 Back
BackLipids, Membranes, and the First Cells: Structure, Function, and Transport
Study Guide - Smart Notes

Chapter 6: Lipids, Membranes, and the First Cells
Plasma Membrane: Structure and Function
The plasma membrane (or cell membrane) is a fundamental structure that separates living cells from their external environment. It acts as a selective barrier, allowing essential materials to enter while keeping harmful substances out, and facilitates chemical reactions necessary for life by sequestering appropriate chemicals.
Selective permeability: Controls what enters and exits the cell.
Compartmentalization: Holds molecules together for efficient chemical reactions.
6.1 Lipid Structure and Function
Lipids are carbon-containing compounds that are insoluble in water due to their high proportion of nonpolar C–C and C–H bonds. Hydrocarbons are nonpolar molecules containing only carbon and hydrogen, making them hydrophobic.
Hydrophobicity: Lipids do not mix with water because electrons are shared equally in C–H bonds.
Key types: Steroids, fats, and phospholipids.
Bond Saturation and Hydrocarbon Structure
Fatty acids are simple lipids made of a hydrocarbon chain bonded to a polar carboxyl group. The degree of saturation affects their structure and physical properties:
Saturated fatty acids: Only single bonds between carbons; straight chains; solid at room temperature.
Unsaturated fatty acids: One or more double bonds; kinked chains; liquid at room temperature.
Polyunsaturated: Many double bonds; more kinks.

Physical State and Health Implications
Saturation influences the physical state of lipids:
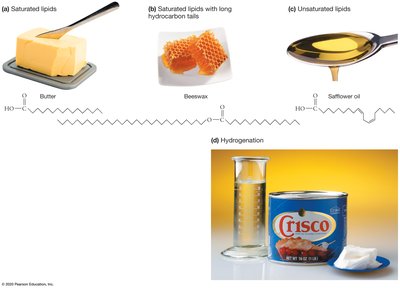
Highly saturated lipids: Solid at room temperature (e.g., butter).
Unsaturated lipids: Liquid at room temperature (e.g., safflower oil).
Hydrogenation: Converts unsaturated fats to saturated, making them solid (e.g., Crisco).
Types of Lipids in Cells
Lipids have diverse structures. The three most important types found in cells are:
Steroids: Bulky, four-ring structure; includes hormones (estrogen, testosterone) and cholesterol.
Fats: Three fatty acids linked to glycerol (triglycerides); primary role is energy storage.
Phospholipids: Glycerol linked to a phosphate group and two hydrocarbon chains; main component of cell membranes.


Formation of Fats and Phospholipids
Fats form via dehydration reactions between the hydroxyl group of glycerol and the carboxyl group of a fatty acid, resulting in an ester linkage. Fatty acids are not polymers.
Phospholipids include a hydrophilic (polar) head and hydrophobic (nonpolar) tails. The head contains glycerol, a phosphate group, and a charged or polar group.

Amphipathic Nature of Membrane Lipids
Membrane lipids are amphipathic, meaning they have both hydrophilic and hydrophobic regions. This property is crucial for membrane formation.
Hydrophilic head: Interacts with water.
Hydrophobic tail: Repels water.
6.2 Phospholipid Bilayers
Amphipathic lipids do not dissolve in water. Instead, they form:
Micelles: Spherical aggregates formed by free fatty acids.
Lipid bilayers: Paired sheets formed by phospholipids, creating the basic structure of cell membranes.

Spontaneous Formation of Bilayers
Phospholipid bilayers form spontaneously in water, requiring no input of energy. The membrane consists of two layers of phospholipid molecules.
Selective Permeability of Lipid Bilayers
Phospholipid bilayers exhibit selective permeability:
Small, nonpolar molecules: Move across quickly (e.g., O2).
Large, polar, or charged molecules: Cross slowly or not at all (e.g., glucose, ions).

Phospholipid Movement within Membranes
Phospholipids are in constant lateral motion but rarely flip to the other side of the bilayer, contributing to membrane fluidity.

6.3 Diffusion and Osmosis Across Lipid Bilayers
Diffusion
Diffusion is the spontaneous movement of molecules and ions from regions of high concentration to low concentration, driven by thermal energy. Equilibrium is reached when molecules are evenly distributed.
Passive transport: Diffusion across membranes without energy input.

Osmosis
Osmosis is a special case of diffusion involving water movement across selectively permeable membranes. Water moves from regions of low solute concentration to high solute concentration, diluting the higher concentration.
Hypertonic: Higher solute concentration outside; cell shrinks.
Hypotonic: Lower solute concentration outside; cell swells.
Isotonic: Equal solute concentration; cell size remains unchanged.


Membranes and Chemical Evolution
The first lipid bilayers likely provided containers for the first replicating molecules, such as RNA. Protocells are simple vesicle-like structures that may have been intermediates in the evolution of cells.
6.4 Proteins Alter Membrane Structure and Function
While phospholipids provide the basic membrane structure, proteins can insert into membranes and form passageways. Proteins are amphipathic, with side chains that can be polar, charged, or nonpolar, allowing them to span the bilayer.
Fluid-Mosaic Model
The fluid-mosaic model describes the membrane as a dynamic mosaic of phospholipids and proteins, with proteins able to move laterally within the membrane.
Membrane Proteins: Integral and Peripheral
Integral (transmembrane) proteins: Span the membrane, with segments facing both interior and exterior.
Peripheral proteins: Bind to membrane lipids without passing through; found on either side of the membrane.
Channel Proteins and Facilitated Diffusion
Ion channels are specialized transmembrane proteins that allow ions to cross membranes, driven by electrochemical gradients (combining concentration and charge gradients).
Channel selectivity: Channel proteins are highly selective, permitting only specific ions or molecules.
Aquaporins: Channel proteins that permit water to cross the membrane.
Gated channels: Open or close in response to signals (e.g., ligand binding, voltage changes).
Facilitated Diffusion via Carrier Proteins
Carrier proteins assist passive transport by selectively picking up solutes on one side of the membrane and releasing them on the other. Example: Glucose transporters.

Pumps and Active Transport
Active transport moves substances against their concentration gradient, requiring energy input (often from ATP). The sodium–potassium pump (Na+/K+-ATPase) is a membrane protein that uses ATP to transport Na+ and K+ ions against their gradients.
Secondary Active Transport (Cotransport)
Electrochemical gradients established by pumps allow cells to engage in secondary active transport, where the movement of one molecule down its gradient powers the movement of another molecule against its gradient.
Summary Table: Passive and Active Mechanisms of Membrane Transport
Transport Type | Energy Required? | Direction | Example |
|---|---|---|---|
Passive (Diffusion) | No | Down gradient | O2 diffusion |
Facilitated Diffusion | No | Down gradient | Glucose via carrier protein |
Active Transport | Yes (ATP) | Against gradient | Na+/K+ pump |
Secondary Active Transport | Indirect (gradient) | Against gradient | Cotransport of glucose |
Key Equations:
Diffusion rate:
Osmosis: (osmotic pressure)
Additional info: The notes expand on brief points with academic context, definitions, and examples to ensure completeness and clarity for exam preparation.
