 Back
BackMetabolism and Energy Transformations in Biological Systems (chpt. 8)
Study Guide - Smart Notes

Metabolism: Overview
Definition and Types of Metabolic Reactions
Metabolism encompasses all chemical reactions occurring within an organism, including both the synthesis and breakdown of molecules. These reactions are organized into metabolic pathways, each step catalyzed by a specific enzyme.
Catabolic Reactions: Break down complex molecules into simpler compounds, releasing energy. Example: Cellular respiration.
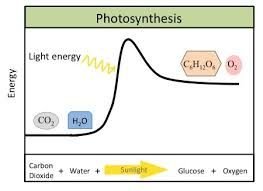
Anabolic Reactions: Consume energy to build complex molecules from simpler ones. Example: Photosynthesis.
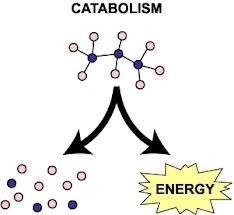
Metabolic Pathways
Metabolic pathways consist of a series of steps, each requiring a specific enzyme. The organization of these pathways allows for regulation and efficiency in cellular processes. 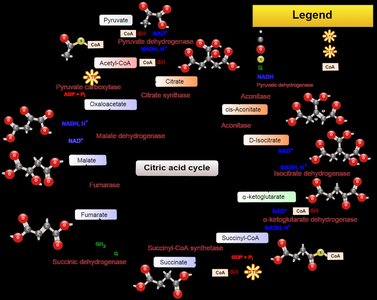
Energy in Biological Systems
Types of Energy
Energy is the capacity to cause change and perform work. Biological systems utilize several forms of energy:
Kinetic Energy: Energy of movement.
Thermal Energy: Kinetic energy of atoms/molecules; heat is the transfer of thermal energy.
Light Energy: Used in photosynthesis.
Chemical Energy: Energy stored in molecular bonds, released during catabolic reactions.
Potential Energy: Energy due to position or structure.

Chemical Energy and Catabolism
Chemical energy is a form of potential energy stored in molecules, released during catabolic reactions such as cellular respiration. 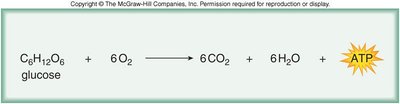
Thermodynamics in Biology
First Law of Thermodynamics
The first law states that energy cannot be created or destroyed, only transferred or transformed. Biological systems, as open systems, exchange energy with their surroundings.
Example: A bear converts chemical energy from food into kinetic energy for movement.

Second Law of Thermodynamics
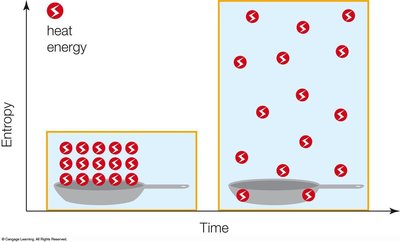
The second law states that every energy transfer increases the entropy (disorder) of the universe. Energy disperses, often as heat, and systems tend toward greater disorder.
Entropy: Measure of disorder; dispersal of energy.
Spontaneous Processes: Occur without energy input, increase entropy.
Free Energy and Metabolic Reactions
Free Energy (G) and Its Change (ΔG)
Free energy is the portion of a system's energy available to do work at constant temperature and pressure. The change in free energy () determines whether a reaction is spontaneous.
Formula: where = change in enthalpy (total energy), = temperature in Kelvin, = change in entropy.
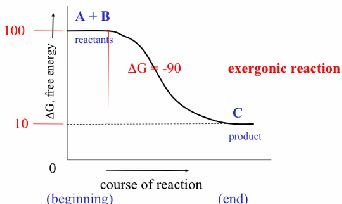
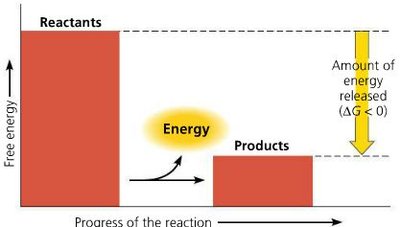
Spontaneous Reactions: (exergonic)
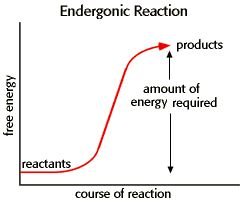
Nonspontaneous Reactions: (endergonic)
Exergonic and Endergonic Reactions
Exergonic Reaction: Net release of free energy (), spontaneous. Example: Cellular respiration.
Endergonic Reaction: Absorbs free energy (), nonspontaneous. Example: Photosynthesis.
ATP and Energy Coupling
Structure and Function of ATP
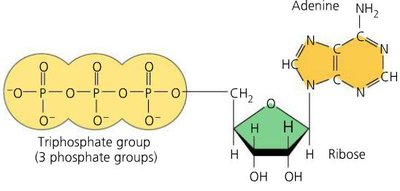
ATP (Adenosine Triphosphate) is the primary energy carrier in cells, consisting of ribose, adenine, and three phosphate groups.
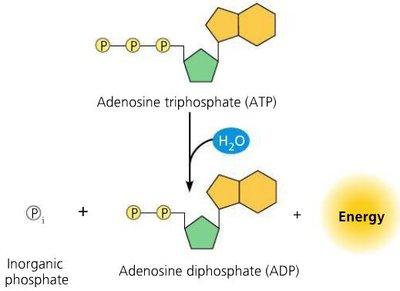
ATP Hydrolysis: Exergonic reaction; releases 7.3 kcal/mol ATP.
Energy Coupling: ATP hydrolysis drives endergonic reactions by transferring a phosphate group to a reactant (phosphorylated intermediate).
ATP Cycle
ATP is continuously regenerated from ADP and inorganic phosphate, a process powered by cellular respiration (endergonic).
Example: A muscle cell uses and regenerates millions of ATP molecules per second.
Enzymes and Regulation of Metabolism
Enzyme Function and Activation Energy
Enzymes are biological catalysts that speed up reactions by lowering the activation energy barrier.
Activation Energy: Energy required to initiate a reaction.
Enzyme Specificity: Enzymes bind specific substrates at their active site, forming an enzyme-substrate complex.
Induced Fit: Active site changes shape to fit substrate.
Factors Affecting Enzyme Activity
Temperature: Enzyme activity increases with temperature up to an optimal point, then decreases due to denaturation.
pH: Each enzyme has an optimal pH; extreme pH can denature enzymes.
Cofactors and Coenzymes: Nonprotein helpers (ions or organic molecules) required for enzyme function.
Enzyme Regulation
Competitive Inhibitors: Bind to active site, blocking substrate.
Noncompetitive Inhibitors: Bind elsewhere, changing enzyme shape and reducing activity.
Allosteric Regulation: Regulatory molecules bind at sites other than the active site, activating or deactivating the enzyme.
Feedback Inhibition: End product of a pathway inhibits an early step, preventing overproduction.
Summary Table: Exergonic vs. Endergonic Reactions
Reaction Type | ΔG | Energy Flow | Spontaneity | Example |
|---|---|---|---|---|
Exergonic | < 0 | Energy released | Spontaneous | Cellular Respiration |
Endergonic | > 0 | Energy absorbed | Nonspontaneous | Photosynthesis |
Key Equations
Free Energy Change:
ATP Hydrolysis:
End of Chapter Review
Catabolism is to anabolism as exergonic is to endergonic.
Metabolic processes that can occur without a net influx of energy are exergonic (e.g., cellular respiration).
Enzymes in thermophilic bacteria have high optimal temperatures, allowing activity in hot springs.
