 Back
BackThe Chemistry of Life: Structure and Function of Proteins 2/18
Study Guide - Smart Notes

The Chemistry of Life
Biological Macromolecules
Biological macromolecules are essential for life and include proteins, lipids, carbohydrates, and nucleic acids. Proteins are polymers made from amino acids and are responsible for a wide range of functions in living organisms.
Proteins and Amino Acids
Structure of Amino Acids
Amino acids are the building blocks of proteins. Each amino acid has a central carbon atom (the alpha carbon) bonded to an amine group, a carboxylic acid group, a hydrogen atom, and a variable "R" group (side chain). The "R" group determines the properties and identity of each amino acid.
Amine Group: NH2
Carboxyl Group: COOH
R Group: Variable side chain
Central Hydrocarbon: Alpha carbon
Amino acids are zwitterionic at neutral pH, meaning they possess both a positive and negative charge.
Classification of Amino Acids
Amino acids are classified based on the properties of their side chains:
Hydrophobic (Nonpolar) Amino Acids: Side chains are nonpolar and tend to avoid water.
Hydrophilic (Polar) Amino Acids: Side chains are polar and interact well with water.
Special Amino Acids: Some amino acids, like cysteine, have unique side chains that can form disulfide bonds.
Special Amino Acids and Disulfide Bonds
Cysteine contains a sulfhydryl (-SH) group, which can form disulfide bonds with another cysteine, stabilizing protein structure. Disulfide bonds are especially important in extracellular proteins and contribute to the strength and stability of protein structures.
Polymerization of Macromolecules
Dehydration Synthesis and Hydrolysis
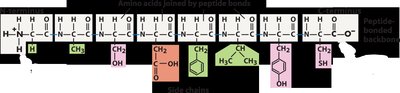
Proteins are formed by linking amino acids through dehydration synthesis (condensation reaction), which removes water to form a peptide bond. The reverse process, hydrolysis, breaks peptide bonds by adding water.
Dehydration Synthesis: Formation of peptide bonds, water is removed.
Hydrolysis: Breaking of peptide bonds, water is added.
Polypeptide Chains and Polarity
Chains of amino acids are called polypeptides. Polypeptides have polarity, with an N-terminus (free amine group) and a C-terminus (free carboxyl group). The sequence of amino acids determines the protein's primary structure.
Protein Structure
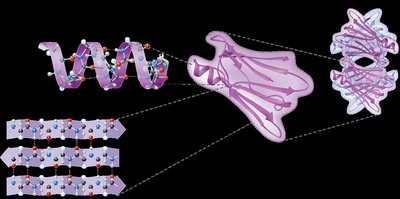
Levels of Protein Structure
Proteins fold into specific structures that are essential for their function. There are four levels of protein structure:
Primary Structure: Sequence of amino acids in a polypeptide chain.
Secondary Structure: Local folding patterns such as alpha helices and beta sheets, stabilized by hydrogen bonds.
Tertiary Structure: Overall three-dimensional shape of a single polypeptide, stabilized by hydrophobic interactions, van der Waals forces, and hydrogen bonds.
Quaternary Structure: Association of multiple polypeptide chains to form a functional protein.
Primary Structure
The primary structure is the linear sequence of amino acids. This sequence determines all higher levels of structure and ultimately the protein's function.
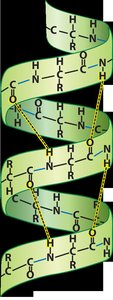
Secondary Structure
Secondary structure arises from hydrogen bonding between the backbone atoms of the polypeptide. The most common secondary structures are the alpha (α) helix and beta (β) pleated sheet.
Alpha Helix: A right-handed coil stabilized by hydrogen bonds.
Beta Sheet: Sheet-like structure stabilized by hydrogen bonds between adjacent strands.
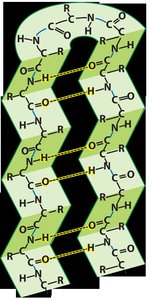
Tertiary Structure
Tertiary structure refers to the overall three-dimensional shape of a polypeptide. It is primarily stabilized by hydrophobic interactions, van der Waals forces, and hydrogen bonds. Hydrophobic side chains tend to cluster in the interior, while polar side chains are exposed to the aqueous environment.
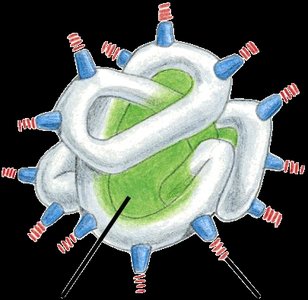
Quaternary Structure
Quaternary structure is the association of two or more polypeptide chains to form a functional protein. This structure is stabilized by noncovalent interactions such as hydrogen bonds and van der Waals forces.
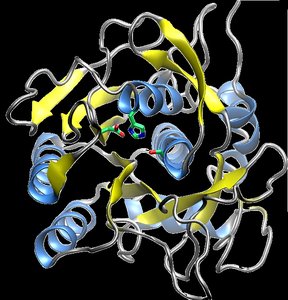
Protein Function and Examples
Functions of Proteins
Proteins perform a wide variety of functions in cells, including catalysis (enzymes), transport, structural support, signaling, regulation, and movement.
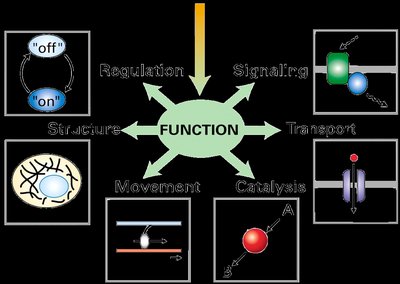
Examples of Protein Function
Luciferase: Enzyme responsible for bioluminescence in fireflies.
Hemoglobin: Protein that transports oxygen in red blood cells.
Collagen: Structural protein stronger than steel, found in connective tissues.

Protein Denaturation
Denaturation and Renaturation
Denaturation is the process by which a protein loses its native structure due to external stress such as heat, pH changes, or ionic strength. This disrupts the noncovalent bonds holding secondary, tertiary, and quaternary structures together. Some proteins can renature if the denaturing agent is removed.
Causes of Denaturation: Heat, pH changes, salt concentration, and other environmental factors.
Effect: Loss of function due to unfolding.
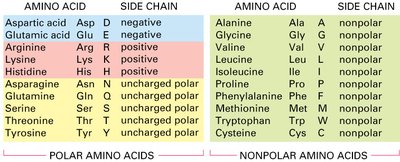
Summary Table: Amino Acid Properties
The following table summarizes the properties of amino acids based on their side chains:
Amino Acid | Abbreviation | Side Chain | Type |
|---|---|---|---|
Aspartic acid | Asp (D) | negative | polar |
Glutamic acid | Glu (E) | negative | polar |
Arginine | Arg (R) | positive | polar |
Lysine | Lys (K) | positive | polar |
Histidine | His (H) | positive | polar |
Asparagine | Asn (N) | uncharged polar | polar |
Glutamine | Gln (Q) | uncharged polar | polar |
Serine | Ser (S) | uncharged polar | polar |
Threonine | Thr (T) | uncharged polar | polar |
Tyrosine | Tyr (Y) | uncharged polar | polar |
Alanine | Ala (A) | nonpolar | nonpolar |
Glycine | Gly (G) | nonpolar | nonpolar |
Valine | Val (V) | nonpolar | nonpolar |
Leucine | Leu (L) | nonpolar | nonpolar |
Isoleucine | Ile (I) | nonpolar | nonpolar |
Proline | Pro (P) | nonpolar | nonpolar |
Phenylalanine | Phe (F) | nonpolar | nonpolar |
Methionine | Met (M) | nonpolar | nonpolar |
Tryptophan | Trp (W) | nonpolar | nonpolar |
Cysteine | Cys (C) | nonpolar | nonpolar |
Key Equations
Peptide Bond Formation
The formation of a peptide bond between two amino acids:
Zwitterion Formation
General zwitterion form of an amino acid:
Conclusion
Understanding the structure and function of proteins is fundamental to biology. The properties of amino acids, the process of polymerization, and the levels of protein structure all contribute to the diverse roles proteins play in living organisms.
