 Back
BackProtein Structure and Function: Chapter 3 Study Notes
Study Guide - Smart Notes

Protein Structure and Function
Introduction to Proteins
Proteins are the most abundant and versatile macromolecules in life, playing essential roles in cellular structure, function, and regulation. Their diversity arises from the unique sequence and chemical properties of their constituent amino acids.
Amino acids are the building blocks of proteins.
Proteins are involved in catalysis, structure, movement, signaling, transport, and defense.
Protein structure determines its function and role in living cells.

Amino Acids and Their Polymerization
Amino acids are organic molecules composed of a central carbon atom (α-carbon) bonded to an amino group, a carboxyl group, a hydrogen atom, and a variable R group (side chain). The combination of amino and carboxyl functional groups is key to their behavior in biological systems.
Structure: Central carbon, amino group (–NH2), carboxyl group (–COOH), hydrogen atom, and R group.
R group: Determines the identity and properties of each amino acid.
Ionization: In water, amino and carboxyl groups ionize, which helps amino acids stay in solution and affects their reactivity.
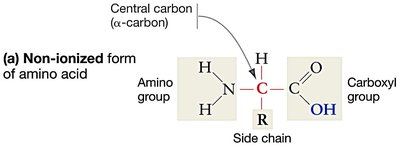
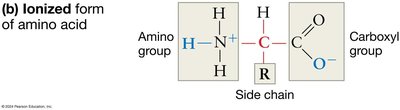
The Nature of Side Chains (R Groups)
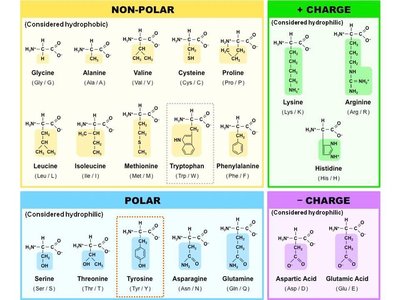
The R group, or side chain, is the part of the amino acid that makes each of the 20 amino acids unique. The properties of amino acids vary because R groups vary in size, polarity, and charge.
Some side chains contain functional groups and can participate in chemical reactions.
Others consist solely of carbon and hydrogen atoms and are nonreactive.
Polarity and Charge of R Groups Affect Solubility
The nature of the R group affects the solubility of amino acids:
Hydrophilic: Polar and charged R groups interact with water.
Hydrophobic: Nonpolar R groups do not form hydrogen bonds with water.
Classification of Amino Acids by R Group
Charged: Can be acidic (negative charge) or basic (positive charge).
Uncharged polar: Contain highly electronegative atoms like oxygen.
Nonpolar: Lack charge and electronegative atoms; hydrophobic.
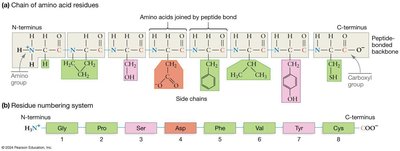
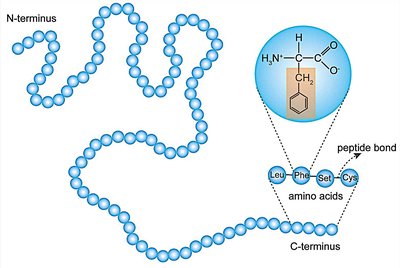
How Do Amino Acids Link to Form Proteins?
Amino acids polymerize via a condensation (dehydration) reaction, forming a peptide bond between the carboxyl group of one amino acid and the amino group of another. This process creates a polypeptide chain.
Peptide bond: C–N covalent bond formed during polymerization.
Directionality: N-terminus (free amino group) to C-terminus (free carboxyl group).
Flexibility: Peptide bond itself cannot rotate, but single bonds adjacent to it can.


Levels of Protein Structure
Primary Structure
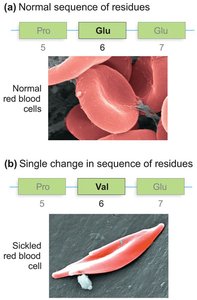
The primary structure of a protein is its unique sequence of amino acids. This sequence is fundamental to all higher levels of protein structure.
There are 20 types of amino acids, allowing for immense diversity.
Single amino acid changes can radically alter protein function (e.g., sickle cell anemia).
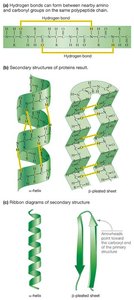
Secondary Structure
Secondary structure is formed by hydrogen bonds between the carbonyl group of one amino acid and the amino group of another. The two main types are:
α-helix
β-pleated sheet
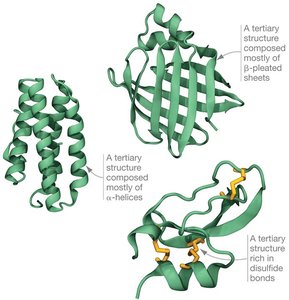
Tertiary Structure
Tertiary structure results from interactions between R groups and between R groups and the peptide backbone, causing the polypeptide to fold into a three-dimensional shape.
Types of interactions: hydrogen bonding, hydrophobic interactions, van der Waals interactions, covalent bonding (disulfide bonds), and ionic bonding.

Quaternary Structure
Quaternary structure arises when two or more polypeptide subunits bond to form a single functional protein. These can be homodimers (identical subunits) or heterodimers (different subunits).
Macromolecular machines: Complexes of multiple proteins that carry out specific functions (e.g., ribosome).
Protein structure is hierarchical: Primary structure determines secondary, which folds into tertiary, and quaternary is based on interactions between tertiary structures.
Protein Folding and Function
Normal protein folding is crucial for function and is often spontaneous, resulting in a more stable, lower-energy state. Denatured proteins lose their function.
Molecular chaperones: Proteins that facilitate proper folding and prevent aggregation (e.g., heat shock protein 90).
Protein shape is flexible and dynamic, regulated by interactions and binding events.
Misfolding can be infectious, as seen in prions (e.g., mad cow disease).
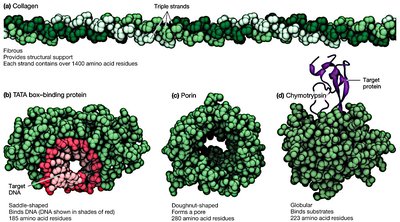
Diversity of Protein Functions
Proteins perform a wide range of functions in cells:
Catalysis: Enzymes speed up chemical reactions.
Structure: Shape cells and body structures.
Movement: Motor proteins move cells or molecules.
Signaling: Convey signals between cells.
Transport: Allow molecules to enter/exit cells or carry them throughout the body.
Defense: Antibodies attack pathogens.
Enzymes as Catalysts
Enzymes are proteins that function as catalysts, holding substrates in precise orientation to make reactions more likely. The active site is the location on an enzyme where substrates bind and react.
Substrates: Reactants in catalyzed reactions.
Active site: Region where substrate binding and reaction occur.
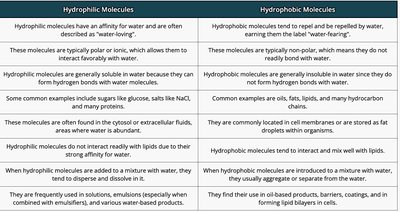
Summary Table: Hydrophilic vs. Hydrophobic Molecules
The following table compares hydrophilic and hydrophobic molecules, which is relevant to understanding amino acid side chain properties:
Hydrophilic Molecules | Hydrophobic Molecules |
|---|---|
Affinity for water; often described as "water-loving" | Repelled by water; "water-fearing" |
Typically polar or ionic; interact favorably with water | Typically non-polar; do not readily bond with water |
Generally soluble in water | Generally insoluble in water |
Examples: glucose, salts, many proteins | Examples: oils, fats, many hydrocarbon chains |
Found in cytosol or extracellular fluids | Found in cell membranes or as fat droplets |
Do not interact readily with lipids | Interact and mix well with lipids |
Added to water: tend to disperse and dissolve | Added to water: usually aggregate or separate |
Used in solutions, emulsions, water-based products | Used in oil-based products, forming lipid bilayers |

Conclusion
Understanding protein structure—from amino acid sequence to complex quaternary assemblies—is fundamental to biology. Protein folding, function, and diversity are central to cellular life and underpin many biological processes.
