 Back
BackUnit 1: Science Practices & The Chemistry of Life – AP Biology Study Notes
Study Guide - Smart Notes

Science Practices in Biology
Overview of Science Practices
In AP Biology, students develop six core science practices that are essential for understanding and applying biological concepts. Mastery of these practices is crucial for success in laboratory work, assessments, and the AP Exam.
Concept Explanation: Ability to explain biological concepts, processes, and models in written form.
Visual Representations: Skill in analyzing diagrams, mathematical models, and flow charts related to biological processes.
Questions and Methods: Formulating testable questions, stating hypotheses, identifying variables and controls, collecting data, and proposing further investigations.
Representing and Describing Data: Constructing and interpreting graphs, charts, and plots; identifying trends and relationships in data.
Statistical Tests and Data Analysis: Performing calculations (means, rates, ratios, percentages), using confidence intervals and error bars, conducting Chi-square tests, and evaluating hypotheses with data.
Argumentation: Making scientific claims, supporting them with evidence, connecting claims to theories, and predicting outcomes in biological systems.
The Chemistry of Life
Topic 1: Water and Life
Water is fundamental to life due to its unique chemical and physical properties, which arise from its molecular structure and hydrogen bonding.
Polarity of Water: Water is a polar molecule because of the unequal sharing of electrons between oxygen and hydrogen atoms, resulting in a partial negative charge near the oxygen and a partial positive charge near the hydrogens.
Hydrogen Bonds: The polarity of water molecules allows them to form hydrogen bonds with each other, which are weak individually but strong collectively, giving water its unique properties.
Specific Heat Capacity and Heat of Vaporization: Water has a high specific heat capacity and heat of vaporization compared to most substances. This allows water to buffer temperature changes in organisms and environments, stabilizing biological systems.
Cohesion, Adhesion, and Surface Tension: Cohesion is the attraction between water molecules, leading to surface tension. Adhesion is the attraction between water molecules and other substances, aiding in capillary action.
Transport in Plants: Water moves through plant tissues via xylem, driven by transpiration, cohesion, and adhesion.
Transpiration: The process by which water evaporates from plant leaves, creating a negative pressure that pulls water upward from roots. Environmental factors such as humidity, temperature, and wind influence transpiration rates.
Additional info: The high heat of vaporization of water helps organisms cool off through sweating and transpiration.
Topic 2: The Elements of Life
Living organisms are composed of a limited set of chemical elements, with carbon, hydrogen, oxygen, nitrogen, phosphorus, and sulfur being the most important for biological macromolecules.
Major Elements: The primary elements in living organisms are carbon (C), hydrogen (H), oxygen (O), nitrogen (N), phosphorus (P), and sulfur (S).
Isotopes: Atoms of the same element with different numbers of neutrons. Radioactive isotopes are used as tracers in biological research (e.g., tracking DNA synthesis).
Role of Carbon: Carbon's ability to form four covalent bonds makes it the backbone of organic molecules, allowing for complex structures.
Nitrogen: Found in amino acids, proteins, and nucleic acids.
Phosphorus: Present in nucleic acids (DNA, RNA) and ATP.
Sulfur: Found in some amino acids (e.g., cysteine, methionine) and vitamins.
Topic 3: Introduction to Biological Macromolecules
Biological macromolecules are large, complex molecules essential for life. They are built from smaller subunits through specific chemical reactions.
Chemical Functional Groups: Groups of atoms (e.g., hydroxyl, carboxyl, amino, phosphate) that confer specific properties and reactivity to molecules.
Monomers and Polymers: Monomers are small subunits (e.g., amino acids, nucleotides, monosaccharides) that join to form polymers (e.g., proteins, nucleic acids, polysaccharides).
Dehydration Synthesis (Condensation): Chemical reaction that joins monomers by removing a water molecule, forming covalent bonds.
Hydrolysis: The reverse reaction, breaking polymers into monomers by adding water.
Sequence and Properties: The sequence of subcomponents (e.g., amino acids in a protein) determines the molecule's properties and function.
Additional info: Enzymes catalyze both dehydration synthesis and hydrolysis reactions in cells.
Topic 4: Types of Macromolecules
There are four major classes of biological macromolecules: carbohydrates, lipids, nucleic acids, and proteins. Each has distinct structures and functions.
Carbohydrates
Structure: Composed of carbon, hydrogen, and oxygen (typically in a 1:2:1 ratio). Monomers are monosaccharides (e.g., glucose); polymers include starch, glycogen, and cellulose.
Function: Serve as energy sources (glucose), energy storage (starch in plants, glycogen in animals), and structural components (cellulose in plant cell walls).
Structural Differences: The arrangement of monomers and type of glycosidic bonds result in different properties (e.g., digestibility of starch vs. cellulose).
Lipids
Structure: Mostly hydrophobic molecules, including fats, phospholipids, and steroids. Fats are composed of glycerol and fatty acids.
Saturated vs. Unsaturated Fatty Acids: Saturated fatty acids have no double bonds (solid at room temperature); unsaturated fatty acids have one or more double bonds (liquid at room temperature).
Functions: Energy storage (fats), membrane structure (phospholipids), signaling (steroids), and insulation.
Phospholipids: Major component of cell membranes, forming bilayers due to their amphipathic nature (hydrophilic head, hydrophobic tails).
Nucleic Acids
Structure: Polymers of nucleotides, each consisting of a phosphate group, a five-carbon sugar (ribose or deoxyribose), and a nitrogenous base.
Polymerization: Nucleotides are linked by phosphodiester bonds between the 3' hydroxyl and 5' phosphate groups, giving directionality (5' to 3').
DNA vs. RNA: DNA contains deoxyribose and is double-stranded; RNA contains ribose and is usually single-stranded. DNA stores genetic information; RNA is involved in protein synthesis and regulation.
Double Helix: DNA strands are held together by hydrogen bonds between complementary bases (A-T, G-C).
Example: The image below shows the structure of a nucleotide, the monomer of nucleic acids.
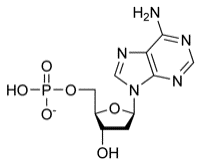
Proteins
Structure: Polymers of amino acids linked by peptide bonds. Each amino acid has an amino group, carboxyl group, hydrogen atom, and a variable R group.
Directionality: Proteins have an N-terminus (amino end) and a C-terminus (carboxyl end).
R Groups: Amino acids differ in their R groups, which can be nonpolar, polar, or charged, affecting protein structure and function.
Levels of Structure:
Primary: Sequence of amino acids.
Secondary: Local folding (α-helices, β-sheets) stabilized by hydrogen bonds.
Tertiary: Overall 3D shape due to interactions among R groups.
Quaternary: Association of multiple polypeptide chains.
Function: Enzymes, structural support, transport, signaling, immune response, and more.
Additional info: Protein function is highly dependent on its three-dimensional structure, which can be altered by changes in pH, temperature, or mutations.
