 Back
BackHydrogen Bonds and the Special Properties of Water
Study Guide - Smart Notes

Hydrogen Bonds and the Special Properties of Water
Introduction
Water is essential for life due to its unique chemical and physical properties, most of which arise from its ability to form hydrogen bonds. These properties are fundamental to biological systems and are critical for processes ranging from cellular structure to temperature regulation.
Hydrogen Bonds
Definition and Nature of Hydrogen Bonds
Hydrogen bond: An electrostatic interaction between a hydrogen atom (with a partial positive charge, δ+) covalently bonded to an electronegative atom (such as O, N, Cl, or F) and another electronegative atom with a lone pair of electrons.
Water is a classic example, where hydrogen bonds form between the hydrogen of one water molecule and the oxygen of another.
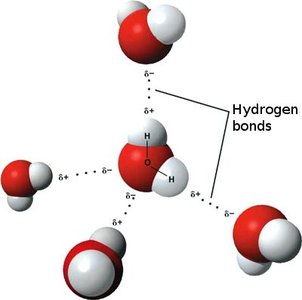
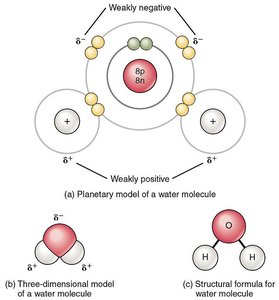
Relative Strength of Chemical Bonds
Covalent bonds are the strongest, followed by ionic bonds, and then hydrogen bonds, which are weaker but significant in large numbers.
Hydrogen bonds are crucial for the structure and function of biological macromolecules.
Biological Importance of Hydrogen Bonds
Proteins: Hydrogen bonds stabilize the three-dimensional structure of proteins, influencing their function.
DNA: The two strands of the DNA double helix are held together by hydrogen bonds between complementary nitrogenous bases (A-T and G-C pairs).
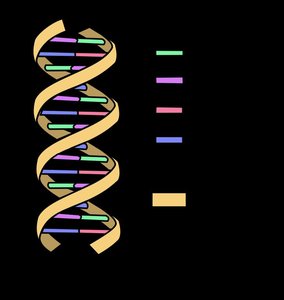
The Special Properties of Water
Water’s ability to form extensive hydrogen bonds gives rise to several unique properties that are essential for life.
1. Temperature Moderation
Water resists changes in temperature, which helps stabilize environments and organisms.
High heat capacity: Water can absorb or release large amounts of heat with only a slight change in its own temperature. The energy required to raise the temperature of 1 gram of water by 1°C is defined as 1 calorie (4.2 Joules).
High heat of vaporization: Water requires a significant amount of energy (540 cal/g) to change from liquid to gas, which moderates Earth's climate and helps organisms cool through evaporation.
2. Cohesion and Adhesion
Cohesion and adhesion are responsible for many of water’s remarkable behaviors, including surface tension and capillary action.
Cohesion: The tendency of water molecules to stick to each other due to hydrogen bonding, resulting in high surface tension (e.g., water droplets forming beads).
Adhesion: The tendency of water molecules to stick to other polar or charged surfaces, such as glass, which contributes to capillary action.
Capillary action: The movement of water within narrow tubes against gravity, essential for water transport in plants.


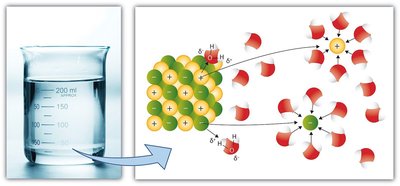
3. Water as the Solvent of Life
Water’s polarity allows it to dissolve a wide variety of substances, making it the universal solvent in biological systems.
Solubility: Water dissolves ionic compounds (like sodium chloride) and many polar molecules, facilitating biochemical reactions and transport of nutrients and waste.
Aqueous solutions: Most cellular processes occur in aqueous solutions, where water surrounds and interacts with solutes.
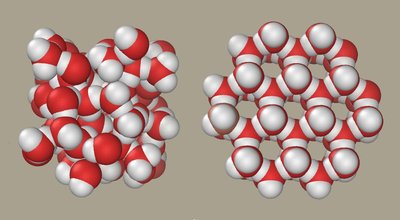
4. Density of Water: Ice Floats
Unlike most substances, water is less dense as a solid than as a liquid, which has profound ecological consequences.
Ice floats: As water freezes, hydrogen bonds stabilize and form a crystalline structure that spaces molecules farther apart, decreasing density.
This property insulates aquatic environments, allowing life to persist under the ice during cold periods.
5. Water and the Search for Extraterrestrial Life
Because water’s properties are so essential for life as we know it, the search for extraterrestrial life often focuses on finding water in other parts of the universe.

Summary Table: Special Properties of Water
Property | Description | Biological Importance |
|---|---|---|
High heat capacity | Absorbs/releases heat with little temperature change | Stabilizes organism and environmental temperatures |
Cohesion & Adhesion | Water molecules stick to each other and to other surfaces | Surface tension, capillary action (water transport in plants) |
Universal solvent | Dissolves many substances due to polarity | Facilitates biochemical reactions and transport |
Lower density as solid | Ice floats on liquid water | Insulates aquatic life in cold climates |
Key Equations
Specific heat of water:
Heat of vaporization:
Where is heat energy, is mass, is specific heat, is temperature change, and is heat of vaporization.
