 Back
BackStructure and Function of Skeletal Muscle: Organization, Contraction, and Repair
Study Guide - Smart Notes

Structure and Function of Skeletal Muscle
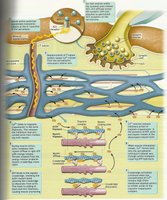
Overview of Skeletal Muscle Structure
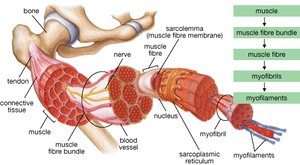
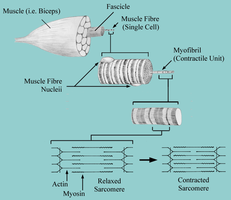
Skeletal muscle is a highly organized tissue responsible for voluntary movements in the body. Its structure is hierarchical, with several levels of organization that contribute to its function and adaptability.
Whole Muscle: Surrounded by the epimysium, a connective tissue sheath.
Muscle Fascicle: Bundles of muscle fibers, each wrapped in perimysium.
Muscle Fiber (Cell): The basic cellular unit, enveloped by the endomysium or sarcolemma.
Myofibrils: Cylindrical structures within each fiber, where hypertrophy (growth) occurs.
Myofilaments: The contractile proteins (actin and myosin) within myofibrils.
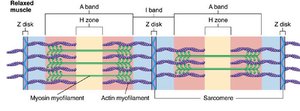
Sarcomere: The Basic Contractile Unit
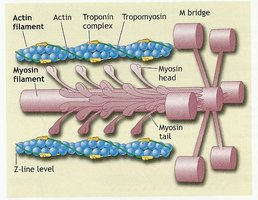
The sarcomere is the fundamental contractile unit of skeletal muscle, extending from one Z-line to the next. It contains organized arrays of proteins that enable contraction.
Myosin: Thick contractile filament.
Actin: Thin contractile filament.
Tropomyosin: Regulatory protein associated with actin.
Troponin: Regulatory protein complex that binds calcium and controls tropomyosin position.
Organization of the Sarcomere
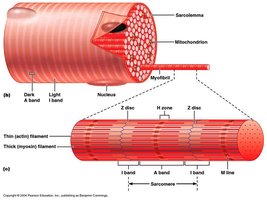
The sarcomere is divided into distinct regions based on the arrangement of thick and thin filaments:
M-line: Center of the sarcomere, where myosin filaments are anchored.
Z-line: Boundary of the sarcomere, where actin filaments are anchored.
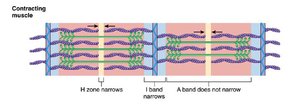
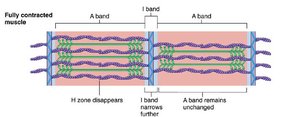
H-zone: Region with only myosin filaments; disappears during contraction.
A-band: Area of overlap between thick and thin filaments.
I-band: Region with only actin filaments; no overlap with myosin.
Muscle Fiber Ultrastructure
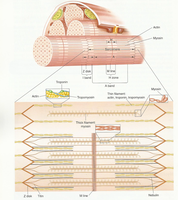
Muscle fibers contain specialized organelles and structures that facilitate contraction:
Sarcoplasmic Reticulum (SR): Stores and releases calcium ions.
Terminal Cisternae: Enlarged areas of the SR adjacent to T-tubules.
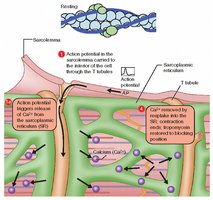
Transverse Tubules (T-tubules): Invaginations of the sarcolemma that transmit action potentials into the fiber.
Mitochondria: Provide ATP for contraction.
Mechanism of Muscle Contraction
Neuromuscular Junction and Excitation
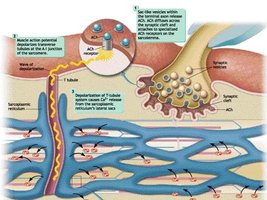
The neuromuscular junction is the synapse between a motor neuron and a muscle fiber. It is essential for initiating muscle contraction.
Motor Unit: A motor neuron and all the muscle fibers it innervates.
Motor End Plate: Region of the muscle fiber membrane that receives the neural signal.
Synaptic Cleft: Gap between the neuron and muscle fiber.
Neurotransmitter (Acetylcholine, Ach): Released from the neuron, binds to receptors on the sarcolemma, initiating an action potential in the muscle fiber.
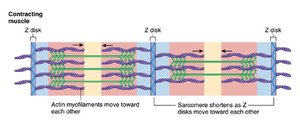
Sliding Filament Theory of Muscle Contraction
Muscle contraction occurs through the sliding of actin filaments over myosin filaments, shortening the sarcomere without changing the length of the filaments themselves.
Force Generation: Produced by the sliding process.
Sarcomere Shortening: Z-lines are pulled toward the center.
Filament Length: Thick and thin filaments remain constant in length.
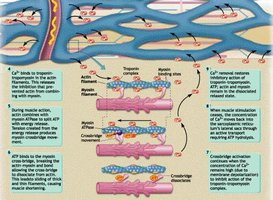
Excitation-Contraction Coupling
This process links the action potential in the muscle fiber to the initiation of contraction:
Nerve impulse triggers Ach release at the neuromuscular junction.
Ach binds to receptors, allowing Na+ and K+ influx, generating a new action potential in the sarcolemma.
Depolarization spreads via T-tubules, causing Ca2+ release from the sarcoplasmic reticulum.
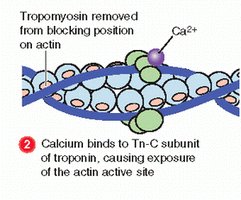
Ca2+ binds to troponin, shifting tropomyosin and exposing actin binding sites.
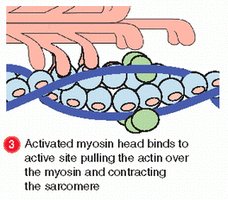
Role of Troponin, Tropomyosin, and Actin
Troponin and tropomyosin regulate muscle contraction by controlling the interaction between actin and myosin:
Troponin: Binds Ca2+, causing a conformational change.
Tropomyosin: Moves away from actin's myosin-binding sites when troponin binds Ca2+.
Actin: Exposed binding sites allow myosin to attach and generate force.
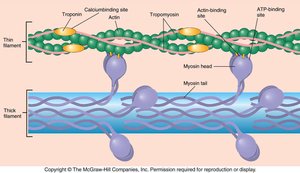
ATP and the Cross-Bridge Cycle
ATP is essential for muscle contraction and relaxation:
ATP binds to myosin, allowing it to detach from actin and re-cock for another cycle.
Energy from ATP hydrolysis is used to power the movement of the myosin head.
Skeletal Muscle Damage and Repair
Types and Mechanisms of Muscle Injury
Muscle injuries can occur due to contusions or contraction-induced damage, especially during eccentric (lengthening) contractions such as plyometrics, negative strength training, or downhill running.
Contraction-Induced Injury: Most common, results in focal sarcomere-level damage.
Immediate Effects: Up to 50% drop in force production post-injury.
Delayed Effects: Delayed onset muscle soreness (DOMS), increased plasma creatine kinase (CK), and lactate dehydrogenase (LDH).

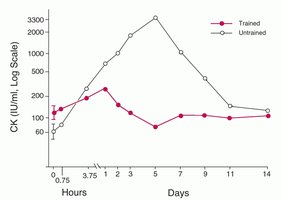
Biochemical Markers of Muscle Damage
Muscle damage is often assessed by measuring enzymes released into the blood:
Creatine Kinase (CK): Peaks 48-72 hours after eccentric exercise; higher in untrained individuals.
Lactate Dehydrogenase (LDH): Increases with hypoxia and contraction, especially with elevated Ca2+.
Role of Calcium and Antioxidants in Muscle Damage
High intracellular Ca2+ exacerbates muscle damage, while antioxidants can mitigate this effect if present chronically in the diet.
High Ca2+: Increases LDH release and muscle damage.
Antioxidants: Reduce Ca2+-induced damage and LDH release.
Mechanism of Contraction-Induced Injury
Muscle injury involves a sequence of events:
Activation and eccentric contraction.
Return to initial length, causing sarcomere heterogeneity and overstretching.
Structural damage leads to force deficit and inflammation.
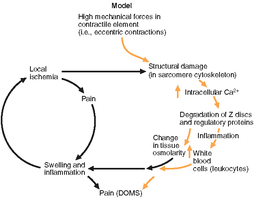
Muscle Repair: Degeneration and Regeneration
Following injury, muscle fibers undergo a process of degeneration and regeneration:
Degeneration: Damaged fibers are cleared by white blood cells (macrophages and leukocytes), recruited by fibroblast growth factor (FGF) and insulin-like growth factor (IGF-1).
Regeneration: Satellite cells (SC) are activated, proliferate, align, and fuse to form new myotubes, which mature into adult muscle fibers.

Role of Cytokines in Muscle Repair
Cytokines are signaling proteins that regulate the inflammatory response and facilitate muscle repair. Interleukin-6 (IL-6) is particularly important for stimulating satellite cell proliferation and muscle growth in response to exercise.
Application to Human Performance
Training status affects susceptibility to muscle injury:
Untrained Athletes: Experience more contraction-induced injury due to greater sarcomere heterogeneity.
Trained Athletes: Develop more homogeneous sarcomere populations, reducing injury risk.
Summary Table: Key Sarcomere Components
Component | Location | Function |
|---|---|---|
Myosin | Thick filament | Force generation, ATPase activity |
Actin | Thin filament | Binds myosin, slides during contraction |
Tropomyosin | Thin filament | Blocks myosin binding sites on actin |
Troponin | Thin filament | Binds Ca2+, moves tropomyosin |
Z-line | Boundary of sarcomere | Anchors actin filaments |
M-line | Center of sarcomere | Anchors myosin filaments |
Key Learning Points
Understand the hierarchical structure of skeletal muscle and the organization of the sarcomere.
Describe the steps of muscle contraction, including excitation-contraction coupling and the sliding filament theory.
Explain the mechanisms and biochemical markers of muscle damage.
Discuss the cellular and molecular processes involved in muscle repair and regeneration.
