 Back
BackThe Chemical Basis of Life: Elements, Compounds, and Atomic Structure
Study Guide - Smart Notes

The Chemistry of Life
Why Study Chemistry in Biology?
Chemistry forms the foundation of biological processes, as all living organisms are composed of chemical elements and compounds. Understanding the chemical basis of life is essential for comprehending cellular structure, function, and metabolism.
Elements and Compounds
Elements are pure substances consisting of only one type of atom and cannot be broken down further by chemical means. Compounds are substances formed from two or more elements in a fixed ratio, resulting in molecules with unique properties.
Element: Pure substance; represented on the periodic table; e.g., Oxygen (O), Carbon (C).
Compound: Combination of elements; e.g., Water (H2O), Brass (CuZn).
Molecule: Smallest unit of a compound retaining its chemical properties.
Example: Water is a compound made of hydrogen and oxygen.
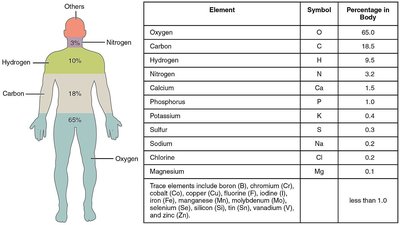
Major Elements in Biological Systems
The human body is primarily composed of a few key elements, each playing vital roles in biological functions.
Oxygen (O): 65% of body mass; essential for cellular respiration.
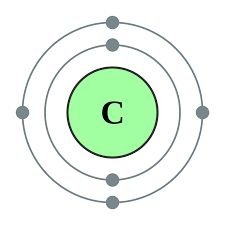
Carbon (C): 18.5%; backbone of organic molecules.
Hydrogen (H): 9.5%; component of water and organic compounds.
Nitrogen (N): 3.2%; found in amino acids and nucleic acids.
Other elements: Calcium, Phosphorus, Potassium, Sulfur, Sodium, Chlorine, Magnesium, and trace elements.
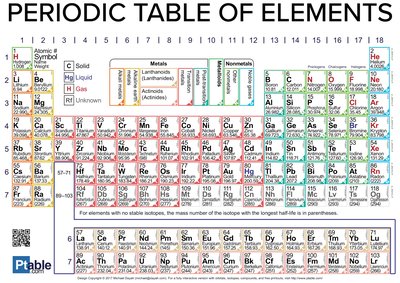
The Periodic Table of Elements
The periodic table organizes elements by atomic number and properties, providing a reference for understanding their chemical behavior and biological significance.
Groups: Columns with similar chemical properties.
Periods: Rows indicating increasing atomic number.
Biologically important elements: Highlighted in the table for their roles in life processes.
Atomic Structure
Atoms and Subatomic Particles
An atom consists of a nucleus containing protons and neutrons, surrounded by electrons in shells. The arrangement of electrons determines chemical reactivity.
Proton: Positively charged particle in the nucleus.
Neutron: Neutral particle in the nucleus.
Electron: Negatively charged particle in orbitals around the nucleus.
Atomic number: Number of protons; defines the element.
Atomic mass: Sum of protons and neutrons; measured in Daltons (Da).
Isotopes: Atoms of the same element with different numbers of neutrons.
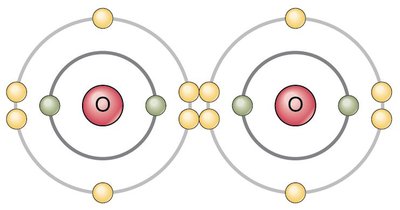
Electron Configuration and Chemical Bonding
Electrons are arranged in shells, with each shell having a maximum capacity. The stability of an atom is determined by the filling of these shells, leading to the formation of chemical bonds.
1st shell: Stable with 2 electrons.
2nd shell: Stable with 8 electrons.
3rd shell: Stable with 8 electrons.
Octet rule: Atoms tend to fill their outermost shell with 8 electrons for stability.

Molecules and Compounds
Formation of Molecules
Atoms combine to form molecules through chemical bonds. Molecules are the smallest units of compounds and retain the compound's properties.
Sodium (Na) and Chlorine (Cl): Combine to form sodium chloride (NaCl), a common salt.
Example: Sodium is a reactive metal, chlorine is a toxic gas, but together they form edible table salt.



Types of Chemical Bonds
Chemical bonds are the forces holding atoms together in molecules. The main types are ionic and covalent bonds.
Ionic bond: Formed by electron transfer and electrostatic attraction between oppositely charged ions (e.g., Na+ and Cl-).
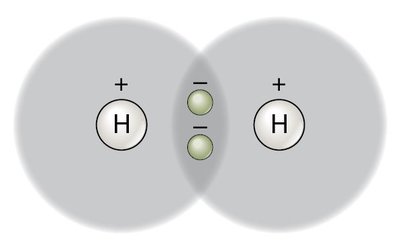
Covalent bond: Formed by sharing electrons between atoms (e.g., H2, O2, CO2).

Electron Configuration and Reactivity
The reactivity of elements is determined by their electron configuration, especially the number of electrons in the outermost shell.
Sodium (Na): Has one electron in its outer shell, making it highly reactive.
Chlorine (Cl): Needs one electron to fill its outer shell, making it reactive.
Example: Sodium reacts with water to produce sodium hydroxide and hydrogen gas.

Covalent Bonds: Polar and Nonpolar
Polar vs. Nonpolar Covalent Bonds
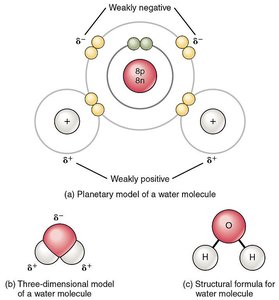
Covalent bonds can be classified as polar or nonpolar based on the sharing of electrons. Electronegativity is the tendency of an atom to attract electrons.
Nonpolar covalent bond: Electrons are shared equally (e.g., H2).
Polar covalent bond: Electrons are shared unequally, resulting in partial charges (e.g., H2O).
Electronegativity: Large differences lead to polar bonds; O, Cl, and N are highly electronegative, H and Na are low, C is intermediate.
Ionic Bonds
Formation of Ionic Bonds
Ionic bonds are formed when electrons are transferred from one atom to another, resulting in charged ions that attract each other.
Example: Sodium (Na) loses an electron to become Na+, chlorine (Cl) gains an electron to become Cl-, forming NaCl.
Summary Table: Elements in the Human Body
The following table summarizes the major elements found in the human body, their symbols, and their approximate percentages.
Element | Symbol | Percentage in Body |
|---|---|---|
Oxygen | O | 65.0 |
Carbon | C | 18.5 |
Hydrogen | H | 9.5 |
Nitrogen | N | 3.2 |
Calcium | Ca | 1.5 |
Phosphorus | P | 1.0 |
Potassium | K | 0.4 |
Sulfur | S | 0.3 |
Sodium | Na | 0.2 |
Chlorine | Cl | 0.2 |
Magnesium | Mg | 0.1 |
Trace elements | - | less than 1.0 |
Additional info:
Trace elements, though present in small amounts, are essential for various biochemical processes.
Understanding atomic structure and chemical bonding is fundamental for topics such as metabolism, genetics, and cellular physiology.
