 Back
BackProteins and Amino Acids: Structure, Classification, and Synthesis
Study Guide - Smart Notes

Proteins and Amino Acids
Introduction to Proteins
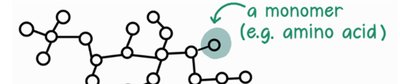
Proteins are essential biological macromolecules composed of amino acids. They perform a vast array of functions within living organisms, including catalyzing metabolic reactions, providing structural support, and regulating cellular processes. Proteins are polymers, meaning they are made up of repeating subunits called monomers—in this case, amino acids.
Structure of Amino Acids
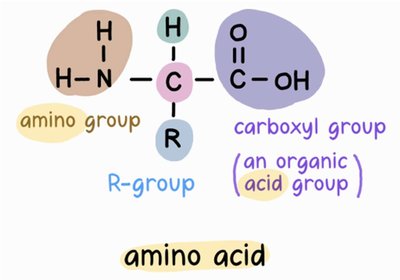
All amino acids share a common basic structure, consisting of a central carbon atom (the alpha carbon) bonded to four different groups: an amino group (-NH2), a carboxyl group (-COOH), a hydrogen atom, and a variable side chain known as the R-group. The R-group determines the unique characteristics of each amino acid.
Examples of Amino Acids and Their R-Groups
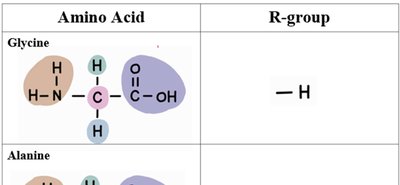
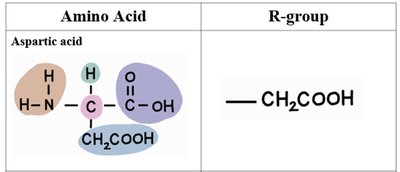
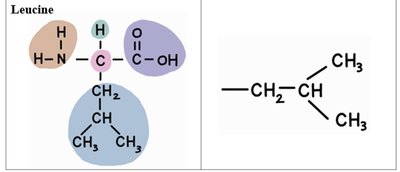
The 20 standard amino acids differ only in their R-groups. Below is a table summarizing the structure and R-groups of several common amino acids:
Amino Acid | R-group |
|---|---|
Glycine | –H |
Alanine | –CH3 |
Serine | –CH2OH |
Aspartic Acid | –CH2COOH |
Leucine | –CH2CH(CH3)2 |
Bonding in Amino Acids
Each atom in an amino acid forms a specific number of bonds: carbon forms four, oxygen forms two, nitrogen forms three, and hydrogen forms one. The carboxyl group (-COOH) and amino group (-NH2) are key functional groups in amino acids.


Classification of Amino Acids
Amino acids can be classified based on the properties of their R-groups:
Nonpolar (hydrophobic): R-group contains only hydrogen or hydrogen and carbon (e.g., Glycine, Alanine, Leucine).
Polar (hydrophilic): R-group contains atoms like oxygen or nitrogen (e.g., Serine).
Acidic: R-group contains a carboxyl group (e.g., Aspartic acid, Glutamic acid).
Basic: R-group contains an amino group (e.g., Lysine).
Proteins as Polymers of Amino Acids
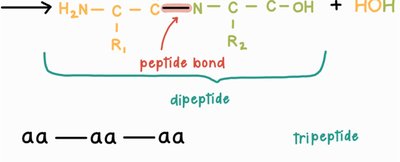
Proteins are polymers formed by linking amino acids together through peptide bonds. The individual amino acids are the monomers, and the resulting chain is called a polypeptide. The sequence and number of amino acids determine the protein's structure and function.
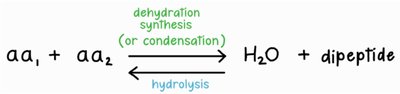
Peptide Bond Formation: Dehydration Synthesis
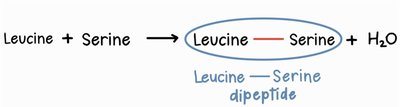
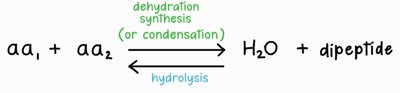
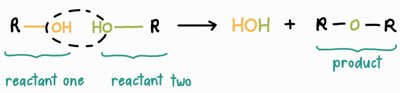
Amino acids are joined together by peptide bonds in a process called dehydration synthesis (or condensation reaction). In this reaction, the carboxyl group of one amino acid reacts with the amino group of another, releasing a molecule of water and forming a covalent bond.
Peptide bond: The covalent bond formed between the carboxyl carbon of one amino acid and the amino nitrogen of another.
Dipeptide: A molecule consisting of two amino acids joined by a peptide bond.
Polypeptide: A chain of many amino acids linked by peptide bonds.
Hydrolysis: Breaking Peptide Bonds
The reverse of dehydration synthesis is hydrolysis, in which a water molecule is added to break a peptide bond, releasing individual amino acids.
Naming Peptides
The number of amino acids in a peptide determines its name:
Dipeptide: Two amino acids
Tripeptide: Three amino acids
Tetrapeptide: Four amino acids
Polypeptide: Many amino acids
The order of amino acids in a peptide is crucial, as different sequences yield different molecules (e.g., tyrosine-valine is different from valine-tyrosine).
From DNA to Protein: The Central Dogma
Proteins are synthesized according to genetic instructions encoded in DNA. The process involves two main steps:
Transcription: DNA is transcribed into messenger RNA (mRNA).
Translation: mRNA is translated into a specific sequence of amino acids, forming a protein.
Summary Table: Amino Acid Classification
Amino Acid | R-group | Classification |
|---|---|---|
Glycine | H | Nonpolar (hydrophobic) |
Alanine | CH3 | Nonpolar (hydrophobic) |
Leucine | CH2CH(CH3)2 | Nonpolar (hydrophobic) |
Serine | CH2OH | Polar (hydrophilic) |
Aspartic Acid | CH2COOH | Acidic |
Glutamic Acid | CH2CH2COOH | Acidic |
Lysine | CH2CH2CH2CH2NH2 | Basic |
Key Equations
Dehydration Synthesis (Peptide Bond Formation):
Hydrolysis (Peptide Bond Breakdown):
Review Questions
Write a reaction to illustrate how a tyrosine-lysine dipeptide forms.
Out of the amino acids mentioned, indicate which are nonpolar, acidic, basic, or just polar.
How many electrons are being shared in the amino acid phenylalanine?
Draw a tripeptide.
