 Back
BackWater and Carbon: The Chemical Basis of Life – Study Notes
Study Guide - Smart Notes

Chapter 2: Water and Carbon – The Chemical Basis of Life
Introduction to Chemical Evolution
Chemical evolution is the leading explanation for the origin of life on Earth. It describes the process by which simple carbon-containing molecules formed increasingly complex substances, eventually leading to molecules capable of self-replication. This transition marked the switch from chemical to biological evolution, fulfilling the five fundamental characteristics of life.
Chemical evolution: Formation of complex molecules from simpler ones.
Biological evolution: Driven by natural selection, where replicating molecules became metabolically active and acquired membranes.
Five characteristics of life: Cellular organization, metabolism, homeostasis, growth, and reproduction.
Properties of Water and the Early Oceans
Water is the foundation of life, making up approximately 75% of a cell's mass. Its unique chemical properties make it an excellent solvent, facilitating the chemical reactions necessary for life.
Solvent: A substance that dissolves solutes to form a solution.
Water as a solvent: Enables reactants to interact more readily, increasing the likelihood of chemical reactions.
Water’s Structure and Its Unique Properties
Water’s molecular structure is responsible for its remarkable properties. It is a small molecule with a bent shape and highly polar covalent bonds, resulting in overall polarity.
Polarity: Oxygen atoms carry a partial negative charge, while hydrogen atoms carry a partial positive charge.
Hydrogen bonds: Weak electrical interactions between the partial charges on water molecules.
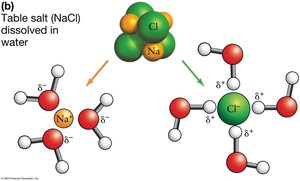
Water as an Efficient Solvent
Water’s polarity allows it to dissolve many substances, especially ions and polar molecules. Hydrophilic molecules interact with water’s partial charges, while hydrophobic molecules do not dissolve and instead cluster together, stabilized by van der Waals interactions.
Hydrophilic: "Water-loving"; ions and polar molecules that dissolve in water.
Hydrophobic: "Water-fearing"; nonpolar molecules that do not dissolve in water.
Hydrophobic interaction: Tendency of nonpolar molecules to aggregate in aqueous solution.
Cohesion, Adhesion, and Surface Tension
Water molecules exhibit cohesion (attraction between like molecules) and adhesion (attraction between unlike molecules). These properties contribute to surface tension, allowing water to resist external forces and behave as if covered by an elastic membrane.
Cohesion: Water molecules stick together due to hydrogen bonding.
Adhesion: Water molecules adhere to surfaces with polar or charged groups.
Surface tension: Cohesive force at the surface of water, making it act like a stretched membrane.

Density of Water: Liquid vs. Solid
Unlike most substances, water is denser as a liquid than as a solid. As water freezes, it forms an open crystal structure due to hydrogen bonding, causing ice to float and insulate aquatic environments.
Crystal structure: Hydrogen bonds hold water molecules apart in ice.
Ecological impact: Ice insulates water below, protecting aquatic life in cold climates.
Water’s Capacity for Absorbing Energy
Water has a high specific heat and heat of vaporization, meaning it can absorb large amounts of energy before changing temperature or state. This property is crucial for temperature regulation in organisms and environments.
Specific heat: Energy required to raise the temperature of 1 gram of a substance by 1°C.
Heat of vaporization: Energy required to convert 1 gram of liquid to gas.
Biological relevance: Sweating cools organisms by utilizing water’s high heat of vaporization.
The Role of Water in Acid–Base Chemical Reactions
Water participates in acid-base reactions, influencing the concentration of protons (hydrogen ions) in solution. Acids donate protons, increasing hydronium ion concentration, while bases accept protons, decreasing it.
Acid: Substance that increases proton concentration in solution.
Base: Substance that decreases proton concentration in solution.
The pH Scale
The pH scale is a logarithmic measure of hydrogen ion concentration, indicating whether a solution is acidic, basic, or neutral. Buffers help maintain stable pH in biological systems, ensuring homeostasis.
pH:
Acidic: pH < 7
Basic: pH > 7
Neutral: pH = 7 (typical of intracellular fluid)
Buffer: Substance that minimizes changes in pH.
Chemical Reactions, Energy, and Chemical Evolution
Chemical evolution may have begun in the atmosphere or deep-sea hydrothermal vents, where simple molecules with high free energy were exposed to kinetic energy, leading to the formation of complex organic compounds.
Atmosphere: Rich in volcanic gases (water vapor, CO2, nitrogen).
Hydrothermal vents: Hot rocks, reactive gases, and minerals with metals.
Spontaneity of Chemical Reactions
Chemical reactions are spontaneous if they proceed without continuous external energy input. Spontaneity is determined by two factors: lower potential energy in products and increased disorder (entropy).
Potential energy: Energy stored in chemical bonds.
Entropy: Measure of disorder in a system.
Investigating Chemical Evolution
Stanley Miller’s 1953 experiment demonstrated that complex organic molecules, such as amino acids, could be synthesized from simple molecules under conditions simulating early Earth. This supported the theory of chemical evolution.
Miller’s experiment: Used heat and electrical sparks to simulate early Earth conditions.
Result: Formation of amino acids, the building blocks of proteins.
Life is Carbon-Based
Carbon is the backbone of organic molecules, capable of forming four covalent bonds and a limitless array of molecular shapes. Organic compounds contain carbon bonded to other elements, enabling diverse chemical behaviors.
Organic compounds: Molecules with carbon bonded to hydrogen, oxygen, nitrogen, phosphorus, or sulfur.
Covalent bonds: Carbon forms single and double bonds, allowing complex structures.
Functional Groups in Organic Molecules
Functional groups are specific groups of atoms within molecules that determine their chemical behavior. Key functional groups include amino, carboxyl, carbonyl, hydroxyl, phosphate, and sulfhydryl.
Amino group (–NH2): Acts as a base, attracts protons.
Carboxyl group (–COOH): Acts as an acid, donates protons.
Carbonyl group (–CO): Links molecules into more complex compounds.
Hydroxyl group (–OH): Acts as a weak acid.
Phosphate group (–PO4): Carries two negative charges.
Sulfhydryl group (–SH): Forms disulfide bonds, stabilizing protein structure.
Assembly of Small Organic Molecules into Macromolecules
Macromolecules are large molecules formed by joining smaller subunits (monomers) through polymerization. Condensation (dehydration) reactions link monomers, releasing water, while hydrolysis breaks polymers into monomers, increasing entropy.
Macromolecule: Large molecule made of monomers.
Polymerization: Process of joining monomers to form polymers.
Condensation reaction: Monomers join, releasing water.
Hydrolysis: Water breaks bonds, releasing monomers.
Equilibrium: Favors free monomers over polymers unless monomer concentration is high.
Additional info: Macromolecules such as proteins, nucleic acids, and carbohydrates may have polymerized early in chemical evolution, forming the basis for life.
