 Back
BackThe Molecules of Life: Structure, Function, and Diversity of Biological Macromolecules
Study Guide - Smart Notes

Chapter 3: The Molecules of Life
Introduction to Biological Molecules
Living organisms are composed of a vast array of molecules, most of which are based on the element carbon. The diversity and complexity of these molecules underlie the structure and function of all living things. This chapter explores the chemistry of carbon, the structure of major biological macromolecules, and their roles in life processes.
Organic Compounds and Carbon Chemistry
Properties of Carbon
Organic compounds are molecules containing carbon and are fundamental to life.
Carbon can form four covalent bonds, allowing for a variety of stable structures, including chains, branches, and rings.
Carbon atoms can bond with hydrogen, oxygen, nitrogen, and other elements, creating diverse molecules.
Examples of carbon skeleton variations:
Length variation
Double bond position
Branching
Ring formation
Simple Organic Compounds
Methane (CH4) is the simplest organic molecule, with one carbon atom bonded to four hydrogens.
Larger hydrocarbons are important as fuels and biological molecules.
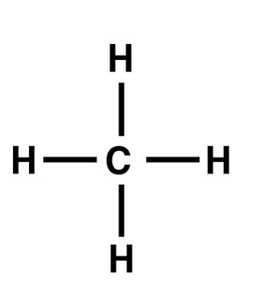
Functional Groups
The chemical behavior of organic molecules is largely determined by functional groups attached to the carbon skeleton.
Common functional groups include hydroxyl, carboxyl, amino, and phosphate groups.
Macromolecules: Polymers and Monomers
Polymer Formation and Breakdown
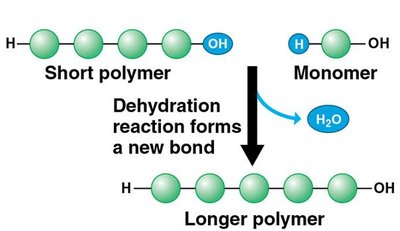
Macromolecules are large molecules formed by joining smaller units called monomers.
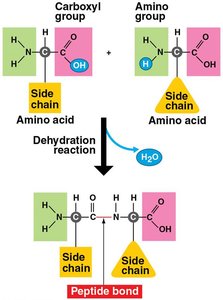
Dehydration reaction: Links monomers by removing a water molecule, forming a polymer.
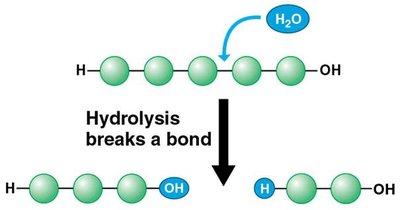
Hydrolysis: Breaks polymers into monomers by adding water.
Categories of Biological Macromolecules
Carbohydrates
Lipids
Proteins
Nucleic acids
Carbohydrates
Monosaccharides
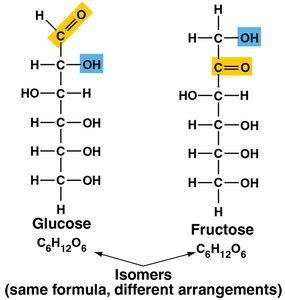
Monosaccharides are the simplest carbohydrates and serve as the building blocks for more complex sugars.
Examples: Glucose and fructose (isomers with the same formula, C6H12O6, but different structures).
Main fuel for cellular work.
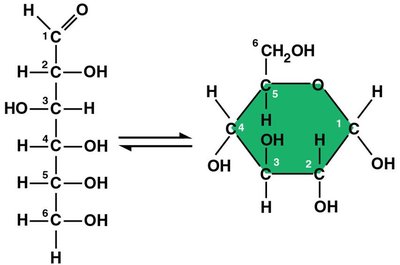
Often form ring structures in aqueous solutions.
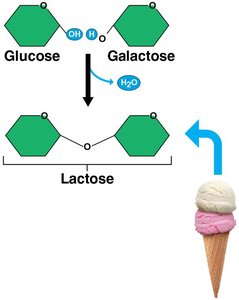
Disaccharides
Disaccharides are formed by joining two monosaccharides via a dehydration reaction.
Examples: Lactose (milk sugar), maltose (malted foods), sucrose (table sugar).
Polysaccharides
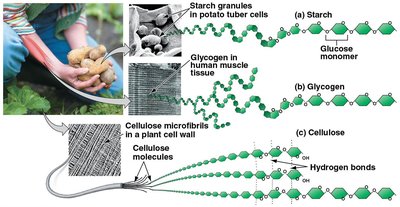
Polysaccharides are complex carbohydrates made of long chains of monosaccharides.
Starch: Energy storage in plants.
Glycogen: Energy storage in animals.
Cellulose: Structural component in plant cell walls; indigestible by animals.
Lipids
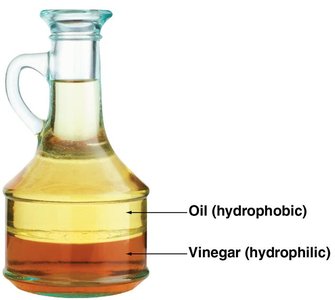
General Properties
Lipids are hydrophobic (water-insoluble) molecules.
They are not always polymers and are structurally diverse.
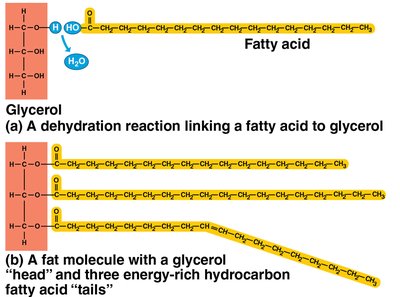
Fats (Triglycerides)
Composed of glycerol and three fatty acids.
Functions: Energy storage, insulation, and cushioning.
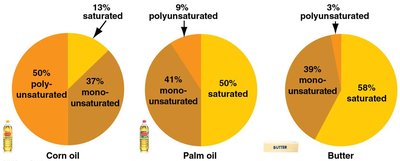
Saturated fats: No double bonds, solid at room temperature, mostly animal fats.
Unsaturated fats: One or more double bonds, liquid at room temperature, mostly plant and fish oils.
Trans fats: Produced by hydrogenation, associated with health risks.




Steroids
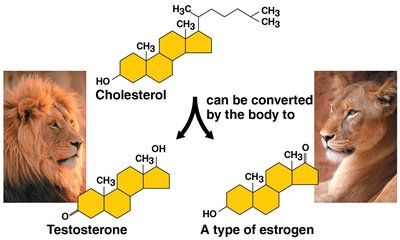
Structure: Four fused carbon rings.
Examples: Cholesterol (cell membrane component), testosterone, and estrogen (hormones).
Synthetic anabolic steroids mimic testosterone and can have medical or harmful effects.

Proteins
Structure and Function
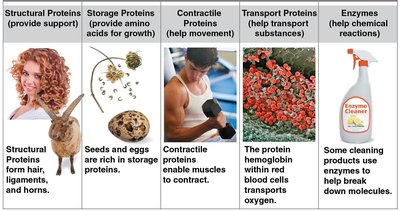
Proteins are polymers of amino acids (20 types).
Roles: Structural support, storage, transport, movement, and catalysis (enzymes).
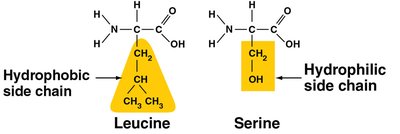
Amino Acids
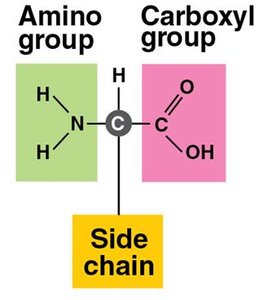
Each amino acid has a central carbon, an amino group, a carboxyl group, a hydrogen atom, and a unique side chain (R group).
Side chains determine properties (hydrophobic/hydrophilic).
Protein Structure
Amino acids are linked by peptide bonds to form polypeptides.
The sequence of amino acids determines the protein's three-dimensional shape and function.
Protein shape is sensitive to environmental conditions (temperature, pH).
Misfolded proteins can cause diseases (e.g., prion diseases, sickle-cell anemia).
Nucleic Acids
DNA and RNA
Nucleic acids store and transmit genetic information.
DNA (deoxyribonucleic acid) and RNA (ribonucleic acid) are polymers of nucleotides.
Each nucleotide consists of a five-carbon sugar, a phosphate group, and a nitrogenous base.
DNA bases: Adenine (A), Thymine (T), Guanine (G), Cytosine (C).
RNA bases: Adenine (A), Uracil (U), Guanine (G), Cytosine (C).
DNA is double-stranded (double helix); RNA is usually single-stranded.
Genetic Information and Evolution
Genes are specific DNA sequences that code for proteins.
Small changes in DNA sequence can affect protein function and lead to genetic disorders (e.g., lactose intolerance, sickle-cell disease).
Evolutionary adaptations can be traced to genetic changes in populations over time.
Case Study: Lactose Intolerance
Lactose intolerance results from the inability to digest lactose due to reduced lactase enzyme production.
Genetic variations in the regulatory region of the lactase gene determine lactose tolerance or intolerance.
Evolutionary perspective: Populations with a history of dairy consumption have higher rates of lactose tolerance due to natural selection.
Summary Table: Major Biological Macromolecules
Macromolecule | Monomer | Function | Example |
|---|---|---|---|
Carbohydrates | Monosaccharide | Energy, structure | Glucose, starch, cellulose |
Lipids | Varied (not always monomers) | Energy storage, membranes, hormones | Fats, oils, steroids |
Proteins | Amino acid | Structure, enzymes, transport | Hemoglobin, enzymes |
Nucleic acids | Nucleotide | Genetic information | DNA, RNA |
Key Equations:
General formula for monosaccharides:
Dehydration reaction:
Hydrolysis:
