 Back
BackProtein Structure and Function: Study Notes
Study Guide - Smart Notes

Chapter 3: Protein Structure and Function
Introduction to Proteins
Proteins are the most abundant and versatile macromolecules in living organisms. They are essential for nearly every biological process, from catalyzing metabolic reactions to providing structural support. Understanding protein structure is fundamental to understanding their diverse functions in cells.
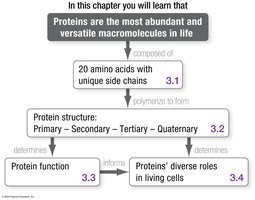
3.1 Amino Acids and Their Polymerization
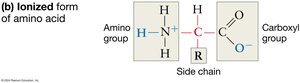
Amino acids are the building blocks of proteins. Each amino acid consists of a central carbon atom (the alpha carbon) bonded to a hydrogen atom, an amino group, a carboxyl group, and a variable side chain known as the R group. The properties of amino acids are determined by their R groups, which can vary in polarity, charge, and reactivity.
General Structure: All amino acids share a core structure but differ in their side chains.
Ionization in Water: In aqueous solutions, amino and carboxyl groups ionize, which helps amino acids stay in solution and affects their chemical reactivity.
Types of R Groups: The 20 major amino acids found in organisms have side chains that can be classified as charged (acidic or basic), uncharged polar, or nonpolar. This classification affects their solubility and interactions.
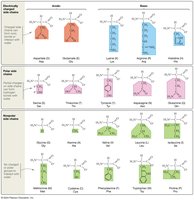
Solubility: Polar and charged R groups are hydrophilic and interact with water, while nonpolar R groups are hydrophobic and do not form hydrogen bonds with water.
Determining Amino Acid Type: To classify an amino acid, check for negative charge (acidic), positive charge (basic), or the presence of an oxygen atom (uncharged polar). If none are present, the amino acid is nonpolar.
Polymerization of Amino Acids
Amino acids link together via peptide bonds, which form between the carboxyl group of one amino acid and the amino group of another through a condensation reaction. This process creates polypeptide chains, which fold into functional proteins.
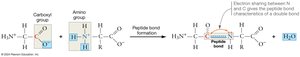
Peptide-Bonded Backbone: The backbone has directionality (N-terminus to C-terminus), R-group orientation (side chains extend out), and flexibility (rotation around single bonds).
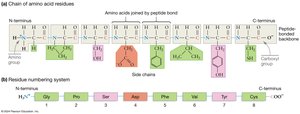
Oligopeptides and Polypeptides: Chains with fewer than 50 amino acids are called oligopeptides or peptides; longer chains are polypeptides. A protein is a complete, functional form of a polypeptide.
3.2 What Do Proteins Look Like?
The structure of a protein determines its function. Proteins exhibit four levels of structural organization:
Primary Structure: The unique sequence of amino acids in a polypeptide. Even a single amino acid change can drastically affect protein function (e.g., sickle cell hemoglobin).
Secondary Structure: Local folding patterns stabilized by hydrogen bonds, such as alpha-helices and beta-pleated sheets.
Tertiary Structure: The overall three-dimensional shape formed by interactions among R groups, including hydrogen bonds, hydrophobic interactions, van der Waals forces, covalent disulfide bonds, and ionic bonds.
Quaternary Structure: The assembly of multiple polypeptide subunits into a functional protein complex (e.g., hemoglobin, ribosome).
Level | Description | Stabilizing Bonds/Interactions |
|---|---|---|
Primary | Sequence of amino acids | Peptide bonds |
Secondary | Alpha-helix, beta-sheet | Hydrogen bonds |
Tertiary | 3D folding of polypeptide | Hydrogen, hydrophobic, van der Waals, covalent, ionic |
Quaternary | Multiple polypeptides | Same as tertiary, between subunits |
*Additional info: Table inferred from "Table 3.1 Protein Structure" summary in the text.*
3.3 Folding and Function
Proper protein folding is essential for function. Folding is often spontaneous and results in a more stable, lower-energy structure. Misfolded proteins can lose function or become harmful (e.g., prions in mad cow disease).
Molecular Chaperones: Specialized proteins that assist in the proper folding of other proteins and prevent aggregation.
Protein Flexibility: Many proteins are dynamic and can adopt multiple conformations until they bind to specific molecules or are activated.
Infectious Misfolding: Prions are misfolded proteins that can induce normal proteins to adopt the same abnormal structure, leading to disease.
3.4 Protein Functions Are as Diverse as Protein Structures
Proteins perform a vast array of functions in cells, including:
Catalysis: Enzymes accelerate chemical reactions by binding substrates at their active sites.
Structure: Provide support and shape to cells and tissues.
Movement: Motor proteins move cells or molecules within cells.
Signaling: Transmit signals between cells.
Transport: Move molecules across membranes or throughout the body.
Defense: Antibodies recognize and neutralize pathogens.
Enzyme Function
Enzymes are highly specific catalysts. They bind substrates in a precise orientation at the active site, lowering the activation energy required for reactions to proceed.
Proteins and the Origin of Life
Experiments suggest that amino acids could have formed and polymerized under prebiotic Earth conditions, but it remains unclear if proteins were the first self-replicating molecules. Nucleic acids are now considered more likely candidates for the origin of life due to their information-carrying capacity.
