 Back
BackLipids, Membranes, and the First Cells: Study Notes
Study Guide - Smart Notes

Chapter 6: Lipids, Membranes, and the First Cells
Introduction
This chapter explores the structure and function of lipids, the formation and properties of biological membranes, and the mechanisms by which substances move across these membranes. Understanding these concepts is fundamental to cell biology and the origin of life.
Plasma Membrane: The Boundary of Life
Functions of the Plasma Membrane
Selective Barrier: Regulates entry and exit of materials, maintaining cellular integrity.
Protection: Prevents entry of harmful substances.
Facilitation of Chemical Reactions: Provides a controlled environment for essential biochemical reactions.
6.1 Lipid Structure and Function
Definition and Properties of Lipids
Lipids: Carbon-containing compounds that are insoluble in water due to their high proportion of nonpolar bonds.
Hydrocarbons: Nonpolar molecules composed only of carbon and hydrogen; hydrophobic in nature.
Fatty Acids and Bond Saturation
Fatty Acids: Simple lipids with a hydrocarbon chain and a polar carboxyl group.
Saturated Fatty Acids: Only single bonds between carbons; maximum hydrogen atoms; solid at room temperature.
Unsaturated Fatty Acids: One or more double bonds; causes kinks; liquid at room temperature.
Polyunsaturated: Multiple double bonds; increased fluidity.
Types of Lipids in Cells
Steroids: Bulky, four-ring structure; includes hormones (estrogen, testosterone) and cholesterol.
Fats (Triacylglycerols/Triglycerides): Three fatty acids linked to glycerol; energy storage; formed by dehydration reactions and ester linkages.
Phospholipids: Glycerol linked to a phosphate group and two hydrocarbon chains; primary component of cell membranes.
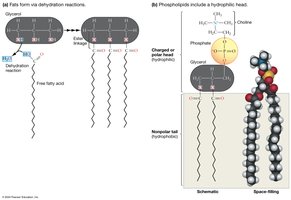
How Membrane Lipids Interact with Water
Amphipathic Nature of Membrane Lipids
Hydrophilic Head: Contains glycerol, phosphate, and a charged or polar group; interacts with water.
Hydrophobic Tail: Nonpolar hydrocarbon chains; repels water.
Amphipathic: Molecules with both hydrophilic and hydrophobic regions (e.g., phospholipids, cholesterol).
6.2 Phospholipid Bilayers
Formation and Structure
Micelles: Spherical aggregates formed by free fatty acids.
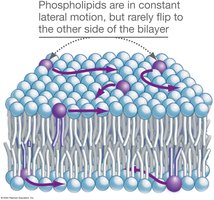
Lipid Bilayers: Paired sheets of phospholipids; form spontaneously in water without energy input.
Selective Permeability
Small/Nonpolar Molecules: Move quickly across bilayers (e.g., O2).
Large/Polar/Charged Molecules: Move slowly or not at all (e.g., glucose).
6.3 How Substances Move Across Lipid Bilayers: Diffusion and Osmosis
Diffusion
Definition: Spontaneous movement of molecules from high to low concentration (down a concentration gradient).
Equilibrium: Random distribution of molecules; no net movement.
Passive Transport: Diffusion across membranes without energy input.
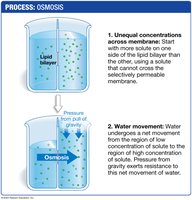
Osmosis
Definition: Diffusion of water across a selectively permeable membrane.
Direction: Water moves from low solute concentration to high solute concentration.
Hypertonic Solution: Higher solute concentration outside; water leaves cell; cell shrinks.
Hypotonic Solution: Lower solute concentration outside; water enters cell; cell swells.
Isotonic Solution: Equal solute concentration; no net water movement; cell size unchanged.
Membranes and Chemical Evolution
Protocells and the Origin of Life
Protocells: Simple vesicle-like structures with nucleic acids; possible intermediates in cellular evolution.
Role of Lipid Bilayers: Provided containers for the first replicating molecules (e.g., RNA).
6.4 Proteins Alter Membrane Structure and Function
Role of Membrane Proteins
Amphipathic Proteins: Insert into membranes; side chains can be polar, charged, or nonpolar.
Fluid-Mosaic Model: Membrane is a dynamic mosaic of phospholipids and proteins; proteins can span the membrane (integral) or attach to one side (peripheral).
Types of Membrane Proteins
Integral (Transmembrane) Proteins: Span the membrane; segments face both interior and exterior.
Peripheral Proteins: Bind to membrane lipids without passing through; found on interior or exterior.
Channel Proteins and Diffusion
Ion Channels: Specialized proteins allowing ions to cross membranes; establish electrochemical gradients.
Channel Selectivity: Channel proteins are selective; only specific ions or molecules pass through.
Aquaporins: Permit water to cross the plasma membrane.
Gated Channels: Open or close in response to signals; regulate flow of ions and molecules.
Facilitated Diffusion: Transmembrane proteins assist passive transport; no energy required.
Carrier Proteins
Mechanism: Selectively pick up solute on one side and release it on the other; example: GLUT-1 (glucose transporter).
Pumps and Active Transport
Active Transport: Moves substances against their concentration gradient; requires energy (usually ATP).
Sodium–Potassium Pump: Uses ATP to transport Na+ and K+ ions against their gradients.
Secondary Active Transport (Cotransport): Uses electrochemical gradients established by pumps to move other molecules; ATP is not directly used for transport.
Summary Table: Types of Membrane Transport
Transport Type | Energy Required? | Direction | Example |
|---|---|---|---|
Passive Diffusion | No | Down gradient | O2 across membrane |
Facilitated Diffusion | No | Down gradient | GLUT-1 (glucose transporter) |
Active Transport | Yes (ATP) | Against gradient | Sodium–Potassium Pump |
Secondary Active Transport | Indirect (gradient) | Against gradient | Cotransport of glucose |
Additional info: The notes expand on brief textbook points to provide definitions, examples, and context for each concept. Images included are directly relevant to the explanation of lipid structure, membrane dynamics, and osmosis.
