 Back
BackMembrane Structure and Function: Study Notes for Biology College Students
Study Guide - Smart Notes

Membrane Structure and Function
Overview of the Plasma Membrane
The plasma membrane is a fundamental cellular structure that defines the boundary of the cell and regulates the movement of substances in and out. It exhibits selective permeability, allowing certain molecules to cross more easily than others, thus maintaining unique internal environments for both the cell and its organelles.
Selective permeability: Essential for cell survival, enables regulation of internal conditions.
Organelles: Each organelle is surrounded by its own membrane, maintaining specialized environments.
Structure of the Plasma Membrane
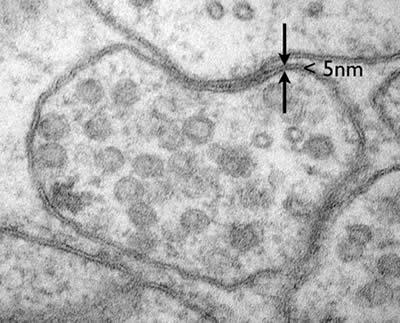
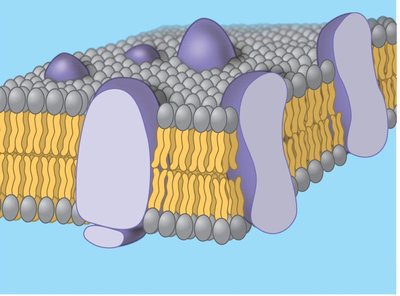
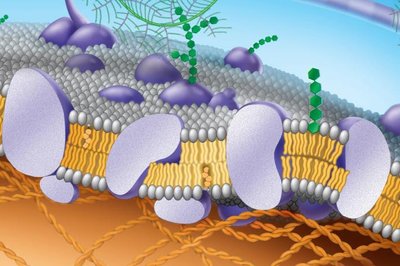
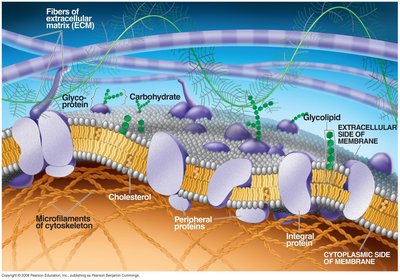
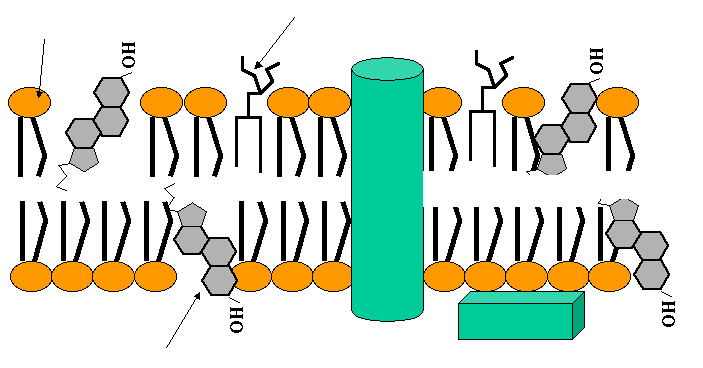
The plasma membrane is composed of a phospholipid bilayer less than 5 nm thick, with embedded proteins and other molecules. The Fluid-Mosaic Model describes this structure as a mosaic of proteins floating in or attached to a fluid lipid bilayer.
Phospholipid bilayer: Two layers of phospholipids with hydrophilic heads facing outward and hydrophobic tails inward.
Proteins: Integral and peripheral proteins are distributed throughout the membrane.
Fluid-Mosaic Model: Proposed by Singer and Nicolson in 1972; emphasizes both fluidity and mosaicism.

Phospholipids: Structure and Properties
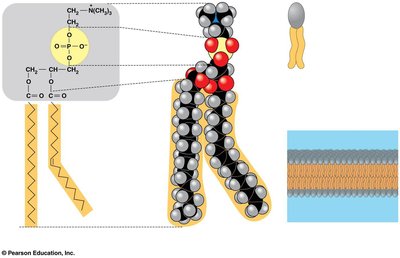
Phospholipids are amphipathic molecules, meaning they contain both hydrophilic (water-loving) and hydrophobic (water-fearing) regions. This property is crucial for the formation of the bilayer.
Hydrophilic head: Composed of choline, phosphate, and glycerol.
Hydrophobic tails: Fatty acids, often with kinks due to cis double bonds (unsaturated).
Amphipathic nature: Drives self-assembly into bilayers in aqueous environments.
Membrane Fluidity and Mosaicism
Membrane fluidity is determined by the types of fatty acids present and the presence of cholesterol. The mosaic aspect refers to the distribution of proteins and lipids in specialized patches.
Fluidity: Lipids and some proteins move laterally; weak hydrophobic interactions allow this movement.
Mosaicism: Membranes are packed with proteins and lipids grouped in defined regions.
Dynamic structure: Current models emphasize a dynamically structured mosaic.
Factors Affecting Membrane Fluidity
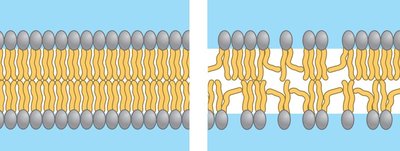
Membrane fluidity is influenced by temperature, the saturation of fatty acid tails, and cholesterol content.
Unsaturated fatty acids: Kinks prevent close packing, increasing fluidity.
Saturated fatty acids: Pack closely, decreasing fluidity.
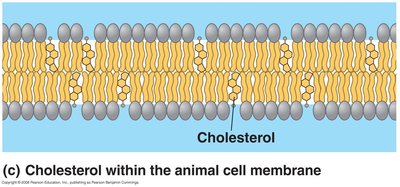
Cholesterol: Acts as a temperature buffer, maintaining fluidity at low temperatures and reducing fluidity at high temperatures.
Membrane Proteins: Types and Functions
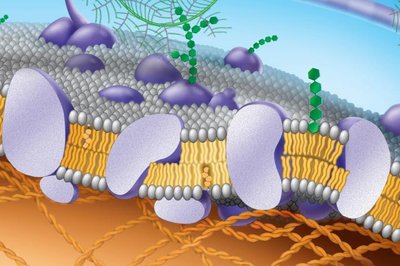
Membrane proteins are essential for the specific functions of the membrane. They are classified as integral (span the membrane) or peripheral (attached to the membrane surface).
Integral proteins: Have hydrophilic and hydrophobic regions, cross the membrane.
Peripheral proteins: Attach to the inner surface, often via integral proteins.
Support: Attachments to the cytoskeleton and extracellular matrix provide structural support.
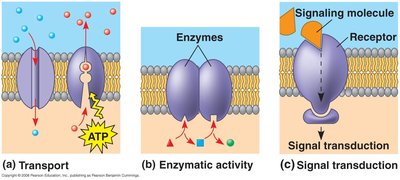
Functions of Membrane Proteins
Membrane proteins perform six major functions:
Transport: Channel and pump proteins move substances across the membrane.
Enzyme activity: Metabolic enzymes catalyze reactions.
Signal transduction: Receptors transmit signals from outside to inside the cell.
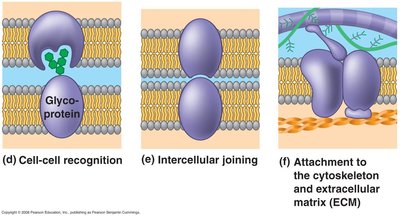
Cell-to-cell recognition: Glycoproteins serve as identification tags.
Intercellular joining: Proteins form junctions between cells.
Attachment: Proteins anchor the membrane to the cytoskeleton and ECM.
Membrane Carbohydrates
Membrane carbohydrates are usually short-branched polysaccharides attached to lipids (glycolipids) or proteins (glycoproteins). They play roles in cell recognition and signaling.
Glycolipids: Lipids with covalently attached carbohydrates.
Glycoproteins: Proteins with covalently attached carbohydrates.
Formation and Embedding of Glycoproteins
Glycoproteins are synthesized in the endoplasmic reticulum (ER), modified in the Golgi apparatus, and embedded in the plasma membrane. The carbohydrate portion ends up on the extracellular side after vesicle fusion.
Phospholipids: Synthesized in RER and SER.
Proteins: Synthesized in RER.
Cholesterol: Synthesized in SER.
Modification: Occurs in the Golgi apparatus.
Transport Across the Membrane
The plasma membrane regulates the transport of molecules, allowing some to pass easily while others require assistance.
Easily crossing molecules: Nonpolar, hydrophobic molecules (e.g., O2, CO2, hydrocarbons).
Difficult or non-crossing molecules: Polar molecules and ions (e.g., water, Na+, Cl-, glucose).
Transport proteins: Facilitate movement of polar molecules and ions.
Mechanisms of Transport
There are several mechanisms by which molecules move across the membrane:
Passive transport: Includes diffusion and osmosis; does not require energy.
Facilitated diffusion: Passive transport aided by proteins (channel and carrier proteins).
Active transport: Uses energy (usually ATP) to move molecules against their concentration gradient.
Cotransport: Coupled transport of two substances, one down its gradient and one against.
Exocytosis: Secretion of large molecules by vesicle fusion.
Endocytosis: Uptake of molecules by vesicle formation (phagocytosis, pinocytosis, receptor-mediated).
Diffusion and Osmosis
Diffusion is the movement of molecules from high to low concentration. Osmosis is the movement of water across a selectively permeable membrane.
Diffusion: Spontaneous, each molecule moves down its own gradient.
Osmosis: Water moves from areas of higher free water concentration to lower, or from lower solute concentration to higher.
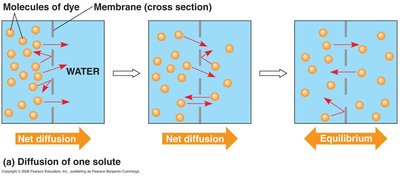
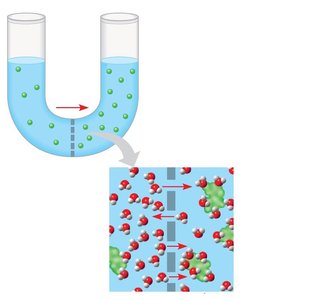
Tonicity and Its Effects on Cells
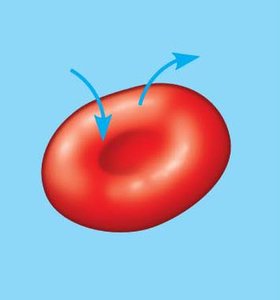
Tonicity describes the ability of a solution to cause a cell to gain or lose water, depending on the concentration of nonpenetrating solutes.
Isotonic: Solute concentration equal inside and outside; no net water movement.
Hypotonic: Lower solute concentration outside; water enters cell, may burst.
Hypertonic: Higher solute concentration outside; water leaves cell, may shrink.

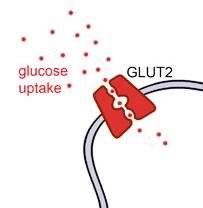
Facilitated Diffusion
Facilitated diffusion is passive transport aided by proteins. Channel proteins and carrier proteins help molecules move down their concentration gradient without energy input.
Channel proteins: Provide corridors for specific molecules.
Carrier proteins: Change shape to move molecules across.
Example: Glucose enters pancreatic beta cells via Glut2 carrier protein.
Active Transport
Active transport uses energy (usually ATP) to move molecules against their concentration gradient. Only carrier proteins are involved.
Sodium-potassium pump: Moves 3 Na+ out and 2 K+ in per cycle, maintaining membrane potential.
Membrane potential: Negative inside due to unequal ion distribution; electrochemical gradient drives ion movement.
Cotransport
Cotransport involves the coupled movement of two substances, one down its gradient and one against, using energy stored in the membrane.
Example: Sucrose-H+ cotransporter in plant cells.
ATP: Indirectly provides energy for cotransport.
Bulk Transport: Exocytosis and Endocytosis
Cells move large molecules via exocytosis (secretion) and endocytosis (uptake). Endocytosis includes phagocytosis, pinocytosis, and receptor-mediated endocytosis.
Exocytosis: Vesicles fuse with plasma membrane to release contents.
Endocytosis: Cell forms vesicles to take in substances; receptor-mediated endocytosis allows specific uptake.
Electrochemical Gradient
The electrochemical gradient is the combined effect of the concentration gradient and membrane potential on ion movement. It influences the direction and rate of ion transport across membranes.
Membrane potential: Favors movement of cations into the cell and anions out.
Example: Diffusion of Na+ into nerve cells during stimulation.
Summary Table: Types of Membrane Transport
Transport Type | Energy Required | Protein Involved | Direction | Example |
|---|---|---|---|---|
Diffusion | No | No | Down gradient | O2, CO2 |
Osmosis | No | Sometimes (aquaporins) | Down gradient | Water |
Facilitated Diffusion | No | Yes (channel/carrier) | Down gradient | Glucose, ions |
Active Transport | Yes (ATP) | Yes (carrier) | Against gradient | Na+/K+ pump |
Cotransport | Indirect (ATP) | Yes (cotransporter) | Against gradient (one molecule) | Sucrose-H+ cotransport |
Exocytosis | Yes | Vesicle fusion | Out of cell | Chylomicron release |
Endocytosis | Yes | Vesicle formation | Into cell | Phagocytosis, pinocytosis |
Key Equations
Membrane Potential:
Diffusion:
Osmosis:
Additional info:
Membrane fluidity is crucial for the function of enzymatic proteins and for cellular processes such as endocytosis and exocytosis.
Organisms in cold environments adapt by increasing unsaturated fatty acids in their membranes.
Familial hypercholesterolemia is caused by defective LDL receptor proteins, leading to high blood cholesterol.
