 Back
BackLipids: Structure, Types, and Biological Functions
Study Guide - Smart Notes

Lipids: Structure and Classification
Overview of Lipid Classes
Lipids are a diverse group of hydrophobic biomolecules essential for cellular structure and energy storage. The four main classes of lipids are:
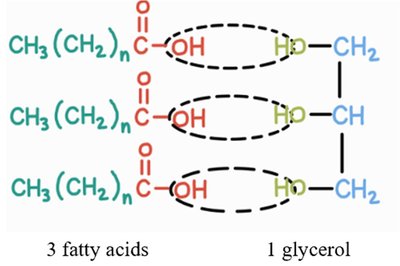
Triglycerides (Triacylglycerols): Commonly known as fats and oils, composed of three fatty acids and one glycerol molecule.
Phospholipids (Phosphoglycerides): Key components of cell membranes, consisting of two fatty acids, one phosphate group, and one glycerol.
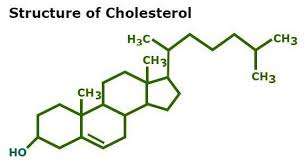
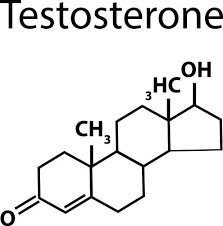
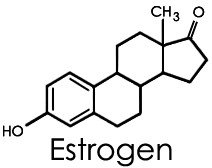
Steroids: Includes cholesterol and sex hormones (e.g., estrogen, progesterone, testosterone), characterized by a four-ring structure.
Waxes: Formed from one saturated fatty acid and one alcohol (not glycerol).
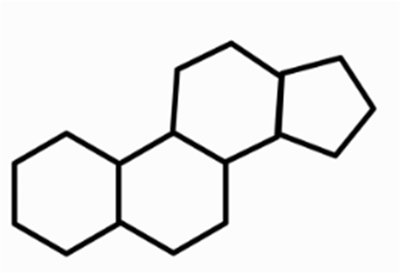
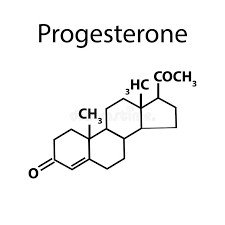
Steroids: Structure and Examples
Steroids are lipids with a characteristic four-ring structure. They play vital roles in cell signaling and membrane structure.
Basic Structure: Four fused carbon rings.
Examples: Cholesterol, estrogen, progesterone, testosterone.
Triglycerides: Structure and Properties
Composition and Formation
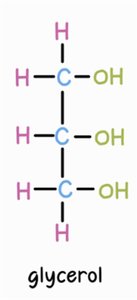
Triglycerides are formed by the esterification of three fatty acids with one glycerol molecule.
Glycerol: A three-carbon alcohol with hydroxyl groups.
Fatty Acids: Organic acids with a carboxyl group (-COOH); can be saturated or unsaturated.
Formation: Dehydration synthesis links fatty acids to glycerol, forming ester bonds and releasing water.

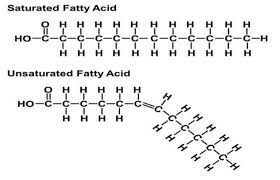
Fatty Acids: Saturated vs. Unsaturated
Fatty acids are classified based on the presence of double bonds.
Saturated Fatty Acids: Lack C=C bonds; solid at room temperature.
Unsaturated Fatty Acids: Have at least one C=C bond; usually liquid at room temperature.

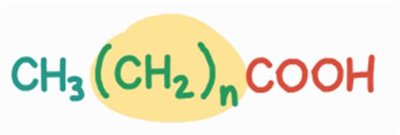
General Formula and Examples
The general formula for fatty acids is:
Saturated:
Monounsaturated:
Polyunsaturated: (where n = number of double bonds)
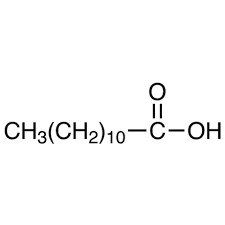
Specific Fatty Acids
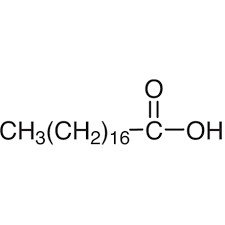
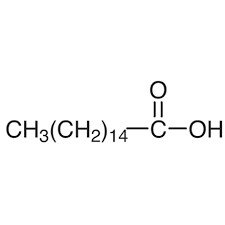
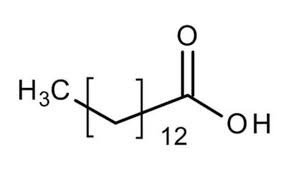
Lauric Acid: C12 saturated fatty acid (12:0),
Stearic Acid: C18 saturated fatty acid (18:0),
Palmitic Acid: C16 saturated fatty acid (16:0),
Myristic Acid: C14 saturated fatty acid (14:0),
Palmitoleic Acid: C16 monounsaturated fatty acid (16:1-7),
Oleic Acid: C18 monounsaturated fatty acid (18:1-9),
Linoleic Acid: C18 di-unsaturated fatty acid (18:2-6,9),
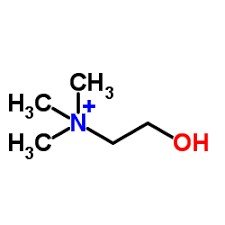
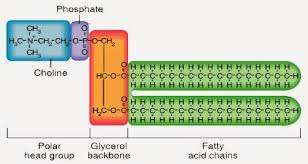
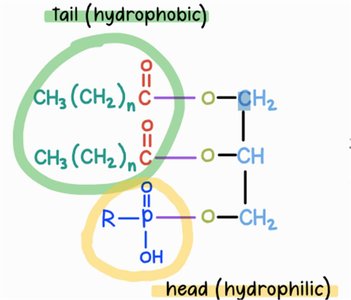
Phospholipids: Structure and Function
Composition and Amphipathic Nature
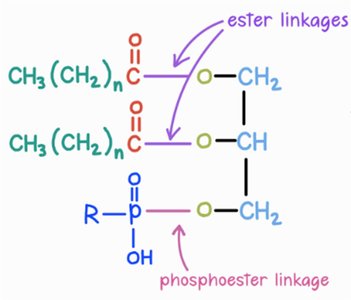
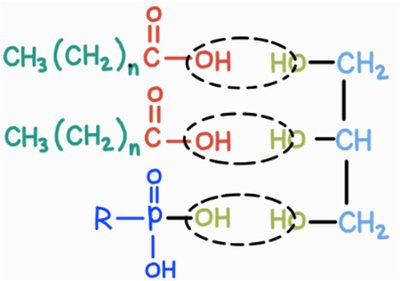
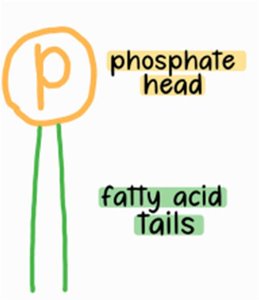
Phospholipids are major components of cell membranes, consisting of two fatty acids, one phosphate group, and one glycerol.
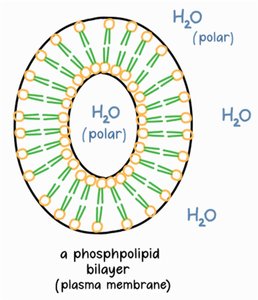
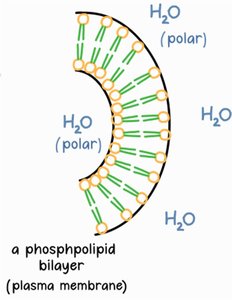
Amphipathic: Possess both hydrophilic (phosphate head) and hydrophobic (fatty acid tails) regions.
Arrangement: Form bilayers in aqueous environments, with tails facing inward and heads facing outward.

Phospholipid Linkages
Ester Linkages: Connect fatty acids to glycerol.
Phosphoester Linkage: Connects phosphate group to glycerol.
Comparison: Triglycerides have three ester linkages; phospholipids have two ester and one phosphoester linkage.

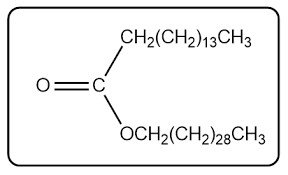
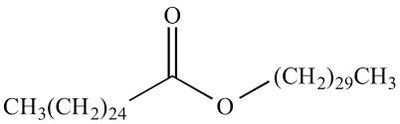
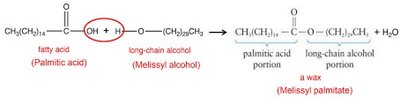
Waxes: Structure and Function
Composition and Examples
Waxes are esters formed from one saturated fatty acid and one long-chain alcohol.
Structure: Not based on glycerol; contain a fatty acid and an alcohol.
Examples: Melissyl palmitate, other wax esters.
Short-Chain Fatty Acids: Acetic Acid
Properties and Biological Role
Acetic acid (CH3COOH) is the simplest short-chain fatty acid, highly water-soluble due to its small size and polar carboxyl group.
Produced by: Microbial fermentation in the gut.
Role: Key in energy metabolism; does not typically form triglycerides.
Summary Table: Saturated vs. Unsaturated Fatty Acids
Saturated | Unsaturated |
|---|---|
Lack C=C bonds Solid at room temperature (25°C) | Have at least one C=C bond |

Example Problems
Triglyceride Molecular Formula Calculation
Problem 1: What is the molecular formula of a triglyceride with lauric acid, stearic acid, and palmitoleic acid? Answer: C49H92O6
Problem 2: A triglyceride has a molecular formula of C51H96O6. Two monomers are palmitoleic acid and myristic acid. What is the third fatty acid? Answer: Stearic acid
Key Terms and Definitions
Hydrophilic: Water-loving; polar.
Hydrophobic: Water-fearing; nonpolar.
Lipophilic: Fat-loving; nonpolar.
Lipophobic: Fat-fearing; polar.
Amphipathic: Molecule with both hydrophilic and hydrophobic regions.
Ester Linkage: Bond formed between an acid and an alcohol via dehydration synthesis.
Phosphoester Linkage: Bond between a phosphate group and an alcohol.
Additional info:
Acetic acid is highly water-soluble due to its small size and polar carboxyl group, making it distinct from longer-chain fatty acids.
Phospholipids are fundamental to cell membrane structure, forming bilayers that separate internal and external environments.
Steroids, unlike other lipids, do not contain fatty acids or glycerol and lack ester or phosphoester linkages.
