 Back
BackProtein Structure and Function: Study Notes for Biology Students
Study Guide - Smart Notes

Protein Structure and Function
Amino Acids and Their Polymerization
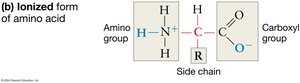
Amino acids are the fundamental building blocks of proteins, each composed of a central carbon atom bonded to four distinct groups: a hydrogen atom, an amino group, a carboxyl group, and a variable R group (side chain). The combination of amino and carboxyl functional groups is crucial for the chemical behavior of amino acids, especially in aqueous environments where these groups ionize, affecting solubility and reactivity.
Core Structure: All amino acids share a common backbone structure, with the R group providing unique properties to each amino acid.
Ionization: In water, the amino group becomes positively charged, and the carboxyl group becomes negatively charged, which helps amino acids stay dissolved and influences their chemical reactivity.
R Group (Side Chain): The R group determines the identity and properties of each amino acid, including its ability to participate in chemical reactions.
The Nature of Side Chains
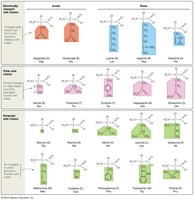
The R group, or side chain, is the variable part of an amino acid that distinguishes the 20 major amino acids found in organisms. Side chains can contain functional groups that participate in chemical reactions, or they may consist solely of carbon and hydrogen atoms.
Classification: Amino acid side chains are classified as charged, uncharged polar, or nonpolar.
Solubility: Polar and charged R groups are hydrophilic and interact with water, while nonpolar R groups are hydrophobic and avoid water.
Identification: The presence of negative or positive charge, or an oxygen atom in the side chain, helps determine the amino acid's classification.
Polymerization of Amino Acids
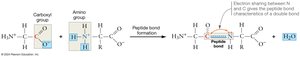
Amino acids link together to form proteins through a process called polymerization, which involves the formation of peptide bonds between the carboxyl group of one amino acid and the amino group of another. This condensation reaction releases a molecule of water.
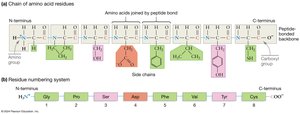
Peptide Bond: A covalent bond formed during polymerization, resulting in a peptide backbone with side chains extending outward.
Directionality: The chain has an N-terminus (free amino group) and a C-terminus (free carboxyl group), conventionally written with the N-terminus on the left.
Flexibility: The peptide bond itself cannot rotate, but the single bonds adjacent to it can, allowing flexibility in the polypeptide chain.
Equation:
Levels of Protein Structure
Proteins exhibit a hierarchical structure, with each level contributing to the molecule's overall shape and function. The four levels are primary, secondary, tertiary, and quaternary structure.
Primary Structure: The unique sequence of amino acids in a polypeptide. Even a single amino acid change can drastically affect protein function (e.g., hemoglobin mutations).
Secondary Structure: Formed by hydrogen bonds between backbone atoms, resulting in alpha-helices and beta-pleated sheets.
Tertiary Structure: The three-dimensional shape formed by interactions among R groups, including hydrogen bonding, hydrophobic interactions, van der Waals forces, covalent (disulfide) bonds, and ionic bonds.
Quaternary Structure: The arrangement of multiple polypeptide subunits into a single functional protein. Examples include dimers (two subunits), homodimers (identical subunits), and heterodimers (different subunits).
Additional info: Macromolecular machines, such as ribosomes, consist of multiple proteins and nucleic acids working together to perform complex cellular functions.
Protein Folding and Function
Proper protein folding is essential for function and is often spontaneous due to chemical interactions. Folded proteins are more stable and have lower potential energy than unfolded (denatured) proteins. Molecular chaperones assist in folding by preventing inappropriate interactions.
Chaperones: Proteins like heat shock protein Hsp90 help newly synthesized polypeptides fold correctly.
Flexibility: Many proteins are dynamic and can adopt multiple shapes until they bind specific molecules.
Misfolding: Misfolded proteins can be infectious, as seen in prions responsible for diseases like mad cow disease.
Additional info: Protein folding is regulated and can be influenced by cellular conditions and interactions with other molecules.
Diversity of Protein Functions
Proteins are essential for nearly all cellular tasks, with functions as diverse as their structures. Their roles include catalysis, structure, movement, signaling, transport, and defense.
Catalysis: Enzymes speed up chemical reactions by binding substrates at their active sites, orienting them for reaction.
Structure: Proteins shape cells and form body structures.
Movement: Motor proteins move cells or molecules within cells.
Signaling: Proteins convey signals between cells.
Transport: Proteins allow molecules to enter and exit cells or carry them throughout the body.
Defense: Antibodies attack pathogens.
Example: Enzymes are highly specific catalysts, holding substrates in precise orientations to facilitate reactions.
Equation:
