 Back
BackStudy Guidance for BIOS 1083 Test 1B: Functional Groups and Isomerism
Study Guide - Smart Notes

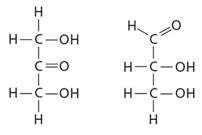
Q41. Choose the phrase that correctly describes the relationship between the two molecules pictured below.
Background
Topic: Isomerism in Organic Chemistry
This question tests your understanding of structural differences between molecules, specifically the types of isomers that can exist in organic compounds.
Key Terms:
Isomers: Molecules with the same molecular formula but different structures.
Structural Isomers: Isomers that differ in the connectivity of their atoms.
Cis-trans Isomers: Isomers that differ in the arrangement around a double bond.
Enantiomers: Isomers that are mirror images of each other.
Step-by-Step Guidance
Examine the molecular formula for both molecules. Are the numbers and types of atoms the same?
Look at the connectivity of atoms in each molecule. Are the atoms connected in the same way, or is there a difference in the arrangement?
Consider whether the molecules are mirror images (enantiomers), differ in arrangement around a double bond (cis-trans isomers), or have different connectivity (structural isomers).
Compare the functional groups and their positions in each molecule. Are they in the same place or different places?
Try solving on your own before revealing the answer!
Final Answer: C. They are structural isomers
The two molecules have the same molecular formula but differ in the connectivity of their atoms, making them structural isomers.
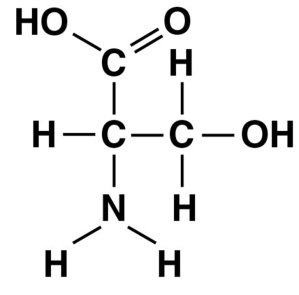
Q44. Name the functional group that is circled in the molecule pictured below.
Background
Topic: Functional Groups in Organic Molecules
This question tests your ability to identify common functional groups in organic chemistry, which are important for understanding chemical reactivity and properties.
Key Terms:
Hydroxyl group:
Carbonyl group:
Carboxyl group:
Amino group:
Step-by-Step Guidance
Locate the circled group in the molecule.
Identify the atoms present in the circled group (look for C, O, H, or N).
Determine if the group contains a double-bonded oxygen (carbonyl), a hydroxyl (), a carboxyl (), or an amino ().
Compare the structure to the definitions of each functional group listed above.
Try solving on your own before revealing the answer!
Final Answer: D. Amino group
The circled group contains a nitrogen atom bonded to two hydrogens, which is characteristic of an amino group ().
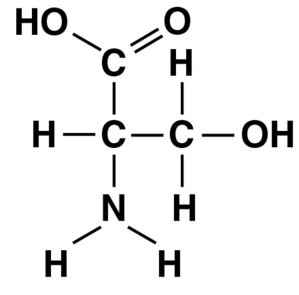
Q45. Name the functional group that is circled in the molecule pictured below.
Background
Topic: Functional Groups in Organic Molecules
This question tests your ability to identify functional groups, which are key to understanding the chemical properties of organic molecules.
Key Terms:
Hydroxyl group:
Carbonyl group:
Carboxyl group:
Amino group:
Step-by-Step Guidance
Find the circled group in the molecule.
Identify the atoms and bonds in the circled group.
Check for the presence of a double-bonded oxygen (carbonyl), a hydroxyl (), a carboxyl (), or an amino ().
Match the structure to the definitions of the functional groups above.
Try solving on your own before revealing the answer!
Final Answer: D. Amino group
The circled group is an amino group (), which is a nitrogen atom bonded to two hydrogens.
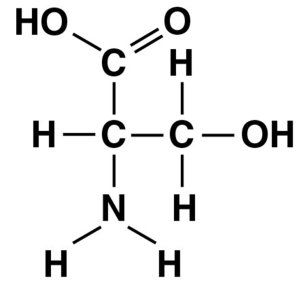
Q46. Name the functional group that is circled in the molecule pictured below.
Background
Topic: Functional Groups in Organic Molecules
This question tests your ability to recognize and name functional groups, which are important for predicting the behavior of organic molecules.
Key Terms:
Hydroxyl group:
Carbonyl group:
Carboxyl group:
Amino group:
Step-by-Step Guidance
Locate the circled functional group in the molecule.
Identify the atoms and their arrangement in the circled group.
Determine if the group matches the structure of a hydroxyl, carbonyl, carboxyl, or amino group.
Compare the group to the definitions provided above.
Try solving on your own before revealing the answer!
Final Answer: D. Amino group
The circled group is an amino group (), which is a nitrogen atom bonded to two hydrogens.
