 Back
BackLEC 9: Metabolism and Energy: Thermodynamics in Biological Systems
Study Guide - Smart Notes

Metabolism and Energy
Overview of Metabolism
Metabolism refers to the totality of an organism’s chemical reactions, managing both material and energy resources within the cell. It is divided into two main categories: anabolism (building molecules) and catabolism (breaking down molecules).
Anabolism: Synthesis of complex molecules from simpler ones (e.g., protein synthesis from amino acids). Requires energy input, often using ATP.
Catabolism: Breakdown of complex molecules into simpler ones (e.g., glucose breakdown in the presence of oxygen). Releases energy, some of which is captured in ATP.
ATP (adenosine triphosphate) acts as the energy currency, cycling between ATP and ADP as energy is used and regenerated.
Cells function as chemical factories, performing thousands of reactions simultaneously to sustain life.
Energy in Biological Systems
Forms of Energy
Energy is defined as the capacity to do work or cause change. In biological systems, energy exists in various forms:
Kinetic Energy: Energy of motion (e.g., heat, moving objects, radiant energy of light).
Potential Energy: Stored energy (e.g., chemical energy in bonds, concentration gradients, electrical potential).
Energy can be transformed from one form to another, but the total amount remains constant.
The First Law of Thermodynamics
The First Law of Thermodynamics states that energy cannot be created or destroyed, only transformed or transferred. In biological systems, this means that cells convert energy from one form to another (e.g., sunlight to chemical energy in sugars).

For example, the energy in gasoline is converted to movement and heat in a car, with some energy lost as heat due to friction.
The Second Law of Thermodynamics
The Second Law of Thermodynamics states that every energy transfer or transformation increases the entropy (disorder) of the universe. Some energy becomes unusable, often lost as heat.
Entropy (S): A measure of disorder or randomness. As entropy increases, less energy is available to do work.
Heat is considered the lowest grade of energy because it increases molecular randomness and is difficult to harness for work.

Biological systems must continually obtain and use energy to maintain order and counteract entropy.
Free Energy and Spontaneity of Reactions
Gibbs Free Energy (G)
Gibbs free energy (G) is the portion of a system’s energy that can perform work. The change in free energy (ΔG) during a process is crucial for understanding whether a reaction will occur spontaneously.
ΔG = Gfinal – Ginitial
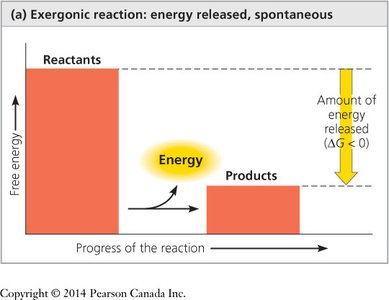
If ΔG is negative, the reaction is spontaneous (exergonic).
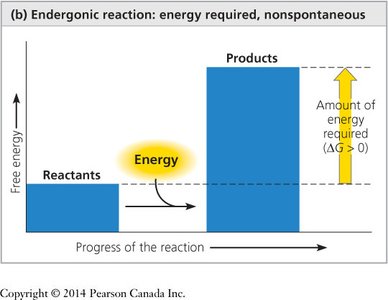
If ΔG is positive, the reaction is non-spontaneous (endergonic).
Systems with high free energy are more ordered and have low entropy, while systems with low free energy are more disordered and have high entropy.
Calculating Free Energy Change
The change in free energy for a chemical reaction can be calculated using the Gibbs equation:
ΔH: Change in enthalpy (total energy)
T: Temperature in Kelvin
ΔS: Change in entropy
This equation helps predict whether a reaction is energetically and/or entropically favorable and whether it will be spontaneous under given conditions.
Examples of Spontaneous and Non-Spontaneous Reactions
Exergonic Reactions: Release free energy (ΔG < 0), spontaneous (e.g., catabolic pathways like glycolysis).
Endergonic Reactions: Absorb free energy (ΔG > 0), non-spontaneous (e.g., anabolic pathways like DNA synthesis).
Energy Coupling in Cells
How Cells Drive Endergonic Reactions
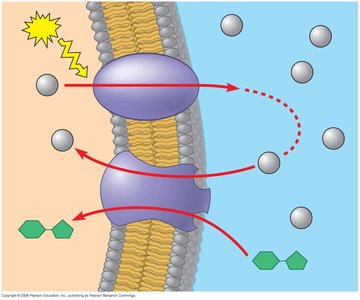
Cells perform work by coupling exergonic and endergonic reactions. The energy released from exergonic reactions (e.g., ATP hydrolysis) is used to drive endergonic processes (e.g., active transport, biosynthesis).
Chemical Work: Synthesis of macromolecules
Transport Work: Pumping substances across membranes
Mechanical Work: Movement of cellular structures
ATP: The Energy Currency of the Cell
Adenosine triphosphate (ATP) stores and transfers energy within cells. The hydrolysis of ATP releases energy that can be used for cellular work:
ATP consists of adenine, ribose, and three phosphate groups.
Energy is stored in the phosphoanhydride bonds between phosphate groups.
Metabolic Pathways and Cellular Respiration
Catabolic Pathways
Catabolic pathways, such as glycolysis, release free energy in a series of reactions. This energy is captured in ATP and used to power cellular processes.
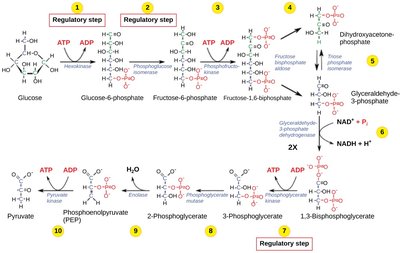
Glycolysis: Breakdown of glucose into pyruvate, generating ATP and NADH.
ATP is regenerated from ADP and inorganic phosphate (Pi) during cellular respiration.
Summary Table: Key Thermodynamic Concepts in Biology
Concept | Definition | Biological Example |
|---|---|---|
Metabolism | Total chemical reactions in a cell | Catabolism and anabolism |
First Law of Thermodynamics | Energy cannot be created or destroyed | Photosynthesis converts light to chemical energy |
Second Law of Thermodynamics | Entropy of the universe increases | Heat loss during metabolism |
Gibbs Free Energy (G) | Energy available to do work | ATP hydrolysis powers cellular work |
Exergonic Reaction | Releases energy (ΔG < 0) | Glucose breakdown |
Endergonic Reaction | Requires energy (ΔG > 0) | Protein synthesis |
Additional info: The notes above integrate foundational thermodynamic principles with their application to biological systems, as required for a college-level understanding of metabolism and energy transformations in cells.
