 Back
BackProtein Structure and Function: Study Notes
Study Guide - Smart Notes

Protein Structure and Function
3.1 Amino Acids and Their Polymerization
Amino acids are the building blocks of proteins, each sharing a common core structure but differing in their side chains (R-groups). The way amino acids link and interact determines the structure and function of proteins.
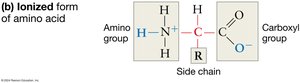
Structure of Amino Acids: Each amino acid consists of a central carbon atom (the alpha carbon) bonded to a hydrogen atom, an amino group (–NH2), a carboxyl group (–COOH), and a variable R-group (side chain).
Ionization in Water: In aqueous solutions, the amino group tends to gain a proton (becoming –NH3+), and the carboxyl group loses a proton (becoming –COO–). These charges help amino acids stay in solution and influence their chemical reactivity.
R-Groups (Side Chains): The R-group is the variable part of an amino acid and determines its unique properties. Some R-groups contain functional groups that can participate in chemical reactions, while others are composed only of carbon and hydrogen.
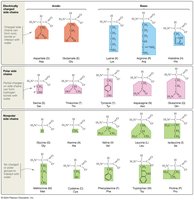
Classification by R-Group Properties: Amino acids are classified based on the polarity and charge of their R-groups:
Charged: Acidic (negative charge) or basic (positive charge).
Uncharged Polar: Contain electronegative atoms like oxygen, making them hydrophilic.
Nonpolar: Lack polar groups, making them hydrophobic.
Solubility: Polar and charged R-groups interact with water (hydrophilic), while nonpolar R-groups do not (hydrophobic).
How Do Amino Acids Link to Form Proteins?
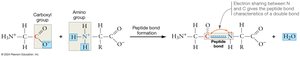
Amino acids are joined by peptide bonds, forming polypeptide chains. This process is a condensation reaction, where a molecule of water is released as the carboxyl group of one amino acid bonds to the amino group of another.
Peptide Bond: A covalent bond formed between the carboxyl carbon of one amino acid and the amino nitrogen of another.
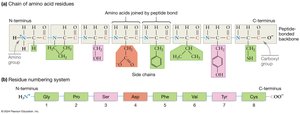
Directionality: Polypeptides have an N-terminus (free amino group) and a C-terminus (free carboxyl group). By convention, sequences are written from N-terminus to C-terminus.
Backbone Flexibility: The peptide bond itself is rigid, but the single bonds on either side allow rotation, giving the polypeptide chain flexibility.
Oligopeptides and Polypeptides: Chains with fewer than 50 amino acids are called oligopeptides or peptides; longer chains are polypeptides. A protein is a complete, functional polypeptide.
3.2 What Do Proteins Look Like?
Levels of Protein Structure
Protein function is determined by its structure, which is organized into four hierarchical levels:
Primary Structure: The unique sequence of amino acids in a protein. Even a single amino acid change can drastically affect protein function (e.g., sickle cell hemoglobin).
Secondary Structure: Regular folding patterns stabilized by hydrogen bonds between backbone atoms. The main types are the alpha-helix and beta-pleated sheet.
Tertiary Structure: The overall three-dimensional shape of a polypeptide, resulting from interactions among R-groups, including hydrogen bonds, hydrophobic interactions, van der Waals forces, covalent disulfide bonds, and ionic bonds.
Quaternary Structure: The arrangement of multiple polypeptide subunits in a protein. Subunits can be identical (homodimers) or different (heterodimers). Some proteins function as part of large macromolecular machines (e.g., ribosomes).
3.3 Folding and Function
Protein Folding and Stability
Proper folding is essential for protein function. Folding is often spontaneous and driven by chemical interactions that stabilize the structure. Misfolded proteins can lose function and may cause disease.
Molecular Chaperones: Specialized proteins that assist in the proper folding of other proteins and prevent inappropriate interactions (e.g., heat shock proteins like Hsp90).
Protein Flexibility: Many proteins are dynamic and can adopt multiple conformations until they bind specific molecules or are activated.
Prions: Infectious, misfolded proteins that can induce normal proteins to adopt the abnormal conformation, leading to diseases such as mad cow disease.
3.4 Protein Functions Are as Diverse as Protein Structures
Major Functions of Proteins
Proteins perform a vast array of functions in cells, including:
Catalysis: Enzymes accelerate chemical reactions by binding substrates at their active sites and lowering activation energy.
Structure: Provide support and shape to cells and tissues (e.g., collagen, keratin).
Movement: Motor proteins move cells or molecules within cells (e.g., actin, myosin).
Signaling: Transmit signals between cells (e.g., hormones, receptors).
Transport: Carry molecules across membranes or throughout the body (e.g., hemoglobin, membrane transporters).
Defense: Antibodies recognize and neutralize pathogens.
Why Are Enzymes Good Catalysts?
Substrate Specificity: Enzymes bind specific reactants (substrates) at their active sites, positioning them to react efficiently.
Active Site: The region of the enzyme where substrates bind and the reaction occurs.
