 Back
BackThe Chemical Basis of Life: Elements, Atoms, and Water’s Role in Biology
Study Guide - Smart Notes

The Chemical Basis of Life
Introduction: Chemistry and Life
All living organisms are fundamentally chemical systems. The chemistry of life is closely tied to water, as life first evolved in aquatic environments and all cells are composed of about 70% water. Understanding the chemical basis of life is essential for explaining biological phenomena, such as how atmospheric compounds can impact coral reefs through chemical reactions.

Elements, Atoms, and Compounds
Elements Essential for Life
Matter is composed of chemical elements, substances that cannot be broken down into other substances by chemical means. About 25 elements are essential for human life, with four elements—oxygen (O), carbon (C), hydrogen (H), and nitrogen (N)—making up about 96% of the mass of most living organisms. Compounds are substances consisting of two or more different elements combined in a fixed ratio.
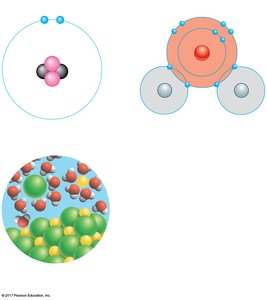
Emergent Properties of Compounds
Compounds often have properties distinct from the elements that compose them. For example, sodium (a reactive metal) and chlorine (a poisonous gas) combine to form sodium chloride (table salt), which is edible and essential for life. This illustrates the concept of emergent properties—new characteristics that arise when elements combine to form compounds.
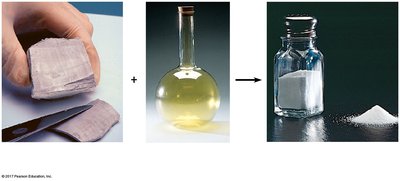
Major and Trace Elements in the Human Body
The human body is primarily composed of a few major elements, with trace elements required in minute amounts for proper physiological function. Deficiencies in trace elements can lead to disease, and some are added to food and water to prevent health issues (e.g., fluoride in water to prevent tooth decay).
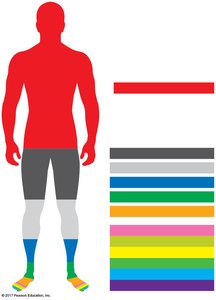
Trace Elements and Health
Trace elements such as iodine and iron are vital for health. Iodine deficiency can cause thyroid gland enlargement (goiter), while iron is essential for oxygen transport in blood. Dietary sources of iron include meats, legumes, and leafy greens.


Atoms: Structure and Properties
Atomic Structure
An atom is the smallest unit of matter that retains the properties of an element. Atoms are composed of three subatomic particles:
Protons: Positively charged, found in the nucleus
Neutrons: No charge, found in the nucleus
Electrons: Negatively charged, orbit the nucleus
The atomic number is the number of protons in an atom, defining the element. The mass number is the sum of protons and neutrons.
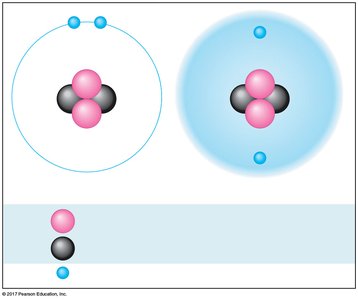
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons. For example, carbon has three naturally occurring isotopes: carbon-12, carbon-13, and carbon-14. Isotopes have the same chemical properties but different mass numbers.
Carbon-12 | Carbon-13 | Carbon-14 | |
|---|---|---|---|
Protons | 6 | 6 | 6 |
Neutrons | 6 | 7 | 8 |
Electrons | 6 | 6 | 6 |
Mass number | 12 | 13 | 14 |

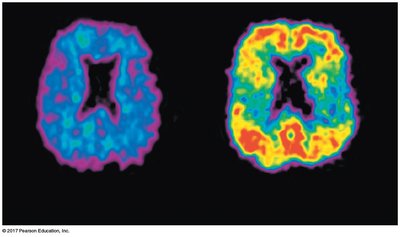
Radioactive Isotopes
Some isotopes are radioactive, meaning they decay spontaneously, releasing particles and energy. Radioactive isotopes are used as tracers in medical imaging and biological research, but they can also pose health risks.

Chemical Bonds
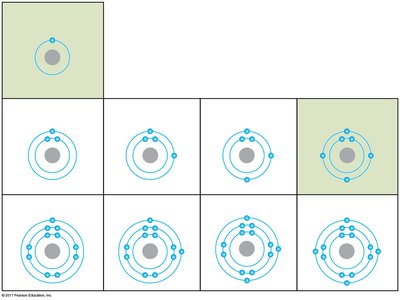
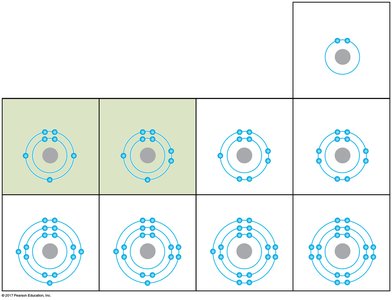
Electron Shells and Chemical Behavior
Electrons occupy specific energy levels called shells. The chemical properties of an atom are determined by the arrangement of electrons in these shells, especially the outermost shell (valence shell). Atoms with incomplete valence shells tend to form chemical bonds to achieve stability.
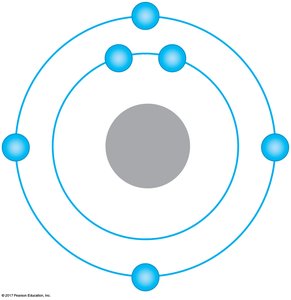
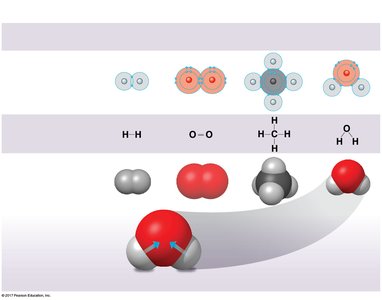
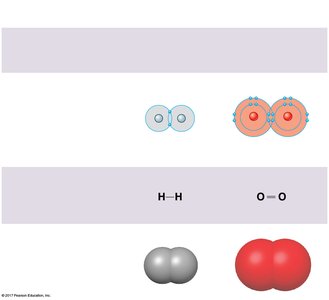
Covalent Bonds
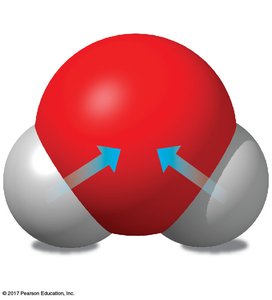
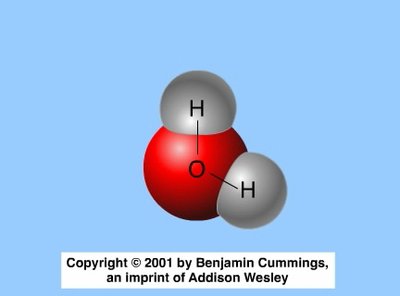
Covalent bonds form when two atoms share one or more pairs of electrons. These bonds can be nonpolar (equal sharing, as in H2 or CH4) or polar (unequal sharing, as in H2O). The polarity of water molecules leads to many of its unique properties.
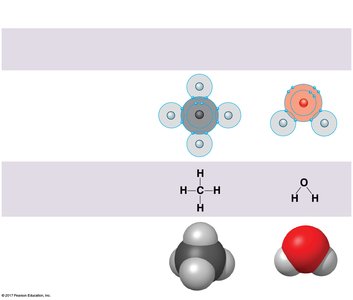
Ionic Bonds
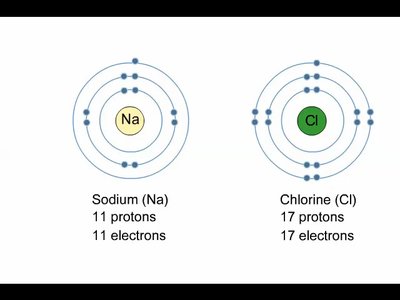
Ionic bonds are formed when one atom transfers electrons to another, resulting in oppositely charged ions that attract each other. For example, sodium (Na) donates an electron to chlorine (Cl), forming Na+ and Cl−, which combine to make sodium chloride (NaCl).



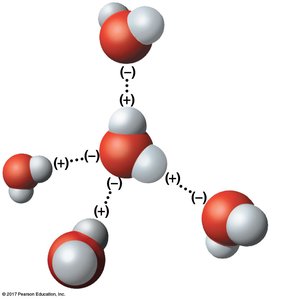
Hydrogen Bonds
Hydrogen bonds are weak attractions between the slightly positive hydrogen atom of one polar molecule and the slightly negative atom of another. In water, hydrogen bonds are responsible for many of its life-supporting properties.
Chemical Reactions
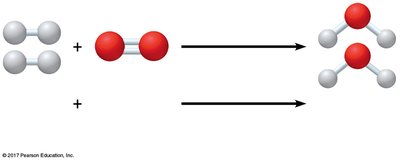
Chemical reactions involve the making and breaking of chemical bonds, resulting in the rearrangement of atoms to form new substances. For example, the reaction of hydrogen and oxygen gases forms water:
Water’s Life-Supporting Properties
Cohesion, Adhesion, and Surface Tension
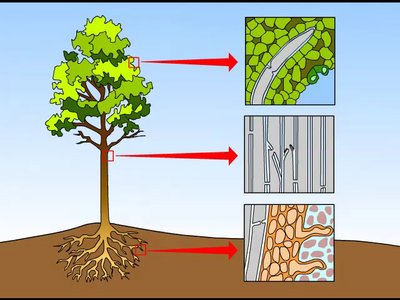
Water molecules stick together (cohesion) due to hydrogen bonding, which also gives water a high surface tension. Adhesion is the tendency of water molecules to stick to other substances, aiding in processes like water transport in plants.

Temperature Moderation
Water moderates temperature by absorbing heat when hydrogen bonds break and releasing heat when they form. This property helps stabilize temperatures in organisms and environments. Evaporative cooling occurs as the most energetic water molecules leave the surface, cooling the remaining liquid.

Ice Floats: Density of Water
Unlike most substances, solid water (ice) is less dense than liquid water due to the stable hydrogen bonds that form a crystalline structure. This allows ice to float, insulating aquatic life in cold climates.


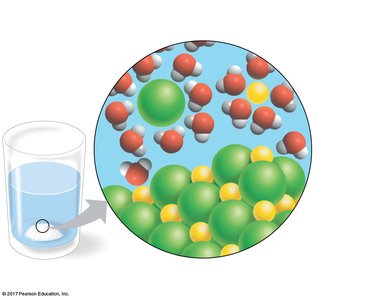
Water as the Solvent of Life
Water’s polarity makes it an excellent solvent, especially for ionic and polar substances. Biological fluids, including blood, are aqueous solutions where water dissolves and transports essential molecules.
Acids, Bases, and pH
The pH scale measures the concentration of hydrogen ions (H+) in a solution. Acids increase H+ concentration, while bases decrease it. Buffers help maintain stable pH in biological systems, which is crucial for cellular function.
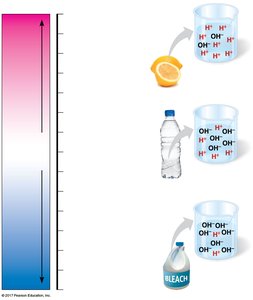
Impact of Chemistry on the Biosphere
Ocean Acidification and Coral Reefs
Rising atmospheric CO2 from fossil fuel combustion dissolves in oceans, forming carbonic acid and lowering ocean pH—a process called ocean acidification. This reduces carbonate ion concentration, impairing the ability of corals and shell-building organisms to form calcium carbonate skeletons, threatening marine ecosystems.
Water and the Search for Extraterrestrial Life
The unique properties of water are essential for life as we know it. The search for extraterrestrial life often focuses on finding water, as its presence may indicate the potential for life-supporting environments elsewhere in the universe.
Summary Table: Types of Chemical Bonds
Bond Type | Description | Example |
|---|---|---|
Covalent | Atoms share electrons | H2O, CH4 |
Ionic | Atoms transfer electrons, forming ions | NaCl |
Hydrogen | Weak attraction between polar molecules | Between H2O molecules |
Additional info: This guide expands on the PowerPoint lecture content with definitions, examples, and academic context to provide a comprehensive overview of the chemical basis of life, suitable for college-level biology students.
