 Back
BackThe Four Major Biomolecules: Structure, Function, and Biological Importance
Study Guide - Smart Notes

Biomolecules (Macromolecules)
Overview of the Four Major Biomolecules
Biomolecules, also known as macromolecules, are large molecules essential for life. The four main classes are proteins, nucleic acids, carbohydrates, and lipids. Each class has unique structures and functions that are critical for cellular processes.
Proteins: Polymers of amino acids, responsible for a vast array of cellular functions.
Nucleic Acids: DNA and RNA, store and transfer genetic information.
Carbohydrates: Sugars and their polymers, provide energy and structural support.
Lipids: Diverse group including fats, phospholipids, and steroids, important for energy storage, membranes, and signaling.
Carbon: The Backbone of Biomolecules
Properties of Carbon
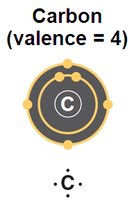
All biomolecules are carbon-based. Carbon's unique ability to form four covalent bonds allows for a variety of stable structures, including chains and rings, which serve as the backbone for complex molecules.
Valence of 4: Carbon can form four covalent bonds, enabling complex branching and ring structures.
Bonding with Many Elements: Carbon bonds with hydrogen, oxygen, nitrogen, and other elements to create diverse molecules.
Examples of Carbon Structures
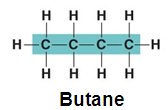
Linear Chains: e.g., Butane
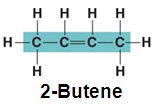
Double Bonds: e.g., 2-Butene
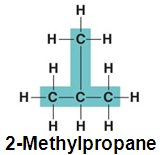
Branched Chains: e.g., 2-Methylpropane
Rings: e.g., Benzene
Polymers and Monomers
Formation and Breakdown of Polymers
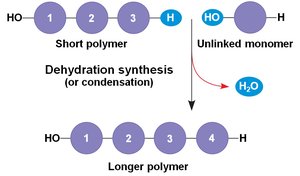
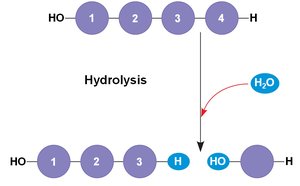
Most biomolecules are polymers, made by linking smaller units called monomers. The process of joining monomers is called dehydration synthesis (or condensation), while breaking them apart is called hydrolysis.
Dehydration Synthesis: Removes a water molecule to form a new bond.
Hydrolysis: Adds a water molecule to break a bond.
Carbohydrates
Structure and Function
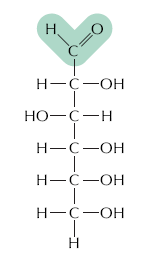
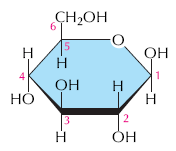
Carbohydrates are composed of carbon, hydrogen, and oxygen, typically with the formula (CH2O)n. They serve as energy sources and structural materials in cells.
Monosaccharides: Simple sugars (e.g., glucose) that are the monomers of carbohydrates.
Disaccharides: Two monosaccharides joined together (e.g., sucrose).
Polysaccharides: Long chains of monosaccharides (e.g., starch, glycogen, cellulose).
Examples and Biological Roles
Glucose (C6H12O6): Main energy source for cells.
Starch and Glycogen: Storage polysaccharides in plants and animals, respectively.
Cellulose: Structural polysaccharide in plant cell walls.
Lipids
Types and Functions
Lipids are hydrophobic molecules that include fats, phospholipids, and steroids. They are not true polymers but are assembled from smaller components.
Fats (Triglycerides): Store energy, composed of glycerol and fatty acids.
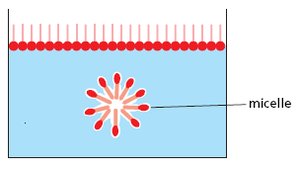
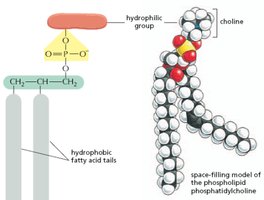
Phospholipids: Major component of cell membranes, with hydrophilic heads and hydrophobic tails.
Steroids: Characterized by four fused carbon rings; include hormones like cholesterol.
Proteins
Structure and Function
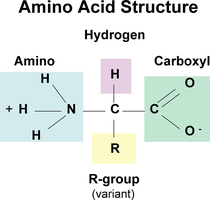
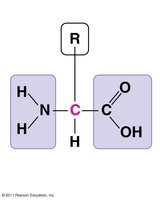
Proteins are polymers of amino acids and perform a vast array of functions in cells, including catalysis, structure, transport, and signaling. The function of a protein is determined by its three-dimensional shape, which is specified by the sequence of amino acids.
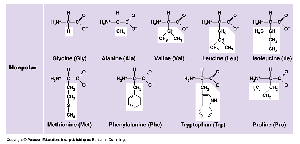
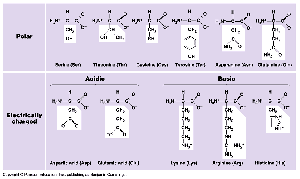
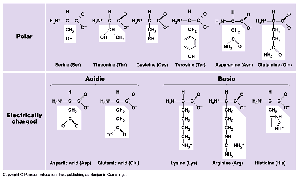
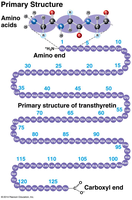
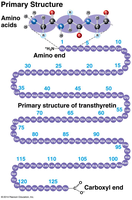
Amino Acids: 20 different types, each with a unique side chain (R-group).
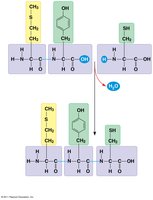
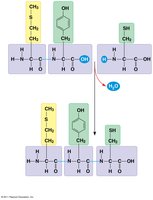
Peptide Bonds: Link amino acids to form polypeptides.
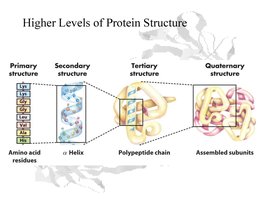
Levels of Structure:
Primary: Sequence of amino acids.
Secondary: Local folding (α-helix, β-sheet) via hydrogen bonds.
Tertiary: Overall 3D shape due to R-group interactions.
Quaternary: Association of multiple polypeptide chains (subunits).
Protein Denaturation
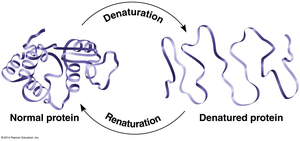
Protein structure is sensitive to environmental conditions such as temperature and pH. Denaturation is the process where proteins lose their structure and, consequently, their function.
Denatured proteins cannot perform their biological roles.

Nucleic Acids
Structure and Function
Nucleic acids are polymers of nucleotides and are responsible for storing, transmitting, and expressing genetic information. The two main types are DNA and RNA.
DNA (Deoxyribonucleic Acid): Stores genetic information.
RNA (Ribonucleic Acid): Transfers genetic information and is involved in protein synthesis.
ATP (Adenosine Triphosphate): Main energy carrier in cells.
cAMP (Cyclic AMP): Functions as a signaling molecule.
Nucleotide Structure
Each nucleotide consists of three components:
Phosphate group
Pentose sugar (deoxyribose in DNA, ribose in RNA)
Nitrogenous base (A, T, C, G in DNA; A, U, C, G in RNA)
DNA Structure
DNA is a double helix composed of two complementary, antiparallel strands held together by hydrogen bonds between specific base pairs (A-T, G-C).
Complementary base pairing: Ensures accurate replication and transcription.
Antiparallel orientation: One strand runs 5' to 3', the other 3' to 5'.
DNA vs. RNA
Feature | DNA | RNA |
|---|---|---|
Sugar | Deoxyribose | Ribose |
Bases | A, T, C, G | A, U, C, G |
Strands | Double-stranded | Single-stranded |
Other Functions of Nucleotides
ATP: Energy currency of the cell.
cAMP: Second messenger in cell signaling.
Summary Table: The Four Major Biomolecules
Biomolecule | Monomer | Polymer | Main Functions |
|---|---|---|---|
Carbohydrates | Monosaccharide | Polysaccharide | Energy, structure |
Lipids | Fatty acids, glycerol | Triglycerides, phospholipids, steroids | Energy storage, membranes, signaling |
Proteins | Amino acid | Polypeptide | Catalysis, structure, transport, signaling |
Nucleic Acids | Nucleotide | DNA, RNA | Information storage and transfer |
