 Back
BackStudy Guide: Proteins, Energy, and Enzymes in Biology
Study Guide - Smart Notes

Q1. Draw and describe the structure of an amino acid, including the central carbon, carboxyl group, amino group, and side chain.
Background
Topic: Amino Acid Structure
This question tests your understanding of the basic building blocks of proteins—amino acids—and their structural components.
Key Terms:
Central (alpha) carbon (Cα): The central atom to which all other groups are attached.
Amino group (-NH2): A functional group containing nitrogen and hydrogen.
Carboxyl group (-COOH): A functional group containing carbon, oxygen, and hydrogen.
Side chain (R group): The variable group that determines the identity and properties of the amino acid.
Step-by-Step Guidance
Draw a central carbon atom (Cα).
Attach an amino group (-NH2) to one side of the central carbon.
Attach a carboxyl group (-COOH) to the opposite side of the central carbon.
Attach a hydrogen atom to the central carbon.
Attach a variable side chain (R group) to the central carbon. The R group is what makes each amino acid unique.
Try solving on your own before revealing the answer!
Q2. Predict whether a side chain is nonpolar, polar uncharged, or polar charged.
Background
Topic: Amino Acid Side Chains
This question tests your ability to classify amino acids based on the chemical properties of their side chains (R groups).
Key Terms:
Nonpolar side chains: Typically contain hydrocarbons (C and H only).
Polar uncharged side chains: Contain electronegative atoms (like O or N) but no charge.
Polar charged side chains: Contain groups that are ionized at physiological pH (positive or negative charge).
Step-by-Step Guidance
Look for hydrocarbon chains or rings (C and H only) to identify nonpolar side chains.
Check for the presence of oxygen or nitrogen atoms without a charge to identify polar uncharged side chains.
Look for side chains with a positive (+) or negative (−) charge to identify polar charged side chains.
Try solving on your own before revealing the answer!
Q3. Contrast hydrolysis and condensation reactions.
Background
Topic: Chemical Reactions in Biology
This question tests your understanding of how biological macromolecules are built and broken down.
Key Terms:
Hydrolysis: Breaking a bond by adding water.
Condensation (Dehydration Synthesis): Forming a bond by removing water.
Step-by-Step Guidance
Describe how hydrolysis uses water to break covalent bonds (e.g., in proteins or polysaccharides).
Explain how condensation reactions remove a water molecule to form a new covalent bond between monomers.
Contrast the direction of each reaction (hydrolysis breaks down polymers; condensation builds them).
Try solving on your own before revealing the answer!
Q4. Define the primary structure of a protein and identify and locate the peptide bonds that maintain primary structure.
Background
Topic: Protein Structure
This question tests your understanding of the sequence of amino acids in a protein and the bonds that hold them together.
Key Terms:
Primary structure: The linear sequence of amino acids in a protein.
Peptide bond: The covalent bond between the carboxyl group of one amino acid and the amino group of the next.
Step-by-Step Guidance
Define the primary structure as the unique sequence of amino acids in a polypeptide chain.
Identify the peptide bonds as the links between the carboxyl group of one amino acid and the amino group of the next.
Locate the peptide bonds in a diagram of a polypeptide chain (look for C-N bonds between amino acids).
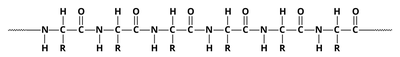
Try solving on your own before revealing the answer!
Q5. Identify and contrast two types of secondary structure and identify the interactions that stabilize these structures.
Background
Topic: Protein Secondary Structure
This question tests your knowledge of alpha helices and beta sheets, and the forces that stabilize them.
Key Terms:
Alpha helix: A coiled structure stabilized by hydrogen bonds.
Beta pleated sheet: A sheet-like structure stabilized by hydrogen bonds between strands.
Hydrogen bonds: The main stabilizing force for secondary structures.
Step-by-Step Guidance
Describe the alpha helix and beta sheet as two common secondary structures in proteins.
Explain how hydrogen bonds form between backbone atoms (not side chains) to stabilize these structures.
Contrast the shapes: alpha helix is a coil, beta sheet is a folded sheet.
Try solving on your own before revealing the answer!
Q6. Identify the level of protein structure that determines the three dimensional shape of a protein and list four types of interactions that contribute to this level of structure.
Background
Topic: Tertiary Structure of Proteins
This question tests your understanding of how proteins fold into their functional shapes.
Key Terms:
Tertiary structure: The overall 3D shape of a single polypeptide chain.
Interactions: Hydrogen bonds, ionic bonds, hydrophobic interactions, disulfide bridges.
Step-by-Step Guidance
Identify tertiary structure as the level responsible for the 3D shape of a protein.
List four types of interactions: hydrogen bonds, ionic bonds, hydrophobic interactions, and disulfide bridges.
Briefly describe each interaction and how it contributes to protein folding.
Try solving on your own before revealing the answer!
Q7. Define the quaternary structure of a protein.
Background
Topic: Protein Structure
This question tests your understanding of how multiple polypeptide chains can assemble into a functional protein complex.
Key Terms:
Quaternary structure: The arrangement of multiple polypeptide subunits in a protein.
Step-by-Step Guidance
Define quaternary structure as the association of two or more polypeptide chains (subunits).
Explain that not all proteins have quaternary structure—only those with multiple subunits.
Try solving on your own before revealing the answer!
Q8. Discuss the role of chaperone proteins in protein folding.
Background
Topic: Protein Folding
This question tests your understanding of how cells ensure proteins fold correctly.
Key Terms:
Chaperone proteins: Proteins that assist other proteins in folding correctly.
Step-by-Step Guidance
Describe how chaperone proteins bind to nascent or misfolded proteins.
Explain that chaperones prevent aggregation and assist in achieving the correct conformation.
Try solving on your own before revealing the answer!
Q9. Predict the potential consequence to protein function if the primary structure of the protein is altered.
Background
Topic: Protein Structure and Function
This question tests your understanding of the relationship between amino acid sequence and protein function.
Key Terms:
Primary structure: The sequence of amino acids.
Mutation: A change in the amino acid sequence.
Step-by-Step Guidance
Explain that the primary structure determines all higher levels of protein structure.
Discuss how a change in sequence can alter folding and function, potentially leading to loss of function or disease.
Try solving on your own before revealing the answer!
Q10. Define and differentiate between potential energy and Gibb’s free energy.
Background
Topic: Energy in Biological Systems
This question tests your understanding of energy concepts relevant to metabolism.
Key Terms:
Potential energy: Stored energy due to position or structure.
Gibbs free energy (G): The energy available to do work in a system at constant temperature and pressure.
Step-by-Step Guidance
Define potential energy as energy stored in chemical bonds or gradients.
Define Gibbs free energy as the portion of a system's energy that can perform work.
Contrast the two: potential energy is a broader concept, while Gibbs free energy is specific to chemical reactions.
Try solving on your own before revealing the answer!
Q11. Define and contrast endergonic and exergonic reactions with respect to changes in Gibb’s free energy.
Background
Topic: Thermodynamics of Reactions
This question tests your understanding of how energy changes during chemical reactions.
Key Terms:
Endergonic reaction: Requires input of energy; ΔG > 0.
Exergonic reaction: Releases energy; ΔG < 0.
Step-by-Step Guidance
Define endergonic and exergonic reactions in terms of Gibbs free energy change (ΔG).
Explain that endergonic reactions are non-spontaneous and require energy input, while exergonic reactions are spontaneous and release energy.
Relate these concepts to biological processes (e.g., ATP hydrolysis is exergonic).
Try solving on your own before revealing the answer!
Q12. Define and contrast anabolic and catabolic reactions and give an example of each.
Background
Topic: Metabolism
This question tests your understanding of the two main types of metabolic pathways.
Key Terms:
Anabolic reactions: Build complex molecules from simpler ones; require energy.
Catabolic reactions: Break down complex molecules into simpler ones; release energy.
Step-by-Step Guidance
Define anabolic reactions and provide an example (e.g., protein synthesis).
Define catabolic reactions and provide an example (e.g., cellular respiration).
Contrast the energy requirements and outcomes of each type of reaction.
Try solving on your own before revealing the answer!
Q13. Correlate the following terms and describe how they relate to each other: endergonic/exergonic, anabolic/catabolic, condensation/hydrolysis.
Background
Topic: Metabolic Pathways and Energy
This question tests your ability to connect different types of reactions and their energy profiles.
Key Terms:
Endergonic: Energy-requiring
Exergonic: Energy-releasing
Anabolic: Building up
Catabolic: Breaking down
Condensation: Building by removing water
Hydrolysis: Breaking by adding water
Step-by-Step Guidance
Match anabolic reactions with endergonic and condensation reactions (building up, energy input, water removal).
Match catabolic reactions with exergonic and hydrolysis reactions (breaking down, energy release, water addition).
Explain how these pairs are related in biological systems.
Try solving on your own before revealing the answer!
Q14. Identify the activation energy, change in free energy (ΔG), and transition state on a graph of the free energy change of a reaction.
Background
Topic: Reaction Energy Diagrams
This question tests your ability to interpret energy diagrams and identify key features.
Key Terms:
Activation energy (Ea): The energy required to reach the transition state.
ΔG (change in free energy): The difference in free energy between reactants and products.
Transition state: The highest energy point along the reaction pathway.
Step-by-Step Guidance
Locate the reactants and products on the y-axis (Gibbs free energy) of the graph.
Identify the peak of the curve as the transition state.
Measure the vertical distance from reactants to the peak for activation energy (Ea).
Measure the vertical distance from reactants to products for ΔG.
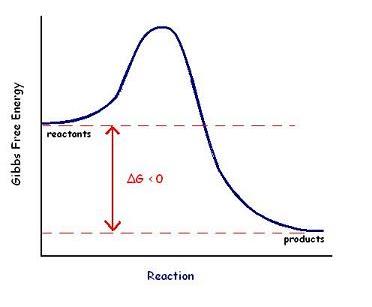
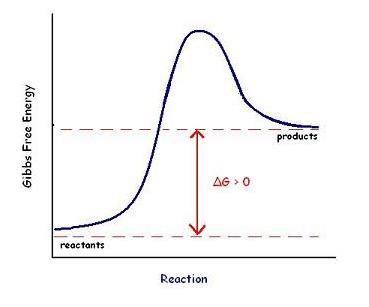
Try solving on your own before revealing the answer!
Q15. Identify whether a reaction is endergonic or exergonic using a graph of the free energy change of a reaction.
Background
Topic: Reaction Energy Diagrams
This question tests your ability to interpret whether a reaction absorbs or releases energy based on a graph.
Key Terms:
Exergonic: ΔG < 0 (products have lower free energy than reactants).
Endergonic: ΔG > 0 (products have higher free energy than reactants).
Step-by-Step Guidance
Examine the relative positions of reactants and products on the y-axis (Gibbs free energy).
If products are lower than reactants, the reaction is exergonic (ΔG < 0).
If products are higher than reactants, the reaction is endergonic (ΔG > 0).


Try solving on your own before revealing the answer!
Q16. Draw how a graph of the free energy change of a reaction is altered in the presence of an enzyme and describe the effect on the rate of the reaction and overall change in free energy (ΔG) of the reaction.
Background
Topic: Enzyme Catalysis
This question tests your understanding of how enzymes affect reaction rates and energy profiles.
Key Terms:
Enzyme: A biological catalyst that speeds up reactions.
Activation energy (Ea): Lowered by enzymes.
ΔG: Unchanged by enzymes.
Step-by-Step Guidance
Draw or visualize a reaction energy diagram with and without an enzyme.
Show that the enzyme lowers the activation energy (the peak is lower), but the difference between reactants and products (ΔG) stays the same.
Explain that lowering activation energy increases the reaction rate.
Try solving on your own before revealing the answer!
Q17. Identify the location of an active site on an enzyme and explain the role of the active site.
Background
Topic: Enzyme Structure and Function
This question tests your understanding of how enzymes interact with substrates.
Key Terms:
Active site: The region on the enzyme where the substrate binds and the reaction occurs.
Step-by-Step Guidance
Identify the active site as a pocket or groove on the enzyme's surface.
Explain that the active site is specific to the substrate and facilitates the chemical reaction.
Try solving on your own before revealing the answer!
Q18. Contrast allosteric inhibition versus competitive inhibition.
Background
Topic: Enzyme Regulation
This question tests your understanding of different mechanisms that regulate enzyme activity.
Key Terms:
Competitive inhibition: Inhibitor binds to the active site, blocking substrate binding.
Allosteric inhibition: Inhibitor binds to a site other than the active site, changing enzyme shape and function.
Step-by-Step Guidance
Describe competitive inhibition as direct competition for the active site.
Describe allosteric inhibition as binding elsewhere, causing a conformational change that reduces activity.
Contrast the reversibility and effects on enzyme kinetics.
Try solving on your own before revealing the answer!
Q19. Describe the process of negative feedback inhibition.
Background
Topic: Metabolic Regulation
This question tests your understanding of how cells regulate metabolic pathways to maintain homeostasis.
Key Terms:
Negative feedback inhibition: The end product of a pathway inhibits an earlier step, preventing overproduction.
Step-by-Step Guidance
Describe how the final product of a metabolic pathway can bind to an enzyme early in the pathway.
Explain that this binding reduces the enzyme's activity, slowing or stopping the pathway.
Try solving on your own before revealing the answer!
Q20. Predict how alterations in enzyme shape could affect enzyme function.
Background
Topic: Enzyme Structure and Function
This question tests your understanding of the relationship between enzyme structure and activity.
Key Terms:
Enzyme conformation: The 3D shape of the enzyme.
Active site: The region where substrate binds.
Step-by-Step Guidance
Explain that enzyme function depends on the precise shape of the active site.
Discuss how changes in shape (due to mutation, pH, temperature) can reduce or eliminate activity.
Try solving on your own before revealing the answer!
Q21. Predict the potential consequence of a nonfunctional enzyme for a cell and discuss the role of enzyme regulation in determining what reactions occur within a cell.
Background
Topic: Enzyme Function and Regulation
This question tests your understanding of the importance of enzymes in cellular metabolism.
Key Terms:
Nonfunctional enzyme: An enzyme that cannot catalyze its reaction.
Enzyme regulation: Mechanisms that control enzyme activity.
Step-by-Step Guidance
Predict that a nonfunctional enzyme can block a metabolic pathway, leading to accumulation of substrates or deficiency of products.
Discuss how cells regulate enzymes to control which reactions occur and when.
Try solving on your own before revealing the answer!
Q22. Describe the role of cofactors and coenzymes in enzyme function and contrast their structures.
Background
Topic: Enzyme Helpers
This question tests your understanding of molecules that assist enzymes in catalysis.
Key Terms:
Cofactor: An inorganic ion (e.g., metal ion) required for enzyme activity.
Coenzyme: An organic molecule (often derived from vitamins) that assists enzymes.
Step-by-Step Guidance
Describe how cofactors and coenzymes bind to enzymes and are necessary for activity.
Contrast their structures: cofactors are usually metal ions, while coenzymes are organic molecules.
Try solving on your own before revealing the answer!
