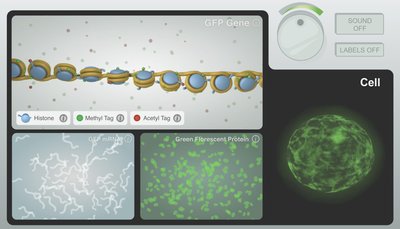
Gene Control via the Epigenome
The epigenome consists of all the chemical modifications to DNA and histone proteins that regulate gene activity. These modifications are dynamic and responsive to environmental cues.
Table: Epigenetic States of Genes
Gene is Active | Gene is Inactive | |
|---|---|---|
DNA around histones | Decondensed | Condensed |
Methyl molecules | Few | Many |
Acetyl molecules | Many | Few |
mRNA transcripts | Many | Few |
Epigenetics and Disease
Turning Genes ‘On’ or ‘Off’ Matters
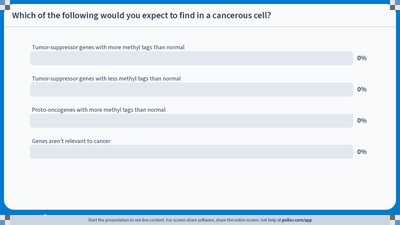
Epigenetic regulation is critical for normal cellular function. Abnormal epigenetic changes can lead to diseases such as cancer.
Tumor-suppressor genes prevent uncontrolled cell division; their silencing by methylation can lead to cancer.
Proto-oncogenes promote cell growth; their abnormal activation can also cause cancer.
Epigenetic Tags and Cellular Memory
Epigenetic Tags Create “Cell Memory”
Epigenetic tags, such as methyl and acetyl groups, serve as cellular memory, ensuring that differentiated cells maintain their identity through cell divisions.
These tags are copied during DNA replication, preserving gene expression patterns.
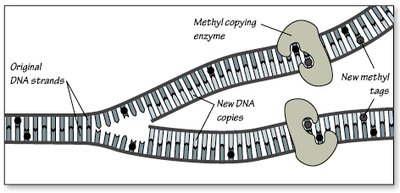
Regulatory Proteins and Enzymes
Gene regulatory proteins recruit enzymes that add or remove epigenetic tags, modulating gene expression in response to internal and external signals.
Case Study: Stress, Behavior, and Epigenetics
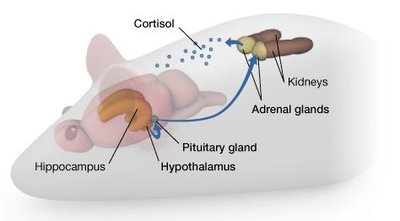
The Stress Circuit – HPA Axis
The hypothalamic-pituitary-adrenal (HPA) axis regulates the stress response by releasing cortisol. Epigenetic regulation of the glucocorticoid receptor (GR) gene in the hippocampus influences how effectively the stress response is shut down.
High methylation of the GR gene reduces receptor expression, leading to prolonged stress responses.
Nature vs. Nurture: Maternal Care and Epigenetics
Experiments in rats show that maternal behavior can alter the epigenetic state of stress-related genes in offspring, affecting their adult behavior.
Pups raised by high-nurturing mothers have less methylation on the GR gene, leading to relaxed behavior.
Pups raised by low-nurturing mothers have more methylation, leading to anxious behavior.
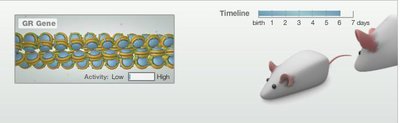
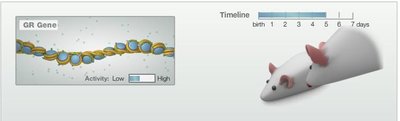
Environmental Influences on the Epigenome
Diet and Epigenetic Tags
Dietary components can influence the addition or removal of epigenetic tags, affecting gene expression and phenotype. Certain nutrients act as methyl donors or cofactors in methylation reactions.
Nutrient | Food Origin | Epigenetic Role |
|---|---|---|
Methionine | Sesame seeds, fish, peppers, spinach | SAM synthesis |
Folic Acid | Leafy vegetables, sunflower seeds, yeast, liver | Methionine synthesis |
Vitamin B12 | Meat, liver, shellfish, milk | Methionine synthesis |
Vitamin B6 | Meats, whole grains, vegetables, nuts | Methionine synthesis |
SAM-e (SAM) | Supplement | Methyl group donor to DNA |
Choline | Egg yolks, liver, soy, meats | Methyl donor to SAM |
Betaine | Wheat, spinach, shellfish, sugar beets | Breaks down toxic byproducts of SAM synthesis |
Resveratrol | Red wine | Removes acetyl groups from histones |
Genistein | Soy products | Increases methylation, cancer prevention |
Sulforaphane | Broccoli | Increases histone acetylation, activates anti-cancer genes |
Butyrate | Produced from dietary fiber fermentation | Increases histone acetylation, activates protective genes |
Diallyl sulphide | Garlic | Increases histone acetylation, activates anti-cancer genes |
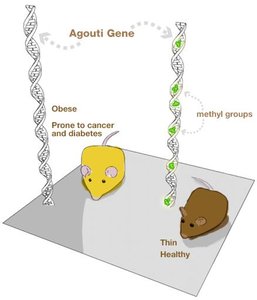
Epigenetic Reprogramming and Imprinting
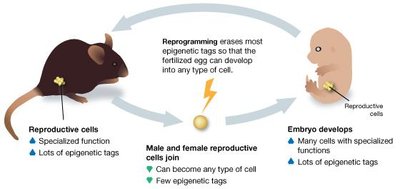
Reprogramming Erases Most Tags
During early development, most epigenetic tags are erased in the zygote, allowing the embryo to develop into any cell type. However, some tags, known as imprints, are retained and passed on to offspring.
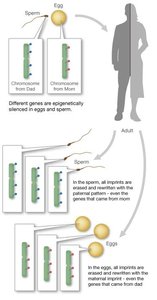
Imprinted Genes
Imprinted genes are expressed in a parent-of-origin-specific manner and escape epigenetic reprogramming. In mammals, about 1% of genes are imprinted, influencing traits such as growth and development.
Examples include the differences in size between ligers and tigons, which result from parent-specific gene expression.
Epigenetic Inheritance
Can Epigenetic Tags Be Passed on for Multiple Generations?
While most epigenetic tags are reset each generation, some evidence suggests that certain epigenetic changes can be inherited across multiple generations, especially if they escape reprogramming. Convincing evidence requires observing changes in the fourth generation.
Environmental exposures (diet, toxins, stress) can potentially affect the epigenome of future generations.
Conclusion
Epigenetics bridges the gap between genotype and phenotype, explaining how environmental factors and life experiences can influence gene expression and organismal traits without altering the DNA sequence. Understanding epigenetics is crucial for fields such as development, disease, and inheritance.
