 Back
BackNutrient Cycling and Biogeochemical Cycles in Ecosystems
Study Guide - Smart Notes

Nutrient Cycling in Ecosystems
Overview of Biogeochemical Cycles
Biogeochemical cycles describe the movement of essential elements and compounds through living organisms (biotic components) and the physical environment (abiotic components). These cycles are crucial for sustaining life, as they ensure the continuous availability of nutrients required for biological processes.
Key Cycles: Water, Carbon, Nitrogen, and Phosphorus cycles are fundamental to ecosystem function.
Importance: These cycles recycle elements, supporting primary production, decomposition, and the structure of biological molecules.
The Water (Hydrologic) Cycle
Importance and Distribution
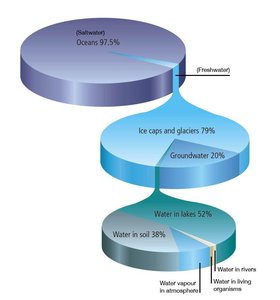
The water cycle is essential for all living organisms, as water is the medium for most biochemical reactions and is involved in nutrient transport and waste removal. The majority of Earth's water is found in the oceans, with only a small fraction available as freshwater.
Oceans: Contain about 97% of Earth's water (mostly saltwater).
Freshwater: Found in ice caps, glaciers, groundwater, lakes, soil, rivers, and living organisms.
Key Processes in the Water Cycle
The water cycle involves the continuous movement of water between the atmosphere, land, and oceans through various physical processes.
Evaporation: Water changes from liquid to vapor due to solar energy.
Condensation: Water vapor cools and forms clouds.
Precipitation: Water returns to Earth's surface as rain, snow, fog, or hail.
Transpiration: Water loss from plants to the atmosphere.
Runoff and Groundwater Flow: Water moves over land and through soil to return to oceans.

The Carbon Cycle
Role of Carbon in Living Organisms
Carbon is a fundamental component of all organic molecules, including carbohydrates, proteins, nucleic acids, and lipids. The carbon cycle describes the movement of carbon among the atmosphere, living organisms, and other reservoirs.
Photosynthesis: Plants and other photosynthetic organisms convert atmospheric CO2 into organic molecules (sugars).
Cellular Respiration: Organisms break down sugars to release energy, returning CO2 to the atmosphere.
Major Reservoirs: Atmosphere, living organisms, fossil fuels, soil, and sediments in aquatic ecosystems.

Key Processes in the Carbon Cycle
Photosynthesis:
Cellular Respiration:
Decomposition: Breakdown of dead organisms returns carbon to the soil and atmosphere.
Combustion: Burning of fossil fuels and wood releases CO2.
The Nitrogen Cycle
Role of Nitrogen in Living Organisms
Nitrogen is a key element in proteins and nucleic acids. Although atmospheric nitrogen (N2) is abundant, most organisms cannot use it directly and rely on specific processes to convert it into usable forms.
Major Reservoir: N2 gas makes up about 78% of the atmosphere.
Other Reservoirs: Soil, aquatic sediments, dissolved in water, and biomass.
Key Processes in the Nitrogen Cycle
Nitrogen Fixation: Conversion of N2 gas to ammonia (NH3) by bacteria and industrial processes.
Ammonification: Decomposition of organic nitrogen to ammonia.
Nitrification: Conversion of ammonia to nitrite (NO2-) and then to nitrate (NO3-) by bacteria.
Denitrification: Conversion of nitrate back to N2 gas by bacteria, returning it to the atmosphere.

The Phosphorus Cycle
Role of Phosphorus in Living Organisms
Phosphorus is a vital component of phospholipids (cell membranes), nucleic acids (DNA, RNA), and ATP (energy transfer molecule). Unlike other cycles, phosphorus does not have a significant atmospheric component.
Major Reservoirs: Sedimentary rocks of marine origin, soil, and dissolved in oceans.
Biological Importance: Essential for energy transfer, genetic material, and cell structure.
Key Processes in the Phosphorus Cycle
Weathering: Releases phosphate ions (PO43-) from rocks into soil and water.
Uptake by Plants: Plants absorb phosphate from soil; animals obtain it by eating plants.
Decomposition: Returns phosphorus to soil and water from dead organisms and waste.
Sedimentation: Phosphorus can be lost to deep sediments, eventually returning via geological uplift.

Summary Table: Major Biogeochemical Cycles
Cycle | Main Reservoir(s) | Key Processes | Biological Importance |
|---|---|---|---|
Water | Oceans, atmosphere, freshwater bodies | Evaporation, condensation, precipitation, transpiration, runoff | Solvent for reactions, nutrient transport, temperature regulation |
Carbon | Atmosphere, living organisms, fossil fuels, soil, sediments | Photosynthesis, respiration, decomposition, combustion | Component of all organic molecules |
Nitrogen | Atmosphere (N2), soil, aquatic sediments, biomass | Nitrogen fixation, ammonification, nitrification, denitrification | Proteins, nucleic acids |
Phosphorus | Sedimentary rocks, soil, dissolved in water | Weathering, uptake, decomposition, sedimentation | ATP, nucleic acids, phospholipids |
Review and Application
Biodiversity and Nutrient Cycling: Greater biodiversity often leads to more efficient nutrient cycling and ecosystem stability.
Human Impact: Activities such as burning fossil fuels, fertilizer use, and deforestation can disrupt these cycles, leading to environmental issues like climate change and eutrophication.
