 Back
BackStates of Matter and Intermolecular Forces: Changes of State and Properties
Study Guide - Smart Notes

States of Matter and Changes of State
Overview of States of Matter
The three primary states of matter—gas, liquid, and solid—differ in the arrangement and movement of their particles. These differences are governed by the balance between kinetic energy and intermolecular forces.
Gas: Particles are far apart, move freely, and have little interaction. Gases have neither definite shape nor volume.
Liquid: Particles are closer together than in gases, allowing for flow but not fixed positions. Liquids have definite volume but take the shape of their container.
Solid: Particles are closely packed in a fixed, orderly arrangement. Solids have definite shape and volume.
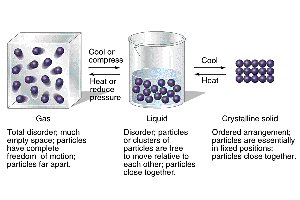
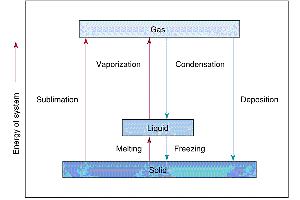
Phase transitions occur when energy is added or removed, causing matter to change from one state to another (e.g., melting, freezing, vaporization, condensation, sublimation, deposition).
Particle Motion and Kinetic Theory
The kinetic molecular theory explains the behavior of particles in different states. In gases, particles move rapidly and randomly; in liquids, they move more slowly and are closer together; in solids, they vibrate in fixed positions.
Kinetic energy increases with temperature, influencing the state of matter.
Intermolecular forces counteract kinetic energy, holding particles together.
Intermolecular Forces
Types of Intermolecular Forces
Intermolecular forces are attractions between molecules that determine physical properties such as boiling and melting points.
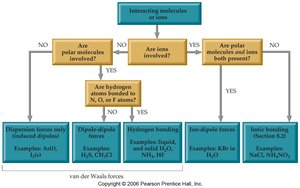
Dispersion (London) forces: Weak attractions present in all molecules, especially nonpolar ones.
Dipole-dipole interactions: Attractions between polar molecules.
Hydrogen bonding: A strong type of dipole-dipole interaction involving H bonded to N, O, or F.
Ion-dipole forces: Attractions between ions and polar molecules.
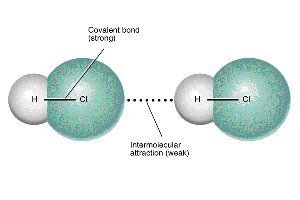
Example: Water exhibits hydrogen bonding, leading to its high boiling point compared to other group 6A hydrides.
Hierarchy and Identification of Forces
The type of intermolecular force present depends on the nature of the molecules involved (polar, nonpolar, ionic).
Dispersion forces are universal but often weak.
Dipole-dipole and hydrogen bonds are stronger and found in polar molecules.
Ion-dipole forces are strongest and occur in solutions of ions in polar solvents.
Physical Properties and Molecular Structure
Boiling and Melting Points
The strength of intermolecular forces directly affects boiling and melting points. Stronger forces require more energy to overcome, resulting in higher transition temperatures.
Boiling point: Temperature at which vapor pressure equals atmospheric pressure.
Melting point: Temperature at which a solid becomes a liquid.
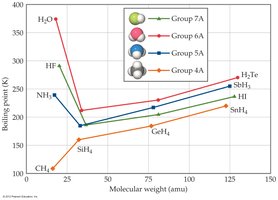
Example: Water (H2O) has a much higher boiling point than expected due to hydrogen bonding.
Structure and Physical Properties
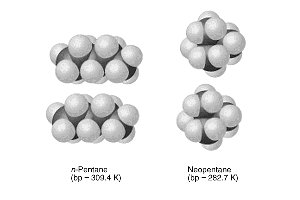
The shape and size of molecules influence their physical properties. For example, n-pentane and neopentane have different boiling points due to differences in molecular shape and surface area.
Phase Changes and Energy
Energy and Phase Transitions
Phase changes involve energy transfer without a change in temperature. The energy required to change state is called latent heat.
Fusion (melting): Solid to liquid
Vaporization: Liquid to gas
Sublimation: Solid to gas
Condensation: Gas to liquid
Freezing: Liquid to solid
Deposition: Gas to solid
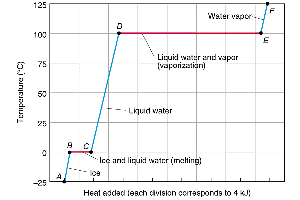
Example: The heating curve of water shows temperature plateaus during phase changes, indicating energy is used to break intermolecular forces rather than increase temperature.
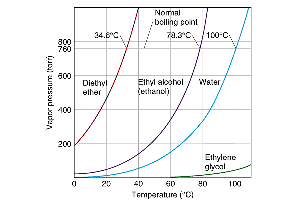
Vapor Pressure and Temperature
Vapor pressure is the pressure exerted by a vapor in equilibrium with its liquid at a given temperature. It increases with temperature as more molecules have enough kinetic energy to escape the liquid phase.
Normal boiling point: The temperature at which vapor pressure equals 1 atm.
Crystalline Solids: Types and Properties
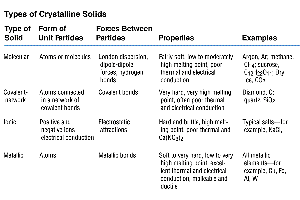
Classification of Crystalline Solids
Crystalline solids are classified based on the nature of their constituent particles and the forces holding them together.
Type of Solid | Form of Unit Particles | Forces Between Particles | Properties | Examples |
|---|---|---|---|---|
Molecular | Atoms or molecules | London dispersion, dipole-dipole, hydrogen bonds | Soft, low melting point, poor conductor | Argon, CO2, I2, H2O |
Covalent-network | Atoms connected by covalent bonds | Covalent bonds | Hard, high melting point, poor conductor | Diamond, SiO2 |
Ionic | Ions | Electrostatic attraction | Hard, brittle, high melting point, good conductor when molten | NaCl, KBr |
Metallic | Atoms | Metallic bonds | Variable hardness and melting point, good conductor | Fe, Cu, Na |

Example: Diamond and graphite are both forms of carbon but differ in structure and properties due to different bonding arrangements.
Summary Table: Intermolecular Forces and Properties
Force Type | Relative Strength | Occurs Between | Example |
|---|---|---|---|
Dispersion | Weakest | All molecules | Ar, I2 |
Dipole-dipole | Intermediate | Polar molecules | CH3Cl |
Hydrogen bond | Strong | H with N, O, or F | H2O, NH3 |
Ion-dipole | Strongest | Ions and polar molecules | Na+ in H2O |
Additional info: Understanding the physical properties of substances and their changes of state is essential for interpreting biological phenomena, such as the behavior of water in cells, the structure of biological membranes, and the function of macromolecules.
