 Back
BackThe Structure and Function of Large Biological Molecules: Proteins
Study Guide - Smart Notes

The Structure and Function of Large Biological Molecules
Macromolecules and Proteins
Macromolecules are the predominant organic molecules in cells, essential for life. Proteins are true polymers made of amino acids, connected by peptide bonds. The structure of a protein determines its function, making proteins central to cellular processes.
Macromolecules: Large molecules composed of thousands of atoms, including proteins, nucleic acids, carbohydrates, and lipids.
Proteins: Polymers of amino acids, performing most cellular work.
Peptide bond: A covalent bond formed by dehydration synthesis between two amino acids.
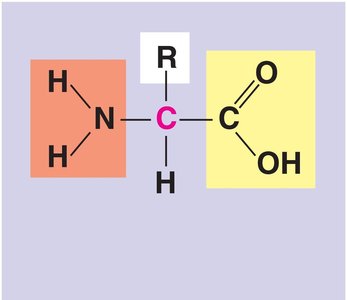
Amino acid: Organic molecule with both an amino group and a carboxyl group.

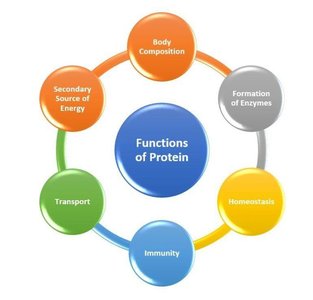
Functions of Proteins
Proteins perform diverse functions in cells, including catalysis, signaling, structural support, and defense. Each function is carried out by specialized protein types.
Enzymes: Catalyze biochemical reactions, creating and breaking down cell components.
Receptors: Recognize hormones and signaling molecules.
Adhesins: Connect cells to one another.
Messengers: Communicate within or outside the cell.
Defense: Recognize, target, and destroy pathogens.
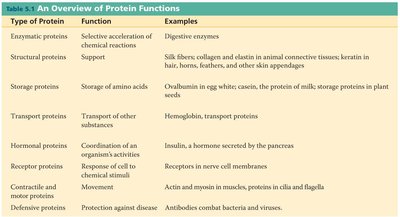
Overview of Protein Functions
Type of Protein | Function | Examples |
|---|---|---|
Enzymatic proteins | Selective acceleration of chemical reactions | Digestive enzymes |
Structural proteins | Support | Silk fibers, collagen, keratin |
Storage proteins | Storage of amino acids | Ovalbumin, casein |
Transport proteins | Transport of other substances | Hemoglobin, transport proteins |
Hormonal proteins | Coordination of activities | Insulin |
Receptor proteins | Response to stimuli | Receptors in nerve cell membranes |
Contractile and motor proteins | Movement | Actin, myosin |
Defensive proteins | Protection against disease | Antibodies |

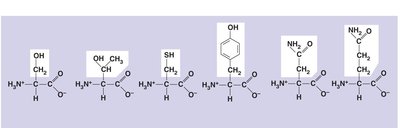
Amino Acids: Structure and Classification
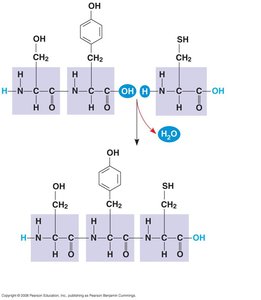
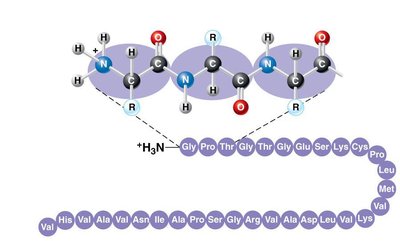
Amino acids are the monomers that make up proteins. Each amino acid contains an amino group, a carboxyl group, a central carbon, and a unique side chain (R group). Amino acids are linked by peptide bonds to form polypeptides.
Dipeptide: Two amino acids linked by a peptide bond.
Polypeptide: Long chain of amino acids linked by peptide bonds.
Peptide bond formation: Occurs via dehydration synthesis.
Classification of Amino Acids
Amino acids are classified based on the properties of their side chains:
Nonpolar: Glycine, Alanine, Valine, Leucine, Isoleucine, Methionine, Phenylalanine, Tryptophan, Proline
Polar: Asparagine, Glutamine, Serine, Threonine, Cysteine, Tyrosine
Acidic: Aspartic acid, Glutamic acid
Basic: Arginine, Histidine, Lysine
Levels of Protein Structure
Proteins have four levels of structure, each contributing to their overall shape and function.
Primary Structure: Linear sequence of amino acids linked by peptide bonds.
Secondary Structure: Regular patterns such as alpha helices and beta sheets, stabilized by hydrogen bonds.
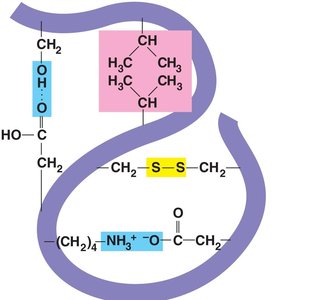
Tertiary Structure: Three-dimensional folding due to interactions between R groups, including hydrophobic interactions, ionic bonds, hydrogen bonds, and disulfide bridges.
Quaternary Structure: Association of multiple polypeptide chains.

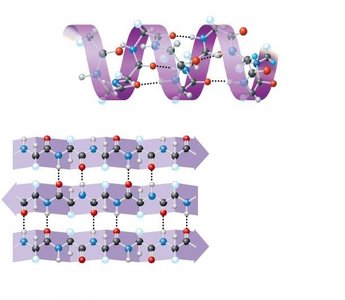
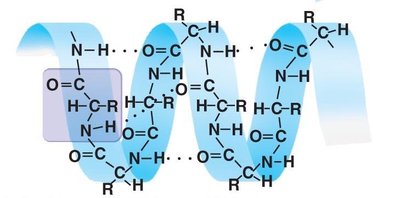
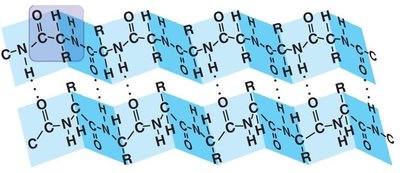
Forces Stabilizing Protein Structure
Tertiary structure is stabilized by several types of interactions:
Hydrophobic interactions and van der Waals forces
Disulfide bridges (covalent bonds between cysteine residues)
Ionic bonds (between charged side chains)
Hydrogen bonds (between polar side chains)
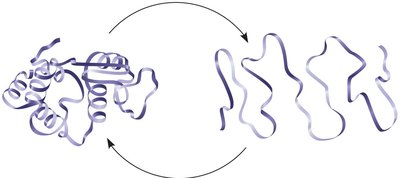
Protein Denaturation and Renaturation
If the environment of a protein changes drastically (e.g., temperature, salinity, pH), the protein can lose its shape and function, a process called denaturation. Some proteins can regain their structure through renaturation.
Denaturation: Loss of protein structure and function due to environmental changes.
Renaturation: Restoration of protein structure under favorable conditions.
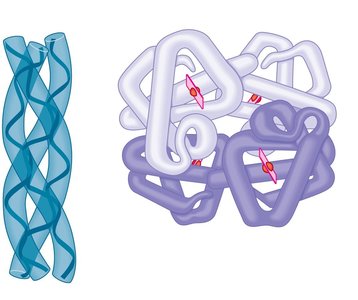
Quaternary Structure
Quaternary structure arises when several polypeptides interact to form a functional protein complex. Examples include collagen and hemoglobin.
Collagen: Structural protein with three polypeptide chains.
Hemoglobin: Oxygen transport protein with four polypeptide chains and heme groups.
 Additional info: The notes have been expanded with academic context to ensure completeness and clarity for exam preparation.
Additional info: The notes have been expanded with academic context to ensure completeness and clarity for exam preparation.
