 Back
BackMitosis and Cell Cycle Control: Structure, Regulation, and Loss of Control
Study Guide - Smart Notes

Mitosis and the Eukaryotic Cell Cycle
Overview of the Cell Cycle
The eukaryotic cell cycle is a series of events that cells undergo to grow and divide. It consists of interphase (G1, S, G2 phases), mitosis (M phase), and cytokinesis (C phase). Mitosis itself is subdivided into five distinct phases: prophase, prometaphase, metaphase, anaphase, and telophase.
Interphase: The cell grows (G1), replicates DNA (S), and prepares for mitosis (G2).
Mitosis: Nuclear division occurs, ensuring each daughter cell receives an identical set of chromosomes.
Cytokinesis: Division of the cytoplasm, resulting in two separate cells.
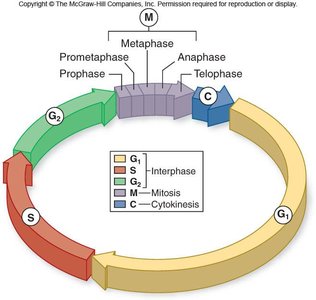
Phases of Mitosis
Mitosis is a highly regulated process that ensures accurate segregation of chromosomes. Each phase is characterized by specific structural changes and events.
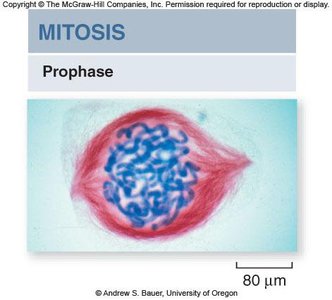
Prophase
During prophase, chromosomes condense and become visible, the nuclear envelope begins to dissolve, and the spindle apparatus starts to form from centrosomes.
Chromosome condensation: Chromatin fibers coil tightly, making chromosomes visible under a microscope.
Spindle formation: Microtubules assemble from centrosomes, which migrate to opposite poles of the cell.
Nuclear envelope breakdown: Prepares the cell for chromosome segregation.
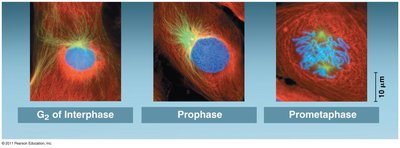
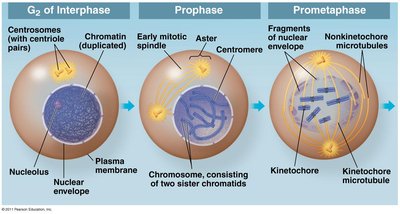
Prometaphase
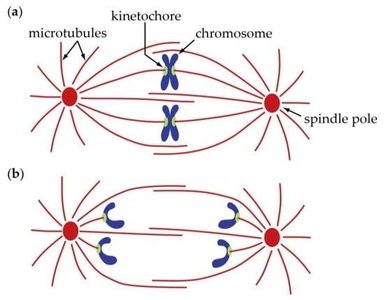
Prometaphase is marked by the complete dissolution of the nuclear envelope and the attachment of chromosomes to spindle microtubules via kinetochores.
Kinetochores: Protein structures at the centromere of each chromosome, serving as attachment sites for spindle microtubules.
Microtubule attachment: Chromosomes begin to move toward the cell center.
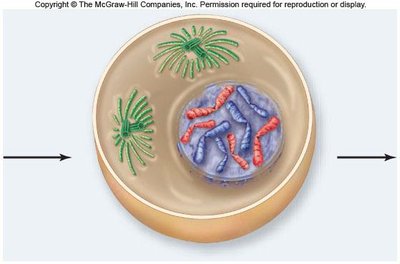
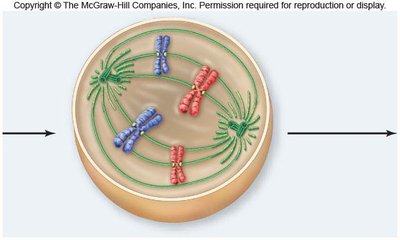
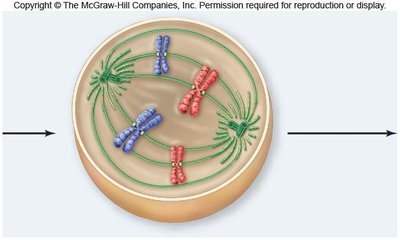
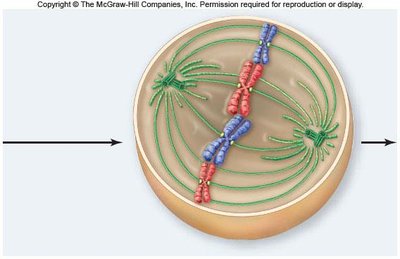
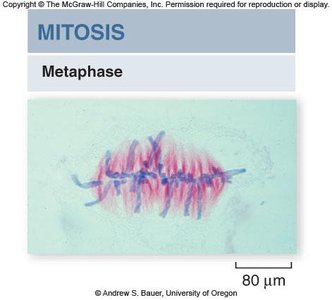
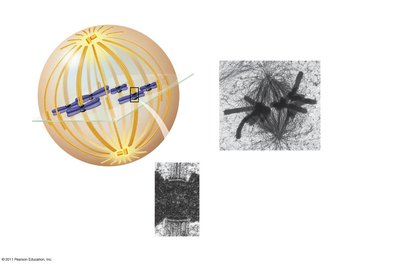
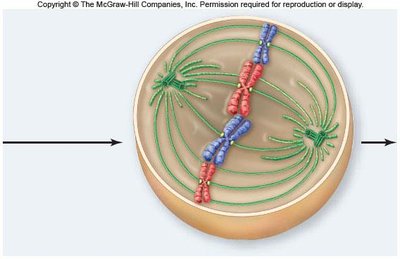
Metaphase
In metaphase, chromosomes align at the metaphase plate, an imaginary plane equidistant from the spindle poles. Microtubules pull at kinetochores, but cohesins keep sister chromatids together.
Metaphase plate: The central region where chromosomes align.
Cohesins: Proteins that hold sister chromatids together until anaphase.
Spindle apparatus: Ensures proper chromosome alignment and segregation.
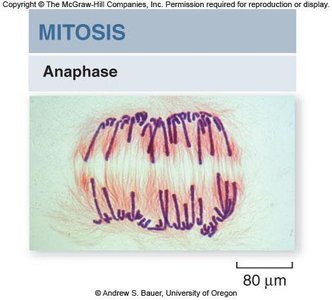
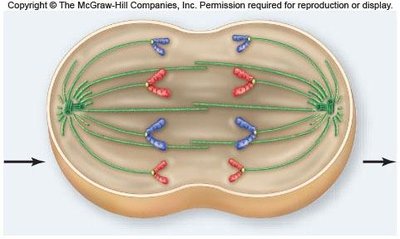
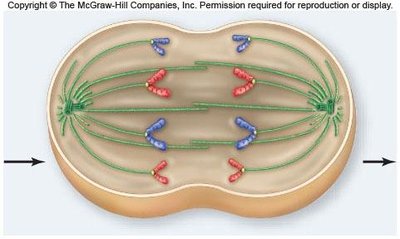
Anaphase
Anaphase begins when cohesins are cleaved, allowing sister chromatids to separate and move toward opposite poles. Nonkinetochore microtubules elongate the cell.
Chromatid separation: Each chromatid becomes an independent chromosome.
Cell elongation: Nonkinetochore microtubules push against each other, lengthening the cell.
Cytokinesis initiation: Begins during anaphase or telophase.
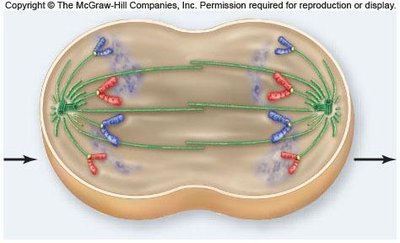
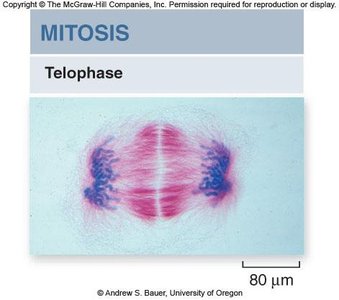
Telophase
Telophase is characterized by the reformation of the nuclear envelope, dissolution of the spindle apparatus, and uncoiling of chromosomes. Mitosis concludes with two genetically identical nuclei.
Nuclear envelope reformation: Chromosomes are enclosed in new nuclei.
Chromosome decondensation: Chromosomes return to their less condensed state.
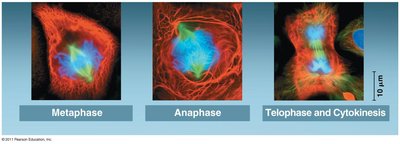
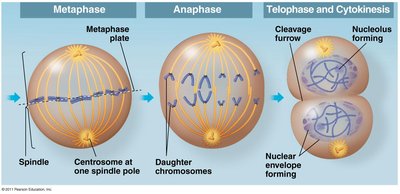
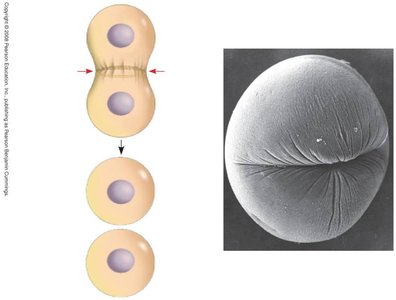
Cytokinesis
Cytokinesis is the physical division of the cell into two daughter cells. In animal cells, this occurs via cleavage furrow formation; in plant cells, a cell plate forms.
Cleavage furrow: Contractile ring of actin filaments pinches the cell in two.
Cell plate (plants): Vesicles fuse to form a new cell wall between daughter cells.



Control of the Eukaryotic Cell Cycle
The cell cycle is tightly regulated by checkpoints and signaling molecules to ensure proper division and prevent errors.
Checkpoints: G1, G2, and M checkpoints monitor cell size, DNA integrity, and chromosome attachment.
Cyclins and CDKs: Cyclin proteins and cyclin-dependent kinases regulate progression through the cell cycle.
Growth factors: External signals, such as platelet-derived growth factor (PDGF), stimulate cell division.
G1 Checkpoint
The G1 checkpoint is the most critical, determining whether the cell will proceed with division or enter a quiescent state (G0).
Go-ahead signal: If received, the cell continues through the cycle; if not, it enters G0.
G2 Checkpoint
Ensures DNA replication is complete and undamaged before mitosis begins. Cyclin activity is regulated by proteins that detect DNA damage.
M Checkpoint (Spindle Checkpoint)
Ensures all chromosomes are properly attached to the spindle before anaphase begins. Activation of separase allows sister chromatids to separate.
Growth Factors and Cell Cycle Regulation
Growth factors, such as PDGF, trigger intracellular signaling pathways that promote cell division, especially during wound healing.
Density-Dependent and Anchorage Dependence
Density-dependent inhibition: Crowded cells stop dividing.
Anchorage dependence: Cells must be attached to a substratum to divide.
Loss of Cell Cycle Control and Cancer
Cancer arises from the loss of normal cell cycle regulation, often due to mutations in key regulatory genes.
Tumor-suppressor genes: Prevent development of cells with mutations (e.g., p53).
Proto-oncogenes: Encode growth factor receptors and signaling proteins; when mutated, become oncogenes that drive uncontrolled growth.
Cancer cells: Ignore normal regulatory signals, may produce their own growth factors, and exhibit neither density-dependent inhibition nor anchorage dependence.
Benign vs. malignant tumors: Benign tumors remain localized; malignant tumors invade tissues and may metastasize.
Summary Table: Cell Cycle Checkpoints
Checkpoint | Main Function | Key Regulators |
|---|---|---|
G1 | Cell size, DNA integrity, external signals | Cyclins, CDKs |
G2 | DNA replication completion, DNA damage | Cyclins, DNA damage sensors |
M (Spindle) | Chromosome attachment to spindle | Regulatory complex, separase |
Key Terms and Concepts
Chromatid: One of two identical halves of a duplicated chromosome.
Centromere: Region where sister chromatids are joined.
Kinetochores: Protein complexes at centromeres for spindle attachment.
Cohesins: Proteins holding sister chromatids together.
Spindle apparatus: Structure made of microtubules for chromosome movement.
Relevant Equations
Cell cycle progression is regulated by cyclin-CDK complexes:
Mitotic spindle force:
Example: Colchicine and Mitotic Spindle Inhibition
Colchicine blocks spindle formation, preventing chromosome alignment and halting the cell cycle at metaphase.
Application: Used experimentally to study mitosis and clinically to treat gout by inhibiting cell division.
Summary
Mitosis and cell cycle control are fundamental to cellular reproduction and organismal growth. Disruption of these processes can lead to diseases such as cancer, highlighting the importance of regulatory checkpoints and signaling pathways.
