 Back
BackThe Working Cell: Energy, Enzymes, and Membrane Function
Study Guide - Smart Notes

The Working Cell
Energy & the Cell
The cell is a dynamic unit that requires energy to perform work, including growth, maintenance, and reproduction. Energy exists in two primary forms: kinetic energy (energy of motion) and potential energy (stored energy, often in chemical bonds).
Kinetic energy: The energy associated with moving objects.
Potential energy: Energy stored due to an object's position or structure, including chemical energy in molecules.
Metabolism: The sum of all chemical reactions in an organism, organized into metabolic pathways that build or break down molecules.
Laws of Thermodynamics
Biological systems obey the laws of thermodynamics:
First Law: Energy cannot be created or destroyed, only transformed.
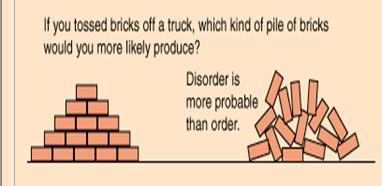
Second Law: Energy transformations increase disorder (entropy), with some energy lost as heat.
Energy Transformations in Cells
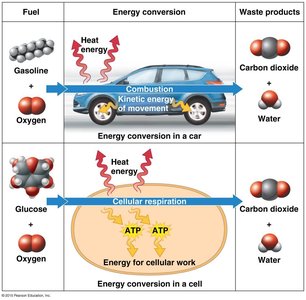
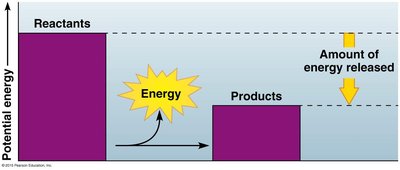
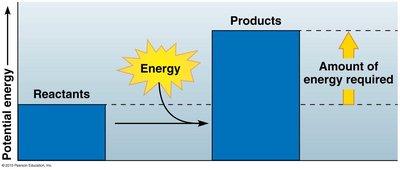
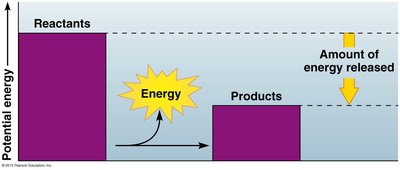
Cells act as chemical factories, transforming energy through thousands of reactions. Some reactions release energy (exergonic), while others require energy (endergonic).
Exergonic reactions: Release energy, e.g., cellular respiration.
Endergonic reactions: Require energy input, e.g., photosynthesis.
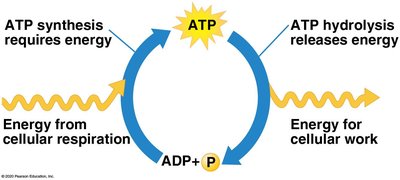
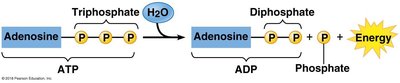
ATP: The Energy Currency of the Cell
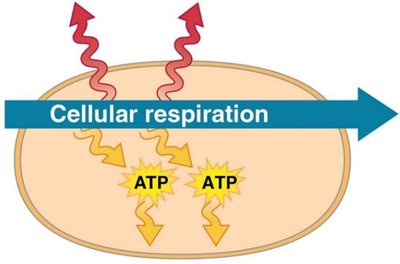
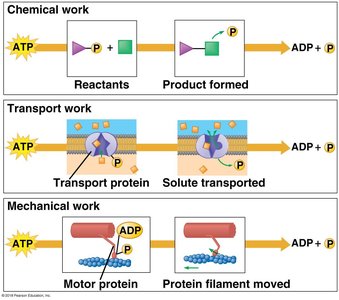
ATP (adenosine triphosphate) is the primary energy carrier in cells. Hydrolysis of ATP releases energy by transferring a phosphate group to another molecule (phosphorylation), powering cellular work.
ATP cycle: Energy from exergonic reactions (e.g., glucose breakdown) is used to regenerate ATP from ADP.
Cellular work: ATP powers chemical, transport, and mechanical work.
How Enzymes Function
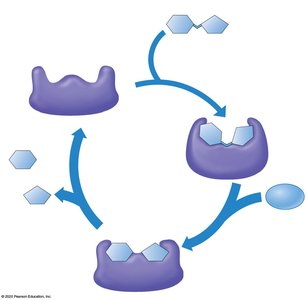
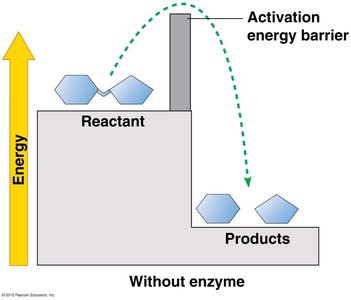
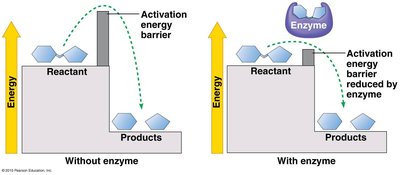
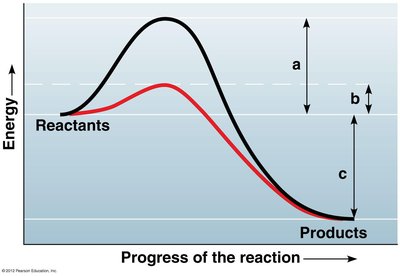
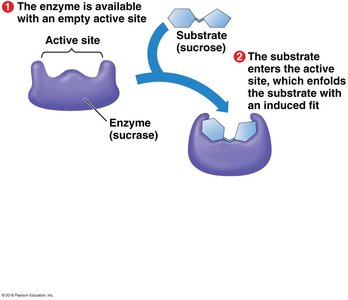
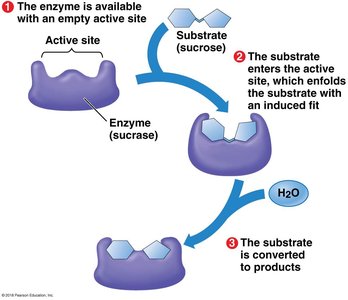
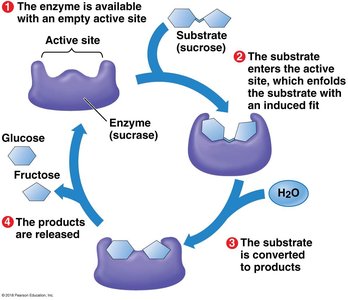
Enzymes are biological catalysts that speed up chemical reactions by lowering the activation energy barrier. Most enzymes are proteins, and their shape determines their specificity for substrates.
Activation energy (EA): The energy required to start a reaction.
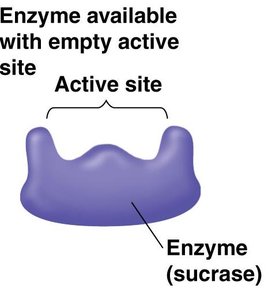
Enzyme specificity: The active site of an enzyme fits only specific substrate molecules.
Enzyme-substrate complex: The substrate binds to the enzyme's active site, forming a temporary association.

Factors Affecting Enzyme Activity
Enzyme activity is influenced by several factors:
Substrate concentration: Higher substrate levels increase reaction rate up to a point.
Enzyme concentration: More enzyme increases reaction rate.
Temperature: Optimal temperature increases reaction rate; extreme temperatures denature enzymes.
pH: Most enzymes function best near neutral pH.
Tables: Enzyme and Substrate Concentration Effects
These tables summarize the relationship between enzyme/substrate concentration and reaction rate.
Lactose conc. (%) | Enzyme conc. (%) | Reaction rate (g/min) |
|---|---|---|
10 | 0 | 0 |
10 | 1 | 25 |
10 | 2 | 50 |
10 | 4 | 100 |
10 | 8 | 200 |
Effect of enzyme concentration: Increasing enzyme concentration increases reaction rate.
Lactose conc. (%) | Enzyme conc. (%) | Reaction rate (g/min) |
|---|---|---|
0 | 2 | 0 |
5 | 2 | 25 |
10 | 2 | 50 |
20 | 2 | 65 |
30 | 2 | 65 |
Effect of substrate concentration: Increasing substrate concentration increases reaction rate up to a saturation point.
Cofactors and Coenzymes
Enzymes often require non-protein helpers called cofactors (inorganic, e.g., zinc, iron, copper) or coenzymes (organic, e.g., vitamins) to function.
Enzyme Inhibition
Enzyme activity can be regulated by inhibitors:
Competitive inhibitors: Bind to the active site, blocking substrate binding.
Noncompetitive inhibitors: Bind elsewhere, changing the enzyme's shape and reducing activity.
Feedback inhibition: The product of a pathway inhibits an enzyme earlier in the pathway.
Applications
Many drugs, pesticides, and poisons act as enzyme inhibitors, including antibiotics, blood pressure medicines, and protease inhibitors for HIV.
Membrane Structure and Function
Cell membranes are composed of a phospholipid bilayer with embedded proteins, forming a fluid mosaic structure. Membrane proteins perform various functions, including transport, signaling, and structural support.

Diffusion and Osmosis
Diffusion is the movement of particles from high to low concentration, driven by random motion. Osmosis is the diffusion of water across a selectively permeable membrane.
Passive transport: Diffusion across membranes without energy input.
Tonicity: The ability of a solution to cause a cell to gain or lose water, depending on solute concentration.
Osmoregulation: The control of water balance, crucial for cell survival.
Facilitated Diffusion and Active Transport
Polar or charged substances cross membranes via facilitated diffusion (using transport proteins, no energy required) or active transport (energy required to move substances against their concentration gradient).
Bulk Transport: Exocytosis and Endocytosis
Cells move large molecules via exocytosis (export) and endocytosis (import), using vesicles that fuse with the membrane.
Phagocytosis: Engulfment of particles by the cell.
Receptor-mediated endocytosis: Specific uptake of molecules using membrane receptors.
Summary: The working cell relies on energy transformations, enzyme activity, and membrane transport to sustain life. Understanding these processes is fundamental to cell biology.
