 Back
BackProtein Structure and Function: Study Notes
Study Guide - Smart Notes

Protein Structure and Function
3.1 Amino Acids and Their Polymerization
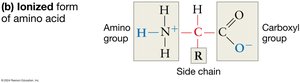
Amino acids are the building blocks of proteins, each composed of a central carbon atom bonded to a hydrogen atom, an amino group, a carboxyl group, and a variable side chain (R group). The combination of amino and carboxyl functional groups is key to the chemical behavior of amino acids, especially in aqueous environments where these groups ionize, affecting solubility and reactivity.
Structure of Amino Acids: All amino acids share a core structure, differing only in their R group.
Ionization in Water: The amino group gains a proton (becomes NH3+), and the carboxyl group loses a proton (becomes COO-), which helps amino acids stay in solution and participate in chemical reactions.
R Groups (Side Chains): The R group determines the unique properties of each amino acid, including their chemical reactivity and solubility.
Classification of Amino Acids: Based on the nature of their R groups, amino acids are classified as charged (acidic or basic), uncharged polar, or nonpolar. This classification affects their solubility and interactions in proteins.
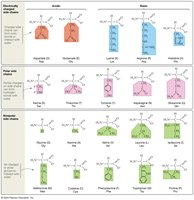
Determining Amino Acid Type: To classify an amino acid, ask: Does the side chain have a negative charge (acidic)? A positive charge (basic)? An oxygen atom (uncharged polar)? If none, it is nonpolar.
How Do Amino Acids Link to Form Proteins?
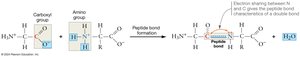
Amino acids polymerize through condensation reactions, forming peptide bonds between the carboxyl group of one amino acid and the amino group of another. This process creates a polypeptide chain, the backbone of protein structure.
Peptide Bond: A covalent bond formed by the removal of a water molecule during polymerization.
Peptide-Bonded Backbone: Has directionality (N-terminus to C-terminus), R-group orientation (side chains extend out), and flexibility (rotation around single bonds adjacent to the peptide bond).
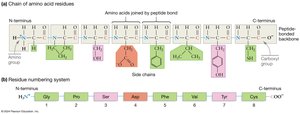
Oligopeptides and Polypeptides: Chains with fewer than 50 amino acids are called oligopeptides or peptides; longer chains are polypeptides. A protein is a complete, functional polypeptide.
3.2 What Do Proteins Look Like?
Levels of Protein Structure
Protein function is determined by its structure, which is organized hierarchically into four levels: primary, secondary, tertiary, and quaternary.
Primary Structure: The unique sequence of amino acids in a protein. Even a single amino acid change can drastically alter protein function (e.g., sickle cell hemoglobin).
Secondary Structure: Formed by hydrogen bonds between backbone atoms, resulting in alpha-helices and beta-pleated sheets.
Tertiary Structure: The overall 3D shape of a polypeptide, determined by interactions among R groups, including hydrogen bonds, hydrophobic interactions, van der Waals forces, covalent (disulfide) bonds, and ionic bonds.
Quaternary Structure: The arrangement of multiple polypeptide subunits in a functional protein complex. Subunits may be identical (homodimers) or different (heterodimers). Some proteins form large macromolecular machines (e.g., ribosomes).
3.3 Folding and Function
Protein Folding
Proper protein folding is essential for function and is often spontaneous due to chemical interactions. Folded proteins are more stable (lower potential energy) than unfolded (denatured) proteins, which are typically nonfunctional.
Molecular Chaperones: Specialized proteins that assist in the correct folding of other proteins and prevent inappropriate interactions (e.g., heat shock proteins like Hsp90).
Protein Flexibility: Many proteins are dynamic and can adopt multiple conformations until stabilized by binding to other molecules.
Prions: Misfolded proteins that can induce normal proteins to adopt an abnormal, disease-causing conformation (e.g., prion protein in mad cow disease).
3.4 Protein Functions Are as Diverse as Protein Structures
Major Functions of Proteins
Proteins perform a vast array of functions in cells, including:
Catalysis: Enzymes accelerate chemical reactions by binding substrates at their active sites and orienting them for reaction.
Structure: Provide support and shape to cells and tissues.
Movement: Motor proteins move cells or molecules within cells.
Signaling: Transmit signals between cells.
Transport: Move molecules across membranes or throughout the body.
Defense: Antibodies recognize and neutralize pathogens.
Why Are Enzymes Good Catalysts?
Substrate Binding: Enzymes have specific active sites where substrates bind, facilitating the chemical reaction by bringing reactants into the correct orientation.
Additional info: The notes above summarize the foundational concepts of protein structure and function, including the chemical properties of amino acids, the hierarchy of protein structure, the importance of folding, and the diverse roles proteins play in biological systems. Understanding these principles is essential for further study in biochemistry, cell biology, and molecular biology.
