 Back
BackTopic4 Metabolism: Energy Transformation and Enzyme Function in Cells
Study Guide - Smart Notes

Metabolism: Energy Transformation and Enzyme Function in Cells
Introduction to Metabolism
Metabolism encompasses all chemical reactions that occur within living organisms, enabling them to maintain life, grow, and reproduce. These reactions are organized into metabolic pathways, where each step is catalyzed by a specific enzyme, transforming a starting molecule into a final product through a series of intermediates.
Anabolic pathways: Synthesize complex molecules from simpler ones, requiring energy input.
Catabolic pathways: Break down complex molecules into simpler ones, releasing energy.
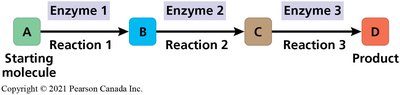
Example: The synthesis of proteins from amino acids (anabolic) and the breakdown of glucose during cellular respiration (catabolic).
Forms of Energy in Biological Systems
Energy is the capacity to do work and exists in various forms within biological systems. Organisms transform energy from one form to another to sustain life processes.
Kinetic energy: Energy of motion (e.g., movement of molecules).
Thermal energy: Random movement of atoms and molecules, perceived as heat.
Potential energy: Stored energy due to position or structure (e.g., chemical bonds).
Chemical energy: A form of potential energy stored in molecular bonds, available for release in chemical reactions.

Example: A diver on a platform has potential energy, which is converted to kinetic energy during the dive.
Laws of Energy Transformation (Thermodynamics)
Biological systems obey the laws of thermodynamics, which govern energy transformations and the direction of metabolic processes.
First Law (Conservation of Energy): Energy cannot be created or destroyed, only transformed from one form to another.
Second Law: Every energy transformation increases the entropy (disorder) of the universe. Some energy is always lost as heat, making energy conversions inefficient.

Example: When a bear eats fish, chemical energy is converted to kinetic energy and heat, increasing entropy.
Biological Order and Entropy
Living organisms maintain order and complexity despite the universal tendency toward disorder (entropy). They do this by coupling energy-releasing (exergonic) reactions with energy-consuming (endergonic) processes, thus maintaining low entropy locally while increasing entropy in their surroundings.

Example: The evolution of complex organisms from simpler ancestors does not violate the second law of thermodynamics because organisms are open systems exchanging energy and matter with their environment.
Free Energy: Spontaneous and Non-Spontaneous Reactions
Free energy (Gibbs free energy, G) is the portion of a system's energy that can perform work at constant temperature and pressure. The change in free energy (ΔG) determines whether a process is spontaneous.
ΔG < 0: Spontaneous (exergonic) reaction; releases energy.
ΔG > 0: Non-spontaneous (endergonic) reaction; requires energy input.
ΔG = 0: System is at equilibrium; no net work can be done.
The equation for free energy change is:
Where:
ΔH: Change in enthalpy (total energy)
T: Absolute temperature in Kelvin
ΔS: Change in entropy
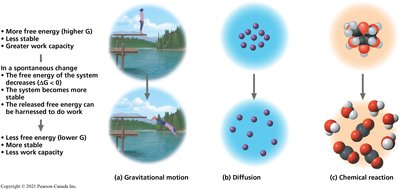
Example: Diffusion of dye in water is a spontaneous process (ΔG < 0).
Exergonic and Endergonic Reactions
Metabolic reactions are classified based on their energy profiles:
Exergonic reactions: Release free energy; products have less free energy than reactants (e.g., cellular respiration).
Endergonic reactions: Absorb free energy; products have more free energy than reactants (e.g., photosynthesis).


Example: Cellular respiration is exergonic (), while photosynthesis is endergonic ().
ATP: Connecting Exergonic and Endergonic Reactions
Adenosine triphosphate (ATP) is the primary energy currency of the cell. It couples exergonic and endergonic reactions, allowing cells to perform work.
ATP hydrolysis: ATP + H2O → ADP + Pi + energy
The energy released is used for cellular work (chemical, transport, and mechanical).
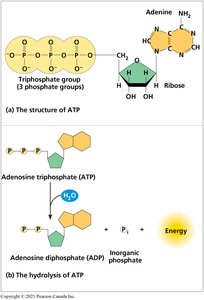
Example: ATP hydrolysis powers muscle contraction, active transport, and biosynthesis.

Enzymes: Biological Catalysts
Enzymes are proteins that catalyze biochemical reactions by lowering the activation energy required for the reaction to proceed, thus increasing the reaction rate without being consumed.
Specificity: Each enzyme acts on a specific substrate, determined by the enzyme's three-dimensional structure.
Mechanisms: Enzymes may orient substrates, strain bonds, provide a favorable microenvironment, or participate directly in the reaction.
Example: Sucrase catalyzes the hydrolysis of sucrose into glucose and fructose.
Summary Table: Key Concepts in Metabolism
Concept | Definition | Example |
|---|---|---|
Metabolism | All chemical reactions in an organism | Cellular respiration, photosynthesis |
Anabolic Pathway | Builds complex molecules, requires energy | Protein synthesis |
Catabolic Pathway | Breaks down molecules, releases energy | Glycolysis |
ATP | Main energy currency of the cell | Muscle contraction |
Enzyme | Protein catalyst, lowers activation energy | Sucrase |
Additional info: These notes integrate foundational concepts from cell biology, biochemistry, and physiology, providing a comprehensive overview of metabolism suitable for college-level biology students.
