 Back
BackLEC 4: Proteins and Nucleic Acids: Structure and Function
Study Guide - Smart Notes

Proteins: Structure and Function
Overview of Proteins
Proteins are essential macromolecules involved in nearly every biological process. Their diverse functions arise from their unique three-dimensional structures, which are determined by the sequence and properties of their amino acid building blocks.
Functions: Enzymes, antibodies, storage proteins, hormones, transporters, structural proteins, and molecular machines.
Structure: Each protein has a unique 3D shape critical for its function.
Amino Acids: Building Blocks of Proteins
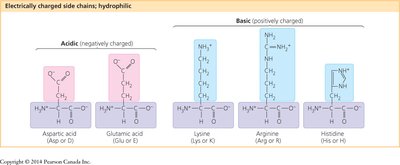
Amino acids are organic molecules with a central carbon atom (α-carbon) bonded to an amino group, a carboxyl group, a hydrogen atom, and a variable side chain (R group). Proteins are polymers of amino acids, linked by peptide bonds.
General Structure: All amino acids share a common backbone but differ in their side chains (R groups).
Enantiomers: Only L-enantiomers are used in proteins.
Classification: Amino acids are grouped by the properties of their side chains: nonpolar, polar, and electrically charged (acidic or basic).
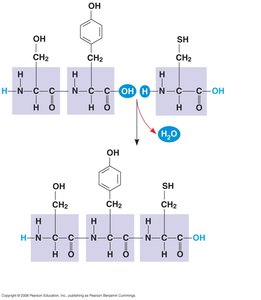
Peptide Bond Formation
Amino acids are joined by peptide bonds through a dehydration reaction, forming polypeptides. Each polypeptide has an amino (N-) terminus and a carboxyl (C-) terminus.
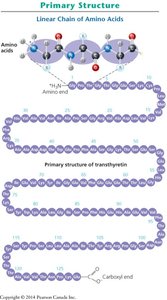
Levels of Protein Structure
Proteins have four levels of structural organization, each contributing to the final shape and function of the molecule.
Primary Structure: The linear sequence of amino acids in a polypeptide, determined by genetic information.
Secondary Structure: Local folding into α-helices and β-pleated sheets, stabilized by hydrogen bonds between backbone atoms.
Tertiary Structure: The overall 3D shape of a polypeptide, resulting from interactions among R groups (hydrophobic interactions, van der Waals forces, ionic bonds, hydrogen bonds, and disulfide bridges).
Quaternary Structure: The association of two or more polypeptide subunits to form a functional protein complex (e.g., hemoglobin).
Protein Denaturation and Folding
Protein structure can be disrupted by changes in pH, salt concentration, temperature, or other environmental factors, leading to denaturation and loss of function. Proper folding is essential for activity, and misfolded proteins are associated with diseases such as Alzheimer's and Parkinson's. Chaperonins are specialized proteins that assist in the correct folding of other proteins.
Denaturation: The process by which a protein loses its native structure and becomes biologically inactive.
Chaperonins: Proteins that help other proteins fold correctly.


Nucleic Acids: DNA and RNA
Overview of Nucleic Acids
Nucleic acids are polymers that store, transmit, and express genetic information. The two main types are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA).
DNA: Stores hereditary information and transmits it to cell descendants.
RNA: Functions in gene expression, including carrying instructions from DNA to ribosomes (mRNA).
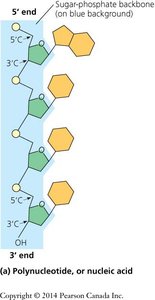
Nucleotide Structure
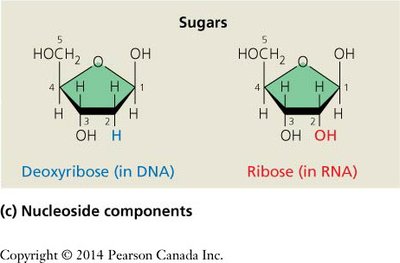
Nucleotides, the monomers of nucleic acids, consist of three components: a nitrogenous base, a five-carbon sugar, and a phosphate group. The sugar is deoxyribose in DNA and ribose in RNA.
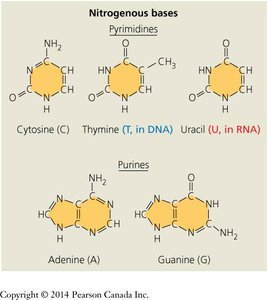
Nitrogenous Bases: Two types: pyrimidines (cytosine, thymine, uracil) and purines (adenine, guanine).
Base Pairing: In DNA, adenine pairs with thymine, and guanine pairs with cytosine. In RNA, uracil replaces thymine.
Formation of Nucleic Acid Polymers
Nucleotides are joined by phosphodiester bonds between the 3′ hydroxyl of one sugar and the 5′ phosphate of the next, forming a sugar-phosphate backbone. Polynucleotide chains have directionality (5′ to 3′).
Structure of DNA and RNA
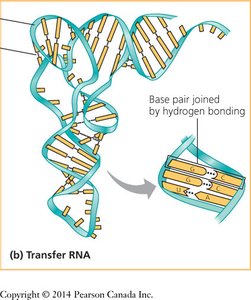
DNA is typically a double helix with two antiparallel strands held together by complementary base pairing. RNA is usually single-stranded but can form complex structures through intramolecular base pairing.
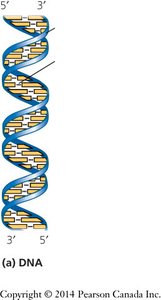
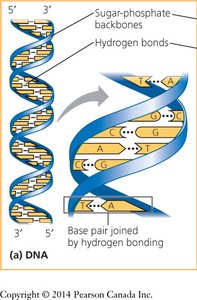
Antiparallel Strands: The two DNA strands run in opposite 5′→3′ directions.
Complementary Base Pairing: Ensures accurate replication and transcription.
RNA Structure: Single-stranded, but can form double-stranded regions by base pairing with itself or other nucleic acids.
Nucleosides and Nucleotides: Nomenclature
Nucleosides consist of a nitrogenous base and a sugar, while nucleotides also include one or more phosphate groups. Nucleotides are named based on their base, sugar, and number of phosphates (e.g., AMP, dGTP).
Summary Table: Key Differences Between DNA and RNA
Feature | DNA | RNA |
|---|---|---|
Sugar | Deoxyribose | Ribose |
Strands | Double-stranded (usually) | Single-stranded (usually) |
Pyrimidine Bases | Cytosine, Thymine | Cytosine, Uracil |
Purine Bases | Adenine, Guanine | Adenine, Guanine |
Main Function | Genetic information storage | Gene expression, protein synthesis |
Key Equations and Concepts
Peptide Bond Formation:
Phosphodiester Bond Formation:
Additional info: Nucleotides can exist as mono-, di-, or triphosphates (e.g., ATP, GTP), which are important in cellular energy transfer and signaling.
