 Back
BackFoundations of Biology: Structure, Chemistry, and Cell Organization
Study Guide - Smart Notes

Introduction to Biology
Definition and Characteristics of Life
Biology is the scientific study of living organisms and their interactions with each other and their environments. All living things share several fundamental characteristics that distinguish them from non-living matter.
Organization (Order): Living systems are highly structured, with hierarchical levels from molecules to organelles, cells, tissues, organs, and organisms.
Energy Processing: Organisms acquire and use energy to maintain organization and drive metabolic processes. For example, plants capture light energy, while animals obtain chemical energy from food.
Response to Stimuli: Organisms detect and respond to environmental changes, such as light, temperature, and chemicals.
Growth, Development, and Reproduction: Organisms grow and develop according to genetic instructions and reproduce to pass genetic information to offspring.
Evolutionary Adaptation: Populations evolve over generations, with traits that enhance survival and reproduction becoming more common.
Classification and Domains of Life
Purpose and Methods of Classification
Classification (taxonomy) organizes biological diversity, allowing scientists to identify species, determine relationships, and track endangered organisms. Methods include:
Structural Comparisons: Grouping by physical traits, such as homologous (common ancestry) and analogous (similar function, different origin) structures.
Fossil Record: Provides evolutionary history and transitional forms, though often incomplete.
Genetic Comparisons: DNA and protein sequence analysis is the most accurate method for determining evolutionary relationships.
Three Domains of Life
Bacteria: Small, unicellular, prokaryotic, with peptidoglycan cell walls and circular chromosomes.
Archaea: Small, unicellular, prokaryotic, with unique membrane lipids and cell walls lacking peptidoglycan; often extremophiles.
Eukarya: Larger, eukaryotic cells with nuclei, multiple linear chromosomes, and membrane-bound organelles; includes animals, plants, fungi, and protists.
Classification Hierarchy: Domain → Kingdom → Phylum → Class → Order → Family → Genus → Species
Chemical Foundations of Life
Matter, Elements, and Atomic Structure
All organisms are composed of matter, which consists of elements. The major elements in living organisms are carbon (C), hydrogen (H), oxygen (O), nitrogen (N), phosphorus (P), and sulfur (S). Atoms are made of protons, neutrons, and electrons, and can form ions (cations and anions).
Chemical Bonds
Ionic Bonds: Formed by electron transfer between atoms, resulting in attraction between oppositely charged ions (e.g., NaCl).
Covalent Bonds: Formed by sharing electrons; can be nonpolar (equal sharing) or polar (unequal sharing).
Weak Interactions: Hydrogen bonds and van der Waals forces are crucial for biological structure and function.
Properties of Water
Cohesion: Water molecules stick together via hydrogen bonds, creating surface tension.
Temperature Moderation: High heat of vaporization stabilizes temperatures.
Lower Density of Ice: Ice floats, preventing bodies of water from freezing solid.
Excellent Solvent: Water dissolves polar and ionic substances, facilitating cellular reactions.
Carbon and Molecular Diversity
Carbon’s Versatility
Carbon can form four covalent bonds, allowing for diverse molecular structures such as chains, branches, rings, and double bonds. This versatility underlies the diversity of biological macromolecules.
Isomers
Structural Isomers: Same formula, different bonding order.
Stereoisomers: Same bonding order, different spatial arrangement (includes geometric and enantiomers).
Pharmacological Importance: Biological systems are stereospecific; for example, only one enantiomer of ibuprofen is active.
Functional Groups
Functional groups (hydroxyl, carbonyl, carboxyl, amino, sulfhydryl, phosphate, methyl) determine the chemical behavior of molecules, affecting reactivity, polarity, and interactions.
Macromolecules: Structure and Function
Carbohydrates
Monosaccharides: Simple sugars (e.g., glucose, ribose) serve as energy sources and building blocks.
Disaccharides: Two monosaccharides joined by glycosidic linkage (e.g., maltose).
Polysaccharides: Long chains for storage (starch, glycogen) or structure (cellulose, chitin).
Lipids
Fats (Triglycerides): Glycerol + 3 fatty acids; energy storage, insulation.
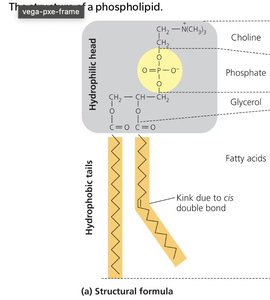
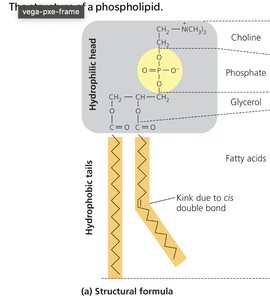
Phospholipids: Glycerol + 2 fatty acids + phosphate; amphipathic, form cell membranes.
Steroids: Four fused rings; e.g., cholesterol, hormones.
Key Point: Lipids are hydrophobic and not true polymers.
Proteins
Amino Acids: 20 types, each with a central carbon, amino group, carboxyl group, hydrogen, and R group.
Peptide Bonds: Link amino acids via dehydration reactions.
Levels of Structure:
Primary: Linear sequence of amino acids.
Secondary: Local folding (α-helix, β-sheet) via hydrogen bonds.
Tertiary: 3D shape from R group interactions.
Quaternary: Multiple polypeptide subunits (e.g., hemoglobin).
Denaturation: Loss of structure and function due to environmental changes.
Nucleic Acids
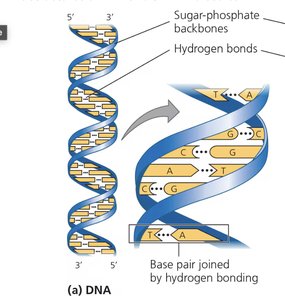
DNA: Double helix of nucleotides (A, T, G, C); stores genetic information.
RNA: Single-stranded; uses uracil instead of thymine; involved in protein synthesis.
Nucleotides: Consist of a phosphate group, 5-carbon sugar, and nitrogenous base.
Cell Membranes and Transport
Phospholipid Bilayer and Fluid Mosaic Model
The plasma membrane is a selectively permeable barrier composed of a phospholipid bilayer with embedded proteins. Phospholipids have hydrophilic heads and hydrophobic tails, causing them to self-assemble into bilayers in aqueous environments.
Integral Proteins: Span the membrane, involved in transport and signaling.
Peripheral Proteins: Loosely attached to the membrane surface.
Other Components: Glycoproteins, glycolipids, and cholesterol contribute to membrane function and fluidity.
Membrane Fluidity
Fluidity is influenced by fatty acid composition (unsaturated tails increase fluidity) and cholesterol (acts as a buffer).
Temperature affects membrane state: high temperature increases fluidity, low temperature can cause solidification.
Transport Mechanisms
Diffusion: Passive movement down a concentration gradient.
Osmosis: Diffusion of water across a selectively permeable membrane.
Facilitated Diffusion: Passive transport aided by channel or carrier proteins.
Active Transport: Movement against a gradient, requiring ATP (e.g., sodium-potassium pump).
Bulk Transport: Endocytosis and exocytosis move large substances via vesicles.
Summary Table: Comparison of Macromolecules
Macromolecule | Monomer | Bond Type | Main Functions |
|---|---|---|---|
Carbohydrates | Monosaccharide | Glycosidic linkage | Energy storage, structure |
Lipids | Fatty acids, glycerol | Ester linkage | Energy storage, membranes, signaling |
Proteins | Amino acid | Peptide bond | Catalysis, structure, transport, signaling |
Nucleic Acids | Nucleotide | Phosphodiester bond | Genetic information, protein synthesis |
Key Equations
Gibbs Free Energy:
ATP Hydrolysis:
Photosynthesis (Overall):
