 Back
BackThe Structure and Function of Large Biological Molecules
Study Guide - Smart Notes

Chapter 5: The Structure and Function of Large Biological Molecules
Introduction to Biomolecules
Large biological molecules, or macromolecules, are essential for life and include carbohydrates, lipids, proteins, and nucleic acids. These molecules are polymers built from monomers, and their structure determines their function in living organisms.
Monomers, Polymers, and Macromolecules
Definitions and Relationships
Monomer: A simple organic molecule that serves as a building block for polymers.
Polymer: A chain of monomers linked together by covalent bonds.
Macromolecule: A large molecule formed by the joining of many polymers; examples include carbohydrates, proteins, and nucleic acids.
Examples of monomer to macromolecule relationships:
Monosaccharide → Carbohydrate → Bread
Glycerol/Fatty acids → Lipids → Bee’s wax
Amino acid → Protein → Hemoglobin
Nucleotide → Nucleic acid → DNA
Synthesis and Breakdown of Polymers
Polymers are synthesized and broken down by specific chemical reactions:
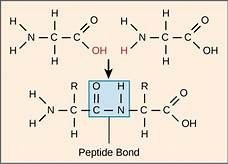
Dehydration Reaction: Joins monomers by removing a water molecule, forming a new bond.
Hydrolysis: Breaks bonds between monomers by adding a water molecule.
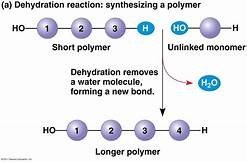
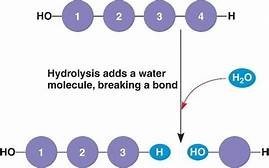
Carbohydrates: Fuel and Building Material
Structure and Classification
Carbohydrates are sugars and their polymers, serving as energy sources and structural materials.
General formula: H-C-OH; ratio of O to H is 1:2.
Aldose: Aldehyde sugar (C=O at the end).
Ketose: Ketone sugar (C=O within the skeleton).
Classified by carbon number: triose (3C), pentose (5C), hexose (6C).
Monosaccharide: Simple sugar (e.g., glucose, 6C).
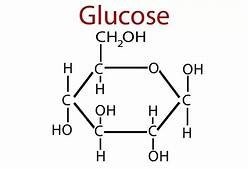
Glucose
Glucose is a common monosaccharide, often forming a ring structure for stability. It is central to cellular respiration and serves as a precursor for amino acids and fatty acids.
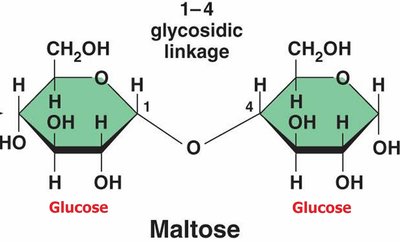
Disaccharides
Disaccharides are formed by joining two monosaccharides via a dehydration reaction, creating a glycosidic linkage.
Maltose: Glucose + Glucose
Sucrose: Glucose + Fructose
Lactose: Glucose + Galactose
Polysaccharides
Polysaccharides are large polymers of monosaccharides, serving storage and structural roles.
Starch: Storage in plants (in plastids such as chloroplasts).
Glycogen: Storage in animals (liver and muscle cells).
Cellulose: Structural component in plant cell walls; straight chains provide strength. Most animals cannot digest cellulose, but some (e.g., cows) have gut microbes that can.
Chitin: Structural polysaccharide in exoskeletons of arthropods.
Lipids: Energy Storage and Membranes
Types and Properties
Lipids are hydrophobic molecules, including fats, oils, phospholipids, and steroids.
Fats: Glycerol + 3 fatty acids; typically animal origin.
Oils: Plant origin; usually unsaturated.
Saturated fatty acids: No double bonds (e.g., lard, butter).
Unsaturated fatty acids: One or more double bonds (e.g., vegetable oils, fish oils).
Trans fats: Produced by hydrogenation; found in processed foods.
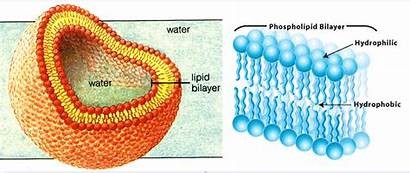
Phospholipids and Steroids
Phospholipids are major components of cell membranes, forming a bilayer with hydrophobic tails and hydrophilic heads. Steroids, such as cholesterol, have four fused rings and contribute to membrane integrity and hormone synthesis.
Proteins: Structure, Enzymes, and Functions
Amino Acids and Peptide Bonds
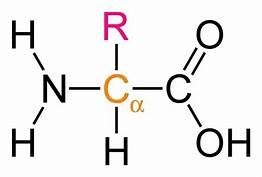
Proteins are polymers of amino acids, linked by peptide bonds. Each amino acid contains an amino group (NH2), a carboxyl group (COOH), a hydrogen atom, and a variable R group attached to a central carbon.
Protein Structure and Function
The function of a protein is determined by its structure, which is organized into four levels:
Primary structure: Sequence of amino acids.
Secondary structure: Alpha helix or beta sheet formed by hydrogen bonding.
Tertiary structure: Overall 3D folding driven by interactions among R groups.
Quaternary structure: Association of two or more polypeptide chains.
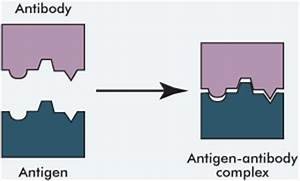
Protein shape is crucial for function, such as enzyme activity or antibody-antigen binding. Denaturation (loss of structure) can occur due to heat, pH changes, or salt concentration, leading to loss of function. Misfolding is implicated in diseases like cystic fibrosis, Alzheimer's, Parkinson's, and mad cow disease.
Nucleic Acids: Storage and Transmission of Genetic Information
Structure and Types
Nucleic acids (DNA and RNA) are polymers of nucleotides, each composed of a phosphate group, a pentose sugar, and a nitrogenous base. Nitrogenous bases are classified as purines (adenine, guanine) or pyrimidines (cytosine, thymine, uracil).
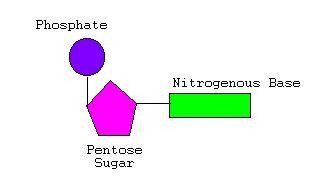
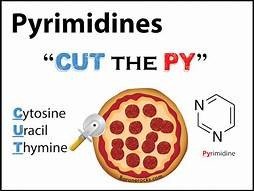
DNA vs. RNA
DNA | RNA | |
|---|---|---|
Sugar | Deoxyribose | Ribose |
Bases | Adenine, Guanine, Thymine, Cytosine | Adenine, Guanine, Uracil, Cytosine |
Strands | Double | Single |
Helix | Yes | No |
Summary Table: Organic Compounds
Macromolecule | Monomer | Function |
|---|---|---|
Proteins | Amino Acids | Enzymes, structural components (e.g., muscle proteins) |
Carbohydrates | Glucose | Energy storage (starch in plants, glycogen in animals), plant cell walls (cellulose) |
Lipids | Glycerol, fatty acids | Long-term energy storage, cell membrane structure |
Nucleic Acids | Nucleotides | Genetic material (DNA), protein synthesis (RNA) |
